| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2021-09-14 15:39:00 UTC |
|---|
| HMDB ID | HMDB0001132 |
|---|
| Secondary Accession Numbers | - HMDB0059646
- HMDB01132
- HMDB59646
|
|---|
| Metabolite Identification |
|---|
| Common Name | Nicotinic acid mononucleotide |
|---|
| Description | Nicotinic acid mononucleotide, also known as nicotinate ribonucleotide, belongs to the class of organic compounds known as nicotinic acid nucleotides. These are pyridine nucleotides in which the pyridine base is nicotinic acid or a derivative thereof. Nicotinic acid mononucleotide is an extremely weak basic (essentially neutral) compound (based on its pKa). Nicotinic acid mononucleotide an intermediate in the cofactor biosynthesis and the nicotinate and nicotinamide metabolism pathways. It is a substrate for nicotinamide riboside kinase, ectonucleotide pyrophosphatase/phosphodiesterase, nicotinamide mononucleotide adenylyltransferase, 5'-nucleotidase, nicotinate-nucleotide pyrophosphorylase, and 5'(3')-deoxyribonucleotidase. |
|---|
| Structure | O[C@H]1[C@@H](O)[C@@H](O[C@@H]1COP(O)(O)=O)[N+]1=CC=CC(=C1)C(O)=O InChI=1S/C11H14NO9P/c13-8-7(5-20-22(17,18)19)21-10(9(8)14)12-3-1-2-6(4-12)11(15)16/h1-4,7-10,13-14H,5H2,(H2-,15,16,17,18,19)/p+1/t7-,8-,9-,10-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| beta-Nicotinate D-ribonucleotide | ChEBI | | Nicotinate ribonucleotide | ChEBI | | Nicotinic acid ribonucleotide | ChEBI | | b-Nicotinate D-ribonucleotide | Generator | | b-Nicotinic acid D-ribonucleotide | Generator | | beta-Nicotinic acid D-ribonucleotide | Generator | | β-Nicotinate D-ribonucleotide | Generator | | β-Nicotinic acid D-ribonucleotide | Generator | | Nicotinic acid D-ribonucleotide | Generator | | Nicotinate D-ribonucleotide | ChEBI | | Nicotinate b-D-ribonucleotide | Generator | | Nicotinate β-D-ribonucleotide | Generator | | Nicotinic acid b-D-ribonucleotide | Generator | | Nicotinic acid beta-D-ribonucleotide | Generator | | Nicotinic acid β-D-ribonucleotide | Generator | | Nicotinate mononucleotide | MeSH | | Nicotinic acid mononucleotide | MeSH | | Deamido nicotinamide ribonucleotide | HMDB | | Deamido-NMN | HMDB | | Nicotinic acid ribotide | HMDB | | Nicotinic mononucleotide | HMDB | | beta-Nicotinic acid mononucleotide | HMDB | | β-Nicotinic acid mononucleotide | HMDB |
| Show more...
|---|
| Chemical Formula | C11H15NO9P |
|---|
| Average Molecular Weight | 336.2119 |
|---|
| Monoisotopic Molecular Weight | 336.048442595 |
|---|
| IUPAC Name | 3-carboxy-1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]-1lambda5-pyridin-1-ylium |
|---|
| Traditional Name | 3-carboxy-1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]-1lambda5-pyridin-1-ylium |
|---|
| CAS Registry Number | 321-02-8 |
|---|
| SMILES | O[C@H]1[C@@H](O)[C@@H](O[C@@H]1COP(O)(O)=O)[N+]1=CC=CC(=C1)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C11H14NO9P/c13-8-7(5-20-22(17,18)19)21-10(9(8)14)12-3-1-2-6(4-12)11(15)16/h1-4,7-10,13-14H,5H2,(H2-,15,16,17,18,19)/p+1/t7-,8-,9-,10-/m1/s1 |
|---|
| InChI Key | JOUIQRNQJGXQDC-ZYUZMQFOSA-O |
|---|
| Chemical Taxonomy |
|---|
| Description | This compound belongs to the class of organic compounds known as nicotinic acid nucleotides. These are pyridine nucleotides, in which the pyridine base is nicotinic acid or a derivative thereof. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | Pyridine nucleotides |
|---|
| Sub Class | Nicotinic acid nucleotides |
|---|
| Direct Parent | Nicotinic acid nucleotides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Nicotinic-acid-nucleotide
- Pentose phosphate
- Pentose-5-phosphate
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Pentose monosaccharide
- Pyridine carboxylic acid
- Pyridine carboxylic acid or derivatives
- Monoalkyl phosphate
- Monosaccharide
- Organic phosphoric acid derivative
- Phosphoric acid ester
- Pyridine
- Pyridinium
- Alkyl phosphate
- Vinylogous amide
- Tetrahydrofuran
- Heteroaromatic compound
- Secondary alcohol
- 1,2-diol
- Carboxylic acid derivative
- Organoheterocyclic compound
- Azacycle
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Oxacycle
- Organopnictogen compound
- Organic nitrogen compound
- Organic oxygen compound
- Organic oxide
- Alcohol
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic cation
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | | Show more...
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| Nicotinic acid mononucleotide,1TMS,isomer #1 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O | 2792.1 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,1TMS,isomer #2 | C[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](COP(=O)(O)O)O[C@H]1[N+]1=CC=CC(C(=O)O)=C1 | 2823.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,1TMS,isomer #3 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O)[C@H]2O)=C1 | 2764.6 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,1TMS,isomer #4 | C[Si](C)(C)OP(=O)(O)OC[C@H]1O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@H](O)[C@@H]1O | 2943.5 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O[Si](C)(C)C)[C@H]2O)=C1 | 2664.2 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TMS,isomer #2 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C | 2711.0 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TMS,isomer #3 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O[Si](C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O | 2825.2 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TMS,isomer #4 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O)[C@H]2O[Si](C)(C)C)=C1 | 2687.6 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TMS,isomer #5 | C[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](COP(=O)(O)O[Si](C)(C)C)O[C@H]1[N+]1=CC=CC(C(=O)O)=C1 | 2858.2 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TMS,isomer #6 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C)[C@@H](O)[C@H]2O)=C1 | 2797.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TMS,isomer #7 | C[Si](C)(C)OP(=O)(OC[C@H]1O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@H](O)[C@@H]1O)O[Si](C)(C)C | 2914.9 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O[Si](C)(C)C)[C@H]2O[Si](C)(C)C)=C1 | 2636.6 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TMS,isomer #2 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O)=C1 | 2761.3 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TMS,isomer #3 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O[Si](C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C | 2780.6 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TMS,isomer #4 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O | 2857.0 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TMS,isomer #5 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C)=C1 | 2783.8 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TMS,isomer #6 | C[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)O[C@H]1[N+]1=CC=CC(C(=O)O)=C1 | 2877.7 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TMS,isomer #7 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O)[C@H]2O)=C1 | 2825.0 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O[Si](C)(C)C)=C1 | 2776.6 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O[Si](C)(C)C)=C1 | 2861.4 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O[Si](C)(C)C)=C1 | 3285.8 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #2 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O)=C1 | 2816.9 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #2 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O)=C1 | 2867.4 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #2 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O)=C1 | 3136.5 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #3 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C | 2851.9 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #3 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C | 2946.7 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #3 | C[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C | 3120.2 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #4 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C)=C1 | 2830.8 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #4 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C)=C1 | 2879.9 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TMS,isomer #4 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C)=C1 | 3169.6 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,5TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O[Si](C)(C)C)=C1 | 2826.8 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,5TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O[Si](C)(C)C)=C1 | 2865.2 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,5TMS,isomer #1 | C[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@H]2O[Si](C)(C)C)=C1 | 3008.3 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O | 3061.1 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](COP(=O)(O)O)O[C@H]1[N+]1=CC=CC(C(=O)O)=C1 | 3079.0 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,1TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O)[C@H]2O)=C1 | 3056.5 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,1TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OP(=O)(O)OC[C@H]1O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@H](O)[C@@H]1O | 3184.5 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O)=C1 | 3173.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C(C)(C)C | 3192.2 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O | 3332.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3187.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)O[C@H]1[N+]1=CC=CC(C(=O)O)=C1 | 3343.9 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@H]2O)=C1 | 3307.9 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,2TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OP(=O)(OC[C@H]1O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@H](O)[C@@H]1O)O[Si](C)(C)C(C)(C)C | 3394.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3344.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O)=C1 | 3474.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C(C)(C)C | 3472.8 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O | 3544.9 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3477.5 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)O[C@H]1[N+]1=CC=CC(C(=O)O)=C1 | 3545.8 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,3TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@H]2O)=C1 | 3528.8 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3641.8 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3581.3 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O)O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3564.4 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O)=C1 | 3680.9 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O)=C1 | 3561.1 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H]2O)=C1 | 3455.0 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C(C)(C)C | 3682.5 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C(C)(C)C | 3668.6 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)O[C@@H]([N+]2=CC=CC(C(=O)O)=C2)[C@@H]1O[Si](C)(C)C(C)(C)C | 3438.9 | Standard polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3678.4 | Semi standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3579.4 | Standard non polar | 33892256 | | Nicotinic acid mononucleotide,4TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)C1=CC=C[N+]([C@@H]2O[C@H](COP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@H]2O[Si](C)(C)C(C)(C)C)=C1 | 3486.7 | Standard polar | 33892256 |
| Show more...
|---|
| Spectra |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid mononucleotide GC-MS (Non-derivatized) - 70eV, Positive | splash10-00kb-9704000000-b4f3d91df4a5fb88cc5e | 2016-09-22 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid mononucleotide GC-MS (3 TMS) - 70eV, Positive | splash10-000b-9221310000-f2425a1cc55147b91d2b | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid mononucleotide GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 10V, Positive-QTOF | splash10-000i-1009000000-227e6a4729b96a705fa4 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 20V, Positive-QTOF | splash10-002b-5239000000-223d08e3fd97af58d374 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 40V, Positive-QTOF | splash10-0007-9500000000-db23a80f58b7ac9781fd | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 10V, Negative-QTOF | splash10-000i-1009000000-f16974a59d3ff46358d6 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 20V, Negative-QTOF | splash10-002b-9001000000-ca67d776dd7d3f5d4650 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 40V, Negative-QTOF | splash10-004i-9000000000-5808f9bb378f2d60cd9b | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 10V, Positive-QTOF | splash10-000i-0479000000-61da4c1926ec7950c051 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 20V, Positive-QTOF | splash10-00si-5950000000-2c01c6fd1632660b38c7 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid mononucleotide 40V, Positive-QTOF | splash10-001i-9500000000-3dbb0f51364659db892e | 2021-09-22 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum |
| Show more...
|---|
| Biological Properties |
|---|
| Cellular Locations | - Cytoplasm
- Mitochondria
- Nucleus
|
|---|
| Biospecimen Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
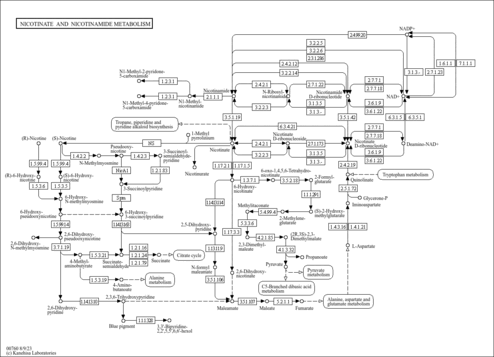
| Pathways | |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | None |
|---|
| Associated OMIM IDs | None |
|---|
| External Links |
|---|
| DrugBank ID | DB02382 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | FDB022444 |
|---|
| KNApSAcK ID | C00007447 |
|---|
| Chemspider ID | 108822 |
|---|
| KEGG Compound ID | C01185 |
|---|
| BioCyc ID | NICOTINATE_NUCLEOTIDE |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PubChem Compound | 121992 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 15763 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | Not Available |
|---|
| MarkerDB ID | Not Available |
|---|
| Good Scents ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| Material Safety Data Sheet (MSDS) | Not Available |
|---|
| General References | Not Available |
|---|