| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2008-09-12 01:12:18 UTC |
|---|
| Update Date | 2022-11-30 19:02:48 UTC |
|---|
| HMDB ID | HMDB0006968 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | CDP-DG(16:0/16:0) |
|---|
| Description | CDP-DG(16:0/16:0) is a cytidine diphosphate diacylglycerol or CDP-diacylglycerol. CDP-diacylglycerol (CDP-DG) is an important branchpoint intermediate in eukaryotic phospholipid biosynthesis and could be a key regulatory molecule in phospholipid metabolism. It is a glycerophospholipid in which a cytidine diphosphate moiety occupies a glycerol substitution site. As is the case with diacylglycerols, CDP-diacylglycerols can have many different combinations of fatty acids of varying lengths and saturation attached at the C-1 and C-2 positions. Fatty acids containing 16, 18 and 20 carbons are the most common. CDP-DG(16:0/16:0), in particular, consists of one chain of palmitic acid at the C-1 position and one chain of palmitic acid at the C-2 position. The palmitic acid moiety is derived from fish oils, milk fats, vegetable oils and animal fats, while the palmitic acid moiety is derived from fish oils, milk fats, vegetable oils and animal fats. CDP-diacylglycerols are intermediates in the synthesis of phosphatidylglycerols (PG, PC, PS, PI), which is catalyzed by CDP-diacyl synthase, synthase, phosphatidylglycerolphosphate (PGP) synthase, phosphatidylinositol (PI) synthase, and phosphatidylserine (PS) synthase. Cytidine diphosphate diacylglycerols are rarely noticed in analyses of lipid compositions of tissues, as they are present is such small amounts, perhaps only 0.05% or so of the total phospholipids. |
|---|
| Structure | [H][C@@](COC(=O)CCCCCCCCCCCCCCC)(COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H](C(O)[C@H]1O)N1C=CC(N)=NC1=O)OC(=O)CCCCCCCCCCCCCCC InChI=1S/C44H81N3O15P2/c1-3-5-7-9-11-13-15-17-19-21-23-25-27-29-39(48)57-33-36(60-40(49)30-28-26-24-22-20-18-16-14-12-10-8-6-4-2)34-58-63(53,54)62-64(55,56)59-35-37-41(50)42(51)43(61-37)47-32-31-38(45)46-44(47)52/h31-32,36-37,41-43,50-51H,3-30,33-35H2,1-2H3,(H,53,54)(H,55,56)(H2,45,46,52)/t36-,37-,41+,42?,43-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 1,2-Dihexadecanoyl-rac-glycero-3-CDP | HMDB | | 1,2-Dipalmitoyl-rac-glycero-3-cytidine-5'-diphosphate | HMDB | | CDP-DG(32:0) | HMDB | | CDP-Diacylglycerol(16:0/16:0) | HMDB | | CDP-Diacylglycerol(32:0) | HMDB |
|
|---|
| Chemical Formula | C44H81N3O15P2 |
|---|
| Average Molecular Weight | 954.086 |
|---|
| Monoisotopic Molecular Weight | 953.514292916 |
|---|
| IUPAC Name | {[(2R,3R,5R)-5-(4-amino-2-oxo-1,2-dihydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}({[(2R)-2,3-bis(hexadecanoyloxy)propoxy](hydroxy)phosphoryl}oxy)phosphinic acid |
|---|
| Traditional Name | [(2R,3R,5R)-5-(4-amino-2-oxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy([(2R)-2,3-bis(hexadecanoyloxy)propoxy(hydroxy)phosphoryl]oxy)phosphinic acid |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H][C@@](COC(=O)CCCCCCCCCCCCCCC)(COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H](C(O)[C@H]1O)N1C=CC(N)=NC1=O)OC(=O)CCCCCCCCCCCCCCC |
|---|
| InChI Identifier | InChI=1S/C44H81N3O15P2/c1-3-5-7-9-11-13-15-17-19-21-23-25-27-29-39(48)57-33-36(60-40(49)30-28-26-24-22-20-18-16-14-12-10-8-6-4-2)34-58-63(53,54)62-64(55,56)59-35-37-41(50)42(51)43(61-37)47-32-31-38(45)46-44(47)52/h31-32,36-37,41-43,50-51H,3-30,33-35H2,1-2H3,(H,53,54)(H,55,56)(H2,45,46,52)/t36-,37-,41+,42?,43-/m1/s1 |
|---|
| InChI Key | ITYHVANGBZMQML-QDFYKRCGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as cdp-diacylglycerols. These are glycerolipids containing a diacylglycerol, with a cytidine diphosphate attached to the oxygen O1 or O2 of the glycerol part. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerophospholipids |
|---|
| Sub Class | CDP-glycerols |
|---|
| Direct Parent | CDP-diacylglycerols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cdp-diacylglycerol
- Pyrimidine ribonucleoside diphosphate
- Diacyl-glycerol-3-pyrophosphate
- Pentose phosphate
- Pentose-5-phosphate
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Organic pyrophosphate
- Pentose monosaccharide
- Aminopyrimidine
- Fatty acid ester
- Pyrimidone
- Monoalkyl phosphate
- Imidolactam
- Pyrimidine
- Fatty acyl
- Alkyl phosphate
- Phosphoric acid ester
- Dicarboxylic acid or derivatives
- Hydropyrimidine
- Organic phosphoric acid derivative
- Monosaccharide
- Tetrahydrofuran
- Heteroaromatic compound
- Secondary alcohol
- Amino acid or derivatives
- 1,2-diol
- Carboxylic acid ester
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Carboxylic acid derivative
- Alcohol
- Organonitrogen compound
- Organic nitrogen compound
- Organooxygen compound
- Amine
- Carbonyl group
- Primary amine
- Organic oxygen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organic oxide
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | |
|---|
| Disposition | |
|---|
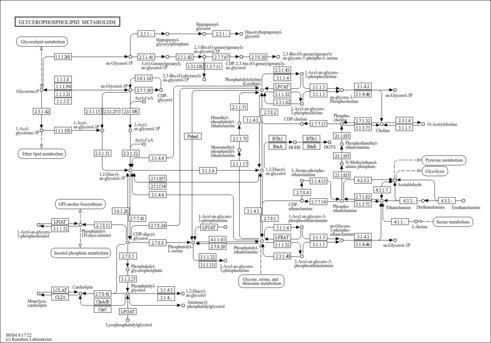
| Process | Naturally occurring processBiological processChemical reactionBiochemical pathwayMetabolic pathway- Cardiolipin Biosynthesis (HMDB: HMDB0006968)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/18:0) (PathBank: SMP0026956)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/18:1(9Z)) (PathBank: SMP0026958)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/18:1(9Z)) (PathBank: SMP0026968)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/18:1(11Z)) (PathBank: SMP0026977)
- Phospholipid Biosynthesis (PathBank: SMP0063652)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/18:2(9Z,12Z)) (PathBank: SMP0068409)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0068413)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/18:1(11Z)) (PathBank: SMP0068417)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0068419)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/18:1(9Z)) (PathBank: SMP0068428)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/18:2(9Z,12Z)) (PathBank: SMP0068429)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/18:0) (PathBank: SMP0068436)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/18:2(9Z,12Z)) (PathBank: SMP0068439)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0068441)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0068442)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/18:0) (PathBank: SMP0068446)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0068449)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0068450)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0068452)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0068473)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0077167)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0068483)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/18:0) (PathBank: SMP0068486)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/18:1(9Z)) (PathBank: SMP0068488)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/18:2(9Z,12Z)) (PathBank: SMP0068489)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0068490)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/16:0) (PathBank: SMP0068494)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:1(11Z)) (PathBank: SMP0068497)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0068501)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/18:1(11Z)) (PathBank: SMP0077154)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/18:0) (PathBank: SMP0077163)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0077169)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/18:0) (PathBank: SMP0077173)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/18:1(11Z)) (PathBank: SMP0077185)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0077188)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/16:0) (PathBank: SMP0077192)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/16:1(9Z)) (PathBank: SMP0077193)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/18:1(11Z)) (PathBank: SMP0077195)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0077199)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/18:0) (PathBank: SMP0077204)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/18:2(9Z,12Z)) (PathBank: SMP0077207)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0077209)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/18:0) (PathBank: SMP0077214)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/18:2(9Z,12Z)) (PathBank: SMP0077217)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/16:1(9Z)) (PathBank: SMP0077223)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/18:1(9Z)) (PathBank: SMP0077226)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/18:1(11Z)) (PathBank: SMP0077235)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:1(13Z)) (PathBank: SMP0080887)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/22:0) (PathBank: SMP0080888)
- Cardiolipin Biosynthesis CL(14:0/16:0/16:0/16:0) (PathBank: SMP0082794)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/16:1(9Z)) (PathBank: SMP0083358)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0083363)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:0) (PathBank: SMP0083390)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/18:1(9Z)) (PathBank: SMP0083396)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0083409)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:0) (PathBank: SMP0083428)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:0) (PathBank: SMP0083435)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0092432)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092444)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0092451)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/16:1(9Z)) (PathBank: SMP0092456)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092474)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/16:0) (PathBank: SMP0092475)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092484)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/18:1(11Z)) (PathBank: SMP0092488)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0092491)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0092492)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/16:0) (PathBank: SMP0092495)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/18:1(11Z)) (PathBank: SMP0092498)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092503)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092514)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/16:1(9Z)) (PathBank: SMP0092516)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:2(9Z,12Z)) (PathBank: SMP0092520)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/22:0) (PathBank: SMP0098235)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/22:1(13Z)) (PathBank: SMP0098236)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0098242)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:1(11Z)) (PathBank: SMP0098244)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0098253)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/22:1(13Z)) (PathBank: SMP0098259)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0098263)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098267)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/22:0) (PathBank: SMP0098268)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/22:1(13Z)) (PathBank: SMP0098269)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098276)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:1(11Z)) (PathBank: SMP0098282)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0098283)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/22:0) (PathBank: SMP0098292)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:1(11Z)) (PathBank: SMP0098295)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0098296)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098297)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/22:0) (PathBank: SMP0098298)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(11Z)/22:0) (PathBank: SMP0098303)
- Phospholipid Biosynthesis CL(15:0cycw5/17:0cycw7/16:0/16:0) (PathBank: SMP0121473)
- Phospholipid Biosynthesis CL(16:0/16:0/14:0/19:0cycw7) (PathBank: SMP0121583)
- Phospholipid Biosynthesis CL(16:0/16:0/16:0/18:1(9Z)) (PathBank: SMP0121585)
- Phospholipid Biosynthesis CL(16:0/16:0/16:1(9Z)/15:0cycw5) (PathBank: SMP0121588)
- Phospholipid Biosynthesis CL(16:0/16:0/16:1(9Z)/16:1(9Z)) (PathBank: SMP0121592)
- Phospholipid Biosynthesis CL(16:0/16:0/18:1(9Z)/14:0) (PathBank: SMP0121593)
- Phospholipid Biosynthesis CL(16:0/16:0/17:0cycw7/16:0) (PathBank: SMP0121599)
- Phospholipid Biosynthesis CL(16:0/16:0/17:0cycw7/17:0cycw7) (PathBank: SMP0121601)
- Phospholipid Biosynthesis CL(16:0/16:0/17:0cycw7/19:0cycw7) (PathBank: SMP0121605)
- Phospholipid Biosynthesis CL(16:0/16:0/18:1(9Z)/16:0) (PathBank: SMP0121607)
- Phospholipid Biosynthesis CL(16:0/16:0/19:0cycw7/14:0) (PathBank: SMP0121612)
- Phospholipid Biosynthesis CL(16:0/16:0/19:0cycw7/16:1(9Z)) (PathBank: SMP0121615)
- Phospholipid Biosynthesis CL(14:0(3-OH)/16:0/16:0/16:0) (PathBank: SMP0122144)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0068410)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/16:0) (PathBank: SMP0068414)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0068421)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/18:1(11Z)) (PathBank: SMP0068457)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/16:1(9Z)) (PathBank: SMP0068465)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/18:1(9Z)) (PathBank: SMP0068468)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/18:0) (PathBank: SMP0068476)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/18:2(9Z,12Z)) (PathBank: SMP0068479)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0068480)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(4Z,7Z,10Z,13Z,16Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0068481)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0068492)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:0) (PathBank: SMP0068496)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0068502)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0077159)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0077179)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/18:1(9Z)) (PathBank: SMP0077186)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/18:1(9Z)) (PathBank: SMP0077206)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0077210)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0077220)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/16:1(9Z)) (PathBank: SMP0077233)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0077239)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/22:0) (PathBank: SMP0080907)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/16:0) (PathBank: SMP0080867)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:1(11Z)) (PathBank: SMP0080896)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:1(13Z)) (PathBank: SMP0080897)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:0) (PathBank: SMP0080904)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:1(13Z)) (PathBank: SMP0080932)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0083388)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0083399)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0083410)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/16:0) (PathBank: SMP0092445)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/16:1(9Z)) (PathBank: SMP0092446)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092464)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/16:0) (PathBank: SMP0092485)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:5(7Z,10Z,13Z,16Z,19Z)/16:0) (PathBank: SMP0092505)
- Phospholipid Biosynthesis CL(16:0/16:0/16:0/17:0cycw7) (PathBank: SMP0121584)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0092521)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092524)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0098230)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:0) (PathBank: SMP0098231)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:0) (PathBank: SMP0098243)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:0) (PathBank: SMP0098264)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:1(11Z)) (PathBank: SMP0098274)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/22:1(13Z)) (PathBank: SMP0098278)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:0) (PathBank: SMP0098281)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/22:1(13Z)) (PathBank: SMP0098286)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0098287)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:1(11Z)) (PathBank: SMP0098289)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/22:1(13Z)) (PathBank: SMP0098293)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(11Z)/20:1(11Z)) (PathBank: SMP0098300)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098306)
- Phospholipid Biosynthesis CL(16:0/16:0/16:0/19:0cycw7) (PathBank: SMP0121586)
- Phospholipid Biosynthesis CL(16:0/16:0/16:1(9Z)/14:0) (PathBank: SMP0121587)
- Phospholipid Biosynthesis CL(16:0/16:0/16:1(9Z)/16:0) (PathBank: SMP0121591)
- Phospholipid Biosynthesis CL(16:0/16:0/16:1(9Z)/19:0cycw7) (PathBank: SMP0121596)
- Phospholipid Biosynthesis CL(16:0/16:0/17:0cycw7/16:1(9Z)) (PathBank: SMP0121600)
- Phospholipid Biosynthesis CL(16:0/16:0/18:1(9Z)/18:1(9Z)) (PathBank: SMP0121610)
- Phospholipid Biosynthesis CL(16:0/16:0/18:1(9Z)/19:0cycw7) (PathBank: SMP0121611)
- Phospholipid Biosynthesis CL(16:0/16:0/19:0cycw7/16:0) (PathBank: SMP0121614)
- Phospholipid Biosynthesis CL(16:0/16:0/19:0cycw7/17:0cycw7) (PathBank: SMP0121616)
- Phospholipid Biosynthesis CL(16:1(9Z)/19:0/16:0/16:0) (PathBank: SMP0122163)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0068431)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/16:0) (PathBank: SMP0068434)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/16:1(9Z)) (PathBank: SMP0068455)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:1(9Z)) (PathBank: SMP0068498)
- Phosphatidylethanolamine Biosynthesis PE(16:0/16:0) (PathBank: SMP0071720)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:1(11Z)) (PathBank: SMP0080875)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:1(13Z)) (PathBank: SMP0080906)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/16:1(9Z)) (PathBank: SMP0083371)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0083418)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0083419)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0083426)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0083427)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0092454)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/16:0/16:0) (PathBank: SMP0097209)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098234)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098246)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098284)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/22:0) (PathBank: SMP0098285)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0098290)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:0) (PathBank: SMP0098294)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(11Z)/22:1(13Z)) (PathBank: SMP0098304)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(8Z,11Z,14Z,17Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098309)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:0/22:1(13Z)) (PathBank: SMP0098313)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:1(13Z)/22:1(13Z)) (PathBank: SMP0098314)
- Phospholipid Biosynthesis CL(15:0cycw5/16:0/16:0/16:0) (PathBank: SMP0121428)
- Phospholipid Biosynthesis CL(15:0cycw5/16:1(9Z)/16:0/16:0) (PathBank: SMP0121452)
- Phospholipid Biosynthesis CL(15:0cycw5/19:0cycw7/16:0/16:0) (PathBank: SMP0121516)
- Phospholipid Biosynthesis CL(16:0/15:0cycw5/16:0/16:0) (PathBank: SMP0121553)
- Phospholipid Biosynthesis CL(16:0/16:0/16:1(9Z)/17:0cycw7) (PathBank: SMP0121594)
- Phospholipid Biosynthesis CL(16:0/16:0/18:1(9Z)/16:1(9Z)) (PathBank: SMP0121608)
- Phospholipid Biosynthesis CL(16:0/16:0/19:0cycw7/15:0cycw5) (PathBank: SMP0121613)
- Phospholipid Biosynthesis CL(16:0/16:0/16:0/16:0) (PathBank: SMP0122211)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:1(13Z)) (PathBank: SMP0080876)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:1(11Z)) (PathBank: SMP0080886)
- Cardiolipin Biosynthesis CL(14:0/14:0/16:0/16:0) (PathBank: SMP0096207)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/16:0/16:0) (PathBank: SMP0096327)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/22:0) (PathBank: SMP0098247)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098257)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098291)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/22:1(13Z)) (PathBank: SMP0098299)
- Cardiolipin Biosynthesis CL(16:0/16:0/22:0/22:0) (PathBank: SMP0098312)
- Phospholipid Biosynthesis CL(15:0cycw5/18:1(9Z)/16:0/16:0) (PathBank: SMP0121493)
- Phospholipid Biosynthesis CL(16:0/16:0/14:0/14:0) (PathBank: SMP0121577)
- Phospholipid Biosynthesis CL(16:0/16:0/14:0/16:1(9Z)) (PathBank: SMP0121579)
- Phospholipid Biosynthesis CL(16:0/16:0/14:0/17:0cycw7) (PathBank: SMP0121581)
- Phospholipid Biosynthesis CL(16:0/16:0/14:0/18:1(9Z)) (PathBank: SMP0121582)
- Phospholipid Biosynthesis CL(16:0/16:0/16:1(9Z)/18:1(9Z)) (PathBank: SMP0121595)
- Phospholipid Biosynthesis CL(16:0/16:0/17:0cycw7/14:0) (PathBank: SMP0121597)
- Phospholipid Biosynthesis CL(16:0/16:0/17:0cycw7/15:0cycw5) (PathBank: SMP0121598)
- Phospholipid Biosynthesis CL(16:0/16:0/17:0cycw7/18:1(9Z)) (PathBank: SMP0121604)
- Phospholipid Biosynthesis CL(16:0/16:0/18:1(9Z)/15:0cycw5) (PathBank: SMP0121606)
- Phospholipid Biosynthesis CL(16:0/16:0/18:1(9Z)/17:0cycw7) (PathBank: SMP0121609)
- Phospholipid Biosynthesis CL(16:0/16:0/19:0cycw7/18:1(9Z)) (PathBank: SMP0121617)
- Phospholipid Biosynthesis CL(17:0/16:1(9Z)/16:0/16:0) (PathBank: SMP0122166)
- Phospholipid Biosynthesis CL(19:0/16:1(9Z)/16:0/16:0) (PathBank: SMP0122186)
- Cardiolipin Biosynthesis CL(10:0/10:0/18:0/22:1(11Z)) (PathBank: SMP0002785)
- Cardiolipin Biosynthesis CL(10:0/10:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0002798)
- Cardiolipin Biosynthesis CL(10:0/10:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0002811)
- Cardiolipin Biosynthesis CL(10:0/10:0/20:0/22:1(11Z)) (PathBank: SMP0002826)
- Cardiolipin Biosynthesis CL(10:0/10:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0002839)
- Cardiolipin Biosynthesis CL(10:0/10:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0002852)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:0/22:1(11Z)) (PathBank: SMP0002864)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/24:0) (PathBank: SMP0002876)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/24:1(11Z)) (PathBank: SMP0002877)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/24:1(9Z)) (PathBank: SMP0002878)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/26:0) (PathBank: SMP0002879)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/26:1(11Z)) (PathBank: SMP0002880)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/26:1(9Z)) (PathBank: SMP0002881)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/28:0) (PathBank: SMP0002882)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/28:1(11Z)) (PathBank: SMP0002883)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/28:1(9Z)) (PathBank: SMP0002884)
- Cardiolipin Biosynthesis CL(10:0/10:0/22:1(11Z)/30:0) (PathBank: SMP0002885)
- Cardiolipin Biosynthesis CL(10:0/12:0/16:0/22:1(11Z)) (PathBank: SMP0003011)
- Cardiolipin Biosynthesis CL(10:0/12:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0003024)
- Cardiolipin Biosynthesis CL(10:0/12:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0003037)
- Cardiolipin Biosynthesis CL(10:0/12:0/18:0/22:1(11Z)) (PathBank: SMP0003054)
- Cardiolipin Biosynthesis CL(10:0/12:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0003070)
- Cardiolipin Biosynthesis CL(10:0/12:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0003086)
- Cardiolipin Biosynthesis CL(10:0/12:0/20:0/22:1(11Z)) (PathBank: SMP0003102)
- Cardiolipin Biosynthesis CL(10:0/12:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0003117)
- Cardiolipin Biosynthesis CL(10:0/12:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0003132)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:0/22:1(11Z)) (PathBank: SMP0003145)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/22:1(11Z)) (PathBank: SMP0003156)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/22:1(9Z)) (PathBank: SMP0003157)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/24:0) (PathBank: SMP0003158)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/24:1(11Z)) (PathBank: SMP0003159)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/24:1(9Z)) (PathBank: SMP0003160)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/26:0) (PathBank: SMP0003161)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/26:1(11Z)) (PathBank: SMP0003162)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/26:1(9Z)) (PathBank: SMP0003163)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/28:0) (PathBank: SMP0003164)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/28:1(11Z)) (PathBank: SMP0003165)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(11Z)/28:1(9Z)) (PathBank: SMP0003166)
- Cardiolipin Biosynthesis CL(10:0/12:0/22:1(9Z)/22:1(11Z)) (PathBank: SMP0003167)
- Cardiolipin Biosynthesis CL(10:0/14:0/14:0/22:1(11Z)) (PathBank: SMP0003219)
- Cardiolipin Biosynthesis CL(10:0/14:0/14:1(11Z)/22:1(11Z)) (PathBank: SMP0003232)
- Cardiolipin Biosynthesis CL(10:0/14:0/14:1(9Z)/22:1(11Z)) (PathBank: SMP0003245)
- Cardiolipin Biosynthesis CL(10:0/14:0/16:0/22:1(11Z)) (PathBank: SMP0003289)
- Cardiolipin Biosynthesis CL(10:0/14:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0003305)
- Cardiolipin Biosynthesis CL(10:0/14:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0003321)
- Cardiolipin Biosynthesis CL(10:0/14:0/18:0/22:1(11Z)) (PathBank: SMP0003340)
- Cardiolipin Biosynthesis CL(10:0/14:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0003358)
- Cardiolipin Biosynthesis CL(10:0/14:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0003376)
- Cardiolipin Biosynthesis CL(10:0/14:0/20:0/22:1(11Z)) (PathBank: SMP0003392)
- Cardiolipin Biosynthesis CL(10:0/14:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0003406)
- Cardiolipin Biosynthesis CL(10:0/14:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0003420)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:0/22:1(11Z)) (PathBank: SMP0003432)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/22:1(11Z)) (PathBank: SMP0003440)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/22:1(9Z)) (PathBank: SMP0003441)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/24:0) (PathBank: SMP0003442)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/24:1(11Z)) (PathBank: SMP0003443)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/24:1(9Z)) (PathBank: SMP0003444)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/26:0) (PathBank: SMP0003445)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/26:1(11Z)) (PathBank: SMP0003446)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(11Z)/26:1(9Z)) (PathBank: SMP0003447)
- Cardiolipin Biosynthesis CL(10:0/14:0/22:1(9Z)/22:1(11Z)) (PathBank: SMP0003448)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0003474)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0003487)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0003530)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0003546)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0003562)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0003581)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0003599)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0003617)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0003633)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0003647)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0003661)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0003673)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0003681)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0003682)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/24:0) (PathBank: SMP0003683)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/24:1(11Z)) (PathBank: SMP0003684)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/24:1(9Z)) (PathBank: SMP0003685)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/26:0) (PathBank: SMP0003686)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/26:1(11Z)) (PathBank: SMP0003687)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(11Z)/26:1(9Z)) (PathBank: SMP0003688)
- Cardiolipin Biosynthesis CL(10:0/14:1(11Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0003689)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0003715)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0003728)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0003771)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0003787)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0003803)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0003822)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0003840)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0003858)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0003874)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0003888)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0003902)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0003914)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0003922)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0003923)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/24:0) (PathBank: SMP0003924)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/24:1(11Z)) (PathBank: SMP0003925)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/24:1(9Z)) (PathBank: SMP0003926)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/26:0) (PathBank: SMP0003927)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/26:1(11Z)) (PathBank: SMP0003928)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(11Z)/26:1(9Z)) (PathBank: SMP0003929)
- Cardiolipin Biosynthesis CL(10:0/14:1(9Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0003930)
- Cardiolipin Biosynthesis CL(10:0/15:0/15:0/22:1(11Z)) (PathBank: SMP0003960)
- Cardiolipin Biosynthesis CL(10:0/15:0/15:1(11Z)/22:1(11Z)) (PathBank: SMP0003976)
- Cardiolipin Biosynthesis CL(10:0/15:0/15:1(9Z)/22:1(11Z)) (PathBank: SMP0003992)
- Cardiolipin Biosynthesis CL(10:0/15:0/22:1(11Z)/23:1(11Z)) (PathBank: SMP0004087)
- Cardiolipin Biosynthesis CL(10:0/15:0/22:1(11Z)/23:1(9Z)) (PathBank: SMP0004088)
- Cardiolipin Biosynthesis CL(10:0/15:0/22:1(11Z)/25:0) (PathBank: SMP0004089)
- Cardiolipin Biosynthesis CL(10:0/15:0/22:1(11Z)/25:1(11Z)) (PathBank: SMP0004090)
- Cardiolipin Biosynthesis CL(10:0/15:0/22:1(11Z)/25:1(9Z)) (PathBank: SMP0004091)
- Cardiolipin Biosynthesis CL(10:0/15:1(11Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0004107)
- Cardiolipin Biosynthesis CL(10:0/15:1(11Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0004123)
- Cardiolipin Biosynthesis CL(10:0/15:1(11Z)/22:1(11Z)/23:1(11Z)) (PathBank: SMP0004218)
- Cardiolipin Biosynthesis CL(10:0/15:1(11Z)/22:1(11Z)/23:1(9Z)) (PathBank: SMP0004219)
- Cardiolipin Biosynthesis CL(10:0/15:1(11Z)/22:1(11Z)/25:0) (PathBank: SMP0004220)
- Cardiolipin Biosynthesis CL(10:0/15:1(11Z)/22:1(11Z)/25:1(11Z)) (PathBank: SMP0004221)
- Cardiolipin Biosynthesis CL(10:0/15:1(11Z)/22:1(11Z)/25:1(9Z)) (PathBank: SMP0004222)
- Cardiolipin Biosynthesis CL(10:0/15:1(9Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0004238)
- Cardiolipin Biosynthesis CL(10:0/15:1(9Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0004254)
- Cardiolipin Biosynthesis CL(10:0/15:1(9Z)/22:1(11Z)/23:1(11Z)) (PathBank: SMP0004349)
- Cardiolipin Biosynthesis CL(10:0/15:1(9Z)/22:1(11Z)/23:1(9Z)) (PathBank: SMP0004350)
- Cardiolipin Biosynthesis CL(10:0/15:1(9Z)/22:1(11Z)/25:0) (PathBank: SMP0004351)
- Cardiolipin Biosynthesis CL(10:0/15:1(9Z)/22:1(11Z)/25:1(11Z)) (PathBank: SMP0004352)
- Cardiolipin Biosynthesis CL(10:0/15:1(9Z)/22:1(11Z)/25:1(9Z)) (PathBank: SMP0004353)
- Cardiolipin Biosynthesis CL(10:0/16:0/16:0/16:0) (PathBank: SMP0004365)
- Cardiolipin Biosynthesis CL(10:0/16:0/16:0/22:1(11Z)) (PathBank: SMP0004373)
- Cardiolipin Biosynthesis CL(10:0/16:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0004392)
- Cardiolipin Biosynthesis CL(10:0/16:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0004411)
- Cardiolipin Biosynthesis CL(10:0/16:0/18:0/22:1(11Z)) (PathBank: SMP0004430)
- Cardiolipin Biosynthesis CL(10:0/16:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0004447)
- Cardiolipin Biosynthesis CL(10:0/16:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0004464)
- Cardiolipin Biosynthesis CL(10:0/16:0/20:0/22:1(11Z)) (PathBank: SMP0004479)
- Cardiolipin Biosynthesis CL(10:0/16:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0004490)
- Cardiolipin Biosynthesis CL(10:0/16:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0004501)
- Cardiolipin Biosynthesis CL(10:0/16:0/22:0/22:1(11Z)) (PathBank: SMP0004510)
- Cardiolipin Biosynthesis CL(10:0/16:0/22:1(11Z)/22:1(11Z)) (PathBank: SMP0004515)
- Cardiolipin Biosynthesis CL(10:0/16:0/22:1(11Z)/22:1(9Z)) (PathBank: SMP0004516)
- Cardiolipin Biosynthesis CL(10:0/16:0/22:1(11Z)/24:0) (PathBank: SMP0004517)
- Cardiolipin Biosynthesis CL(10:0/16:0/22:1(11Z)/24:1(11Z)) (PathBank: SMP0004518)
- Cardiolipin Biosynthesis CL(10:0/16:0/22:1(11Z)/24:1(9Z)) (PathBank: SMP0004519)
- Cardiolipin Biosynthesis CL(10:0/16:0/22:1(9Z)/22:1(11Z)) (PathBank: SMP0004520)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0004536)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0004555)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0004574)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0004591)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0004608)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0004623)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0004634)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0004645)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0004654)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0004659)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0004660)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/22:1(11Z)/24:0) (PathBank: SMP0004661)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/22:1(11Z)/24:1(11Z)) (PathBank: SMP0004662)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/22:1(11Z)/24:1(9Z)) (PathBank: SMP0004663)
- Cardiolipin Biosynthesis CL(10:0/16:1(11Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0004664)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0004680)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0004699)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0004718)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0004735)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0004752)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0004767)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0004778)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0004789)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0004798)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0004803)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0004804)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/22:1(11Z)/24:0) (PathBank: SMP0004805)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/22:1(11Z)/24:1(11Z)) (PathBank: SMP0004806)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/22:1(11Z)/24:1(9Z)) (PathBank: SMP0004807)
- Cardiolipin Biosynthesis CL(10:0/16:1(9Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0004808)
- Cardiolipin Biosynthesis CL(10:0/18:0/18:0/22:1(11Z)) (PathBank: SMP0004824)
- Cardiolipin Biosynthesis CL(10:0/18:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0004838)
- Cardiolipin Biosynthesis CL(10:0/18:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0004852)
- Cardiolipin Biosynthesis CL(10:0/18:0/20:0/22:1(11Z)) (PathBank: SMP0004864)
- Cardiolipin Biosynthesis CL(10:0/18:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0004872)
- Cardiolipin Biosynthesis CL(10:0/18:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0004880)
- Cardiolipin Biosynthesis CL(10:0/18:0/22:0/22:1(11Z)) (PathBank: SMP0004886)
- Cardiolipin Biosynthesis CL(10:0/18:0/22:1(11Z)/22:1(11Z)) (PathBank: SMP0004888)
- Cardiolipin Biosynthesis CL(10:0/18:0/22:1(11Z)/22:1(9Z)) (PathBank: SMP0004889)
- Cardiolipin Biosynthesis CL(10:0/18:0/22:1(9Z)/22:1(11Z)) (PathBank: SMP0004890)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0004898)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0004912)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0004924)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0004932)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0004940)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0004946)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0004948)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0004949)
- Cardiolipin Biosynthesis CL(10:0/18:1(11Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0004950)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0004958)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0004972)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0004984)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0004992)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005000)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0005006)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0005008)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0005009)
- Cardiolipin Biosynthesis CL(10:0/18:1(9Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0005010)
- Cardiolipin Biosynthesis CL(10:0/20:0/20:0/22:1(11Z)) (PathBank: SMP0005017)
- Cardiolipin Biosynthesis CL(10:0/20:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0005022)
- Cardiolipin Biosynthesis CL(10:0/20:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005027)
- Cardiolipin Biosynthesis CL(10:0/20:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0005034)
- Cardiolipin Biosynthesis CL(10:0/20:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005039)
- Cardiolipin Biosynthesis CL(10:0/20:1(13Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0005046)
- Cardiolipin Biosynthesis CL(10:0/20:1(13Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005051)
- Cardiolipin Biosynthesis CL(10:0/22:1(11Z)/10:0/22:1(11Z)) (PathBank: SMP0005054)
- Cardiolipin Biosynthesis CL(10:0/22:1(11Z)/10:0/22:1(9Z)) (PathBank: SMP0005055)
- Cardiolipin Biosynthesis CL(10:0/22:1(9Z)/10:0/22:1(11Z)) (PathBank: SMP0005056)
- Cardiolipin Biosynthesis CL(12:0/12:0/14:0/22:1(11Z)) (PathBank: SMP0005090)
- Cardiolipin Biosynthesis CL(12:0/12:0/14:1(11Z)/22:1(11Z)) (PathBank: SMP0005103)
- Cardiolipin Biosynthesis CL(12:0/12:0/14:1(9Z)/22:1(11Z)) (PathBank: SMP0005116)
- Cardiolipin Biosynthesis CL(12:0/12:0/16:0/22:1(11Z)) (PathBank: SMP0005160)
- Cardiolipin Biosynthesis CL(12:0/12:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0005176)
- Cardiolipin Biosynthesis CL(12:0/12:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0005192)
- Cardiolipin Biosynthesis CL(12:0/12:0/18:0/22:1(11Z)) (PathBank: SMP0005210)
- Cardiolipin Biosynthesis CL(12:0/12:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0005226)
- Cardiolipin Biosynthesis CL(12:0/12:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0005242)
- Cardiolipin Biosynthesis CL(12:0/12:0/20:0/22:1(11Z)) (PathBank: SMP0005257)
- Cardiolipin Biosynthesis CL(12:0/12:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0005269)
- Cardiolipin Biosynthesis CL(12:0/12:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005281)
- Cardiolipin Biosynthesis CL(12:0/12:0/22:0/22:1(11Z)) (PathBank: SMP0005292)
- Cardiolipin Biosynthesis CL(12:0/12:0/22:1(11Z)/24:0) (PathBank: SMP0005300)
- Cardiolipin Biosynthesis CL(12:0/12:0/22:1(11Z)/24:1(11Z)) (PathBank: SMP0005301)
- Cardiolipin Biosynthesis CL(12:0/12:0/22:1(11Z)/24:1(9Z)) (PathBank: SMP0005302)
- Cardiolipin Biosynthesis CL(12:0/12:0/22:1(11Z)/26:0) (PathBank: SMP0005303)
- Cardiolipin Biosynthesis CL(12:0/12:0/22:1(11Z)/26:1(11Z)) (PathBank: SMP0005304)
- Cardiolipin Biosynthesis CL(12:0/12:0/22:1(11Z)/26:1(9Z)) (PathBank: SMP0005305)
- Cardiolipin Biosynthesis CL(12:0/14:0/14:0/22:1(11Z)) (PathBank: SMP0005325)
- Cardiolipin Biosynthesis CL(12:0/14:0/14:1(11Z)/22:1(11Z)) (PathBank: SMP0005341)
- Cardiolipin Biosynthesis CL(12:0/14:0/14:1(9Z)/22:1(11Z)) (PathBank: SMP0005357)
- Cardiolipin Biosynthesis CL(12:0/14:0/16:0/16:0) (PathBank: SMP0005396)
- Cardiolipin Biosynthesis CL(12:0/14:0/16:0/22:1(11Z)) (PathBank: SMP0005404)
- Cardiolipin Biosynthesis CL(12:0/14:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0005423)
- Cardiolipin Biosynthesis CL(12:0/14:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0005442)
- Cardiolipin Biosynthesis CL(12:0/14:0/18:0/22:1(11Z)) (PathBank: SMP0005461)
- Cardiolipin Biosynthesis CL(12:0/14:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0005478)
- Cardiolipin Biosynthesis CL(12:0/14:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0005495)
- Cardiolipin Biosynthesis CL(12:0/14:0/20:0/22:1(11Z)) (PathBank: SMP0005510)
- Cardiolipin Biosynthesis CL(12:0/14:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0005521)
- Cardiolipin Biosynthesis CL(12:0/14:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005532)
- Cardiolipin Biosynthesis CL(12:0/14:0/22:0/22:1(11Z)) (PathBank: SMP0005541)
- Cardiolipin Biosynthesis CL(12:0/14:0/22:1(11Z)/22:1(11Z)) (PathBank: SMP0005546)
- Cardiolipin Biosynthesis CL(12:0/14:0/22:1(11Z)/22:1(9Z)) (PathBank: SMP0005547)
- Cardiolipin Biosynthesis CL(12:0/14:0/22:1(11Z)/24:0) (PathBank: SMP0005548)
- Cardiolipin Biosynthesis CL(12:0/14:0/22:1(11Z)/24:1(11Z)) (PathBank: SMP0005549)
- Cardiolipin Biosynthesis CL(12:0/14:0/22:1(11Z)/24:1(9Z)) (PathBank: SMP0005550)
- Cardiolipin Biosynthesis CL(12:0/14:0/22:1(9Z)/22:1(11Z)) (PathBank: SMP0005551)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0005564)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0005580)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0005626)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0005645)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0005664)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0005683)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0005700)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0005717)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0005732)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0005743)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005754)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0005763)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0005768)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0005769)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/22:1(11Z)/24:0) (PathBank: SMP0005770)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/22:1(11Z)/24:1(11Z)) (PathBank: SMP0005771)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/22:1(11Z)/24:1(9Z)) (PathBank: SMP0005772)
- Cardiolipin Biosynthesis CL(12:0/14:1(11Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0005773)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0005786)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0005802)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0005848)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0005867)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0005886)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0005905)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0005922)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0005939)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0005954)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0005965)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0005976)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0005985)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0005990)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0005991)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/22:1(11Z)/24:0) (PathBank: SMP0005992)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/22:1(11Z)/24:1(11Z)) (PathBank: SMP0005993)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/22:1(11Z)/24:1(9Z)) (PathBank: SMP0005994)
- Cardiolipin Biosynthesis CL(12:0/14:1(9Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0005995)
- Cardiolipin Biosynthesis CL(12:0/15:0/15:0/22:1(11Z)) (PathBank: SMP0006012)
- Cardiolipin Biosynthesis CL(12:0/15:0/15:1(11Z)/22:1(11Z)) (PathBank: SMP0006031)
- Cardiolipin Biosynthesis CL(12:0/15:0/15:1(9Z)/22:1(11Z)) (PathBank: SMP0006050)
- Cardiolipin Biosynthesis CL(12:0/15:0/22:1(11Z)/23:1(11Z)) (PathBank: SMP0006130)
- Cardiolipin Biosynthesis CL(12:0/15:0/22:1(11Z)/23:1(9Z)) (PathBank: SMP0006131)
- Cardiolipin Biosynthesis CL(12:0/15:1(11Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0006141)
- Cardiolipin Biosynthesis CL(12:0/15:1(11Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0006160)
- Cardiolipin Biosynthesis CL(12:0/15:1(11Z)/22:1(11Z)/23:1(11Z)) (PathBank: SMP0006240)
- Cardiolipin Biosynthesis CL(12:0/15:1(11Z)/22:1(11Z)/23:1(9Z)) (PathBank: SMP0006241)
- Cardiolipin Biosynthesis CL(12:0/15:1(9Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0006251)
- Cardiolipin Biosynthesis CL(12:0/15:1(9Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0006270)
- Cardiolipin Biosynthesis CL(12:0/15:1(9Z)/22:1(11Z)/23:1(11Z)) (PathBank: SMP0006350)
- Cardiolipin Biosynthesis CL(12:0/15:1(9Z)/22:1(11Z)/23:1(9Z)) (PathBank: SMP0006351)
- Cardiolipin Biosynthesis CL(12:0/16:0/16:0/16:0) (PathBank: SMP0006354)
- Cardiolipin Biosynthesis CL(12:0/16:0/16:0/22:1(11Z)) (PathBank: SMP0006364)
- Cardiolipin Biosynthesis CL(12:0/16:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0006384)
- Cardiolipin Biosynthesis CL(12:0/16:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0006404)
- Cardiolipin Biosynthesis CL(12:0/16:0/18:0/22:1(11Z)) (PathBank: SMP0006422)
- Cardiolipin Biosynthesis CL(12:0/16:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0006436)
- Cardiolipin Biosynthesis CL(12:0/16:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0006450)
- Cardiolipin Biosynthesis CL(12:0/16:0/20:0/22:1(11Z)) (PathBank: SMP0006462)
- Cardiolipin Biosynthesis CL(12:0/16:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0006470)
- Cardiolipin Biosynthesis CL(12:0/16:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0006478)
- Cardiolipin Biosynthesis CL(12:0/16:0/22:0/22:1(11Z)) (PathBank: SMP0006484)
- Cardiolipin Biosynthesis CL(12:0/16:0/22:1(11Z)/22:1(11Z)) (PathBank: SMP0006486)
- Cardiolipin Biosynthesis CL(12:0/16:0/22:1(11Z)/22:1(9Z)) (PathBank: SMP0006487)
- Cardiolipin Biosynthesis CL(12:0/16:0/22:1(9Z)/22:1(11Z)) (PathBank: SMP0006488)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0006499)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0006519)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0006537)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0006551)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0006565)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0006577)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0006585)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0006593)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0006599)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0006601)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0006602)
- Cardiolipin Biosynthesis CL(12:0/16:1(11Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0006603)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0006614)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0006634)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0006652)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0006666)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0006680)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0006692)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0006700)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0006708)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0006714)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0007042)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0007043)
- Cardiolipin Biosynthesis CL(12:0/16:1(9Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0007044)
- Cardiolipin Biosynthesis CL(12:0/18:0/18:0/22:1(11Z)) (PathBank: SMP0007054)
- Cardiolipin Biosynthesis CL(12:0/18:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0007065)
- Cardiolipin Biosynthesis CL(12:0/18:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0007076)
- Cardiolipin Biosynthesis CL(12:0/18:0/20:0/22:1(11Z)) (PathBank: SMP0007085)
- Cardiolipin Biosynthesis CL(12:0/18:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0007090)
- Cardiolipin Biosynthesis CL(12:0/18:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0007095)
- Cardiolipin Biosynthesis CL(12:0/18:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0007105)
- Cardiolipin Biosynthesis CL(12:0/18:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0007116)
- Cardiolipin Biosynthesis CL(12:0/18:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0007125)
- Cardiolipin Biosynthesis CL(12:0/18:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0007130)
- Cardiolipin Biosynthesis CL(12:0/18:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0007135)
- Cardiolipin Biosynthesis CL(12:0/18:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0007145)
- Cardiolipin Biosynthesis CL(12:0/18:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0007156)
- Cardiolipin Biosynthesis CL(12:0/18:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0007165)
- Cardiolipin Biosynthesis CL(12:0/18:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0007170)
- Cardiolipin Biosynthesis CL(12:0/18:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0007175)
- Cardiolipin Biosynthesis CL(12:0/22:1(11Z)/12:0/22:1(11Z)) (PathBank: SMP0007198)
- Cardiolipin Biosynthesis CL(12:0/22:1(11Z)/12:0/22:1(9Z)) (PathBank: SMP0007199)
- Cardiolipin Biosynthesis CL(12:0/22:1(9Z)/12:0/22:1(11Z)) (PathBank: SMP0007200)
- Cardiolipin Biosynthesis CL(14:0/14:0/14:0/22:1(11Z)) (PathBank: SMP0007219)
- Cardiolipin Biosynthesis CL(14:0/14:0/14:1(11Z)/22:1(11Z)) (PathBank: SMP0007238)
- Cardiolipin Biosynthesis CL(14:0/14:0/14:1(9Z)/22:1(11Z)) (PathBank: SMP0007257)
- Cardiolipin Biosynthesis CL(14:0/14:0/16:0/22:1(11Z)) (PathBank: SMP0007305)
- Cardiolipin Biosynthesis CL(14:0/14:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0007323)
- Cardiolipin Biosynthesis CL(14:0/14:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0007341)
- Cardiolipin Biosynthesis CL(14:0/14:0/18:0/22:1(11Z)) (PathBank: SMP0007358)
- Cardiolipin Biosynthesis CL(14:0/14:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0007370)
- Cardiolipin Biosynthesis CL(14:0/14:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0007382)
- Cardiolipin Biosynthesis CL(14:0/14:0/20:0/22:1(11Z)) (PathBank: SMP0007393)
- Cardiolipin Biosynthesis CL(14:0/14:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0007399)
- Cardiolipin Biosynthesis CL(14:0/14:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0007405)
- Cardiolipin Biosynthesis CL(14:0/14:0/22:0/22:1(11Z)) (PathBank: SMP0007410)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0007419)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0007438)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/16:0/16:0) (PathBank: SMP0007477)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0007487)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0007507)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0007527)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0007545)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0007559)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0007573)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0007585)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0007593)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0007601)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0007607)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0007609)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0007610)
- Cardiolipin Biosynthesis CL(14:0/14:1(11Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0007611)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0007620)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0007639)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0007688)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0007708)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0007728)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0007746)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0007760)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0007774)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0007786)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0007794)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0007802)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0007808)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/22:1(11Z)/22:1(11Z)) (PathBank: SMP0007810)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/22:1(11Z)/22:1(9Z)) (PathBank: SMP0007811)
- Cardiolipin Biosynthesis CL(14:0/14:1(9Z)/22:1(9Z)/22:1(11Z)) (PathBank: SMP0007812)
- Cardiolipin Biosynthesis CL(14:0/15:0/15:0/22:1(11Z)) (PathBank: SMP0007825)
- Cardiolipin Biosynthesis CL(14:0/15:0/15:1(11Z)/22:1(11Z)) (PathBank: SMP0007846)
- Cardiolipin Biosynthesis CL(14:0/15:0/15:1(9Z)/22:1(11Z)) (PathBank: SMP0007867)
- Cardiolipin Biosynthesis CL(14:0/15:1(11Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0007933)
- Cardiolipin Biosynthesis CL(14:0/15:1(11Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0007954)
- Cardiolipin Biosynthesis CL(14:0/15:1(9Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0008020)
- Cardiolipin Biosynthesis CL(14:0/15:1(9Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0008041)
- Cardiolipin Biosynthesis CL(14:0/16:0/16:0/22:1(11Z)) (PathBank: SMP0008108)
- Cardiolipin Biosynthesis CL(14:0/16:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0008125)
- Cardiolipin Biosynthesis CL(14:0/16:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0008142)
- Cardiolipin Biosynthesis CL(14:0/16:0/18:0/22:1(11Z)) (PathBank: SMP0008157)
- Cardiolipin Biosynthesis CL(14:0/16:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008168)
- Cardiolipin Biosynthesis CL(14:0/16:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008179)
- Cardiolipin Biosynthesis CL(14:0/16:0/20:0/22:1(11Z)) (PathBank: SMP0008188)
- Cardiolipin Biosynthesis CL(14:0/16:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0008193)
- Cardiolipin Biosynthesis CL(14:0/16:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0008198)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0008211)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0008228)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0008243)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008254)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008265)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0008274)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0008279)
- Cardiolipin Biosynthesis CL(14:0/16:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0008284)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0008297)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0008314)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0008329)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008340)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008351)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0008360)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0008365)
- Cardiolipin Biosynthesis CL(14:0/16:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0008370)
- Cardiolipin Biosynthesis CL(14:0/18:0/18:0/22:1(11Z)) (PathBank: SMP0008380)
- Cardiolipin Biosynthesis CL(14:0/18:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008388)
- Cardiolipin Biosynthesis CL(14:0/18:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008396)
- Cardiolipin Biosynthesis CL(14:0/18:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008413)
- Cardiolipin Biosynthesis CL(14:0/18:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008421)
- Cardiolipin Biosynthesis CL(14:0/18:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008438)
- Cardiolipin Biosynthesis CL(14:0/18:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008446)
- Cardiolipin Biosynthesis CL(14:0/22:1(11Z)/14:0/22:1(11Z)) (PathBank: SMP0008461)
- Cardiolipin Biosynthesis CL(14:0/22:1(11Z)/14:0/22:1(9Z)) (PathBank: SMP0008462)
- Cardiolipin Biosynthesis CL(14:0/22:1(9Z)/14:0/22:1(11Z)) (PathBank: SMP0008463)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0008472)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0008491)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0008539)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0008557)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0008575)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0008592)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008604)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008616)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0008627)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0008633)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0008639)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0008644)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0008653)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0008672)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0008720)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0008738)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0008756)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0008773)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0008785)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0008797)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0008808)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0008814)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0008820)
- Cardiolipin Biosynthesis CL(14:1(11Z)/14:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0008825)
- Cardiolipin Biosynthesis CL(14:1(11Z)/15:0/15:0/22:1(11Z)) (PathBank: SMP0008837)
- Cardiolipin Biosynthesis CL(14:1(11Z)/15:0/15:1(11Z)/22:1(11Z)) (PathBank: SMP0008858)
- Cardiolipin Biosynthesis CL(14:1(11Z)/15:0/15:1(9Z)/22:1(11Z)) (PathBank: SMP0008879)
- Cardiolipin Biosynthesis CL(14:1(11Z)/15:1(11Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0008945)
- Cardiolipin Biosynthesis CL(14:1(11Z)/15:1(11Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0008966)
- Cardiolipin Biosynthesis CL(14:1(11Z)/15:1(9Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0009032)
- Cardiolipin Biosynthesis CL(14:1(11Z)/15:1(9Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0009053)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/16:0/16:0) (PathBank: SMP0009111)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/16:0/22:1(11Z)) (PathBank: SMP0009115)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0009132)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0009149)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/18:0/22:1(11Z)) (PathBank: SMP0009164)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009180)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/20:0/22:1(11Z)) (PathBank: SMP0009189)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0009194)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0009199)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0009214)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0009231)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0009246)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0009257)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009268)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0009277)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0009282)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0009287)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0009302)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0009319)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0009334)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0009345)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009356)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0009365)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0009370)
- Cardiolipin Biosynthesis CL(14:1(11Z)/16:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0009375)
- Cardiolipin Biosynthesis CL(14:1(11Z)/18:0/18:0/22:1(11Z)) (PathBank: SMP0009386)
- Cardiolipin Biosynthesis CL(14:1(11Z)/18:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0009394)
- Cardiolipin Biosynthesis CL(14:1(11Z)/18:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009402)
- Cardiolipin Biosynthesis CL(14:1(11Z)/18:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0009421)
- Cardiolipin Biosynthesis CL(14:1(11Z)/18:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009429)
- Cardiolipin Biosynthesis CL(14:1(11Z)/18:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0009448)
- Cardiolipin Biosynthesis CL(14:1(11Z)/18:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009456)
- Cardiolipin Biosynthesis CL(14:1(11Z)/22:1(11Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0009477)
- Cardiolipin Biosynthesis CL(14:1(11Z)/22:1(11Z)/14:1(11Z)/22:1(9Z)) (PathBank: SMP0009478)
- Cardiolipin Biosynthesis CL(14:1(11Z)/22:1(11Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0009479)
- Cardiolipin Biosynthesis CL(14:1(11Z)/22:1(11Z)/14:1(9Z)/22:1(9Z)) (PathBank: SMP0009480)
- Cardiolipin Biosynthesis CL(14:1(11Z)/22:1(9Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0009481)
- Cardiolipin Biosynthesis CL(14:1(11Z)/22:1(9Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0009483)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0009492)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0009511)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0009559)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0009577)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0009595)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0009612)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0009624)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009636)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0009647)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0009653)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0009659)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(11Z)/22:0/22:1(11Z)) (PathBank: SMP0009664)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0009673)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0009692)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0009740)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0009758)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0009776)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0009793)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0009805)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0009817)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0009828)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0009834)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0009840)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/22:0/22:1(11Z)) (PathBank: SMP0009845)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:0/15:0/22:1(11Z)) (PathBank: SMP0009857)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:0/15:1(11Z)/22:1(11Z)) (PathBank: SMP0009878)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:0/15:1(9Z)/22:1(11Z)) (PathBank: SMP0009899)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:1(11Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0009965)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:1(11Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0009986)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:1(9Z)/15:1(11Z)/22:1(11Z)) (PathBank: SMP0010052)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:1(9Z)/15:1(9Z)/22:1(11Z)) (PathBank: SMP0010073)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/16:0/22:1(11Z)) (PathBank: SMP0010141)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0010158)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0010175)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/18:0/22:1(11Z)) (PathBank: SMP0010190)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010201)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010212)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/20:0/22:1(11Z)) (PathBank: SMP0010221)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0010226)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0010231)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0010246)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0010263)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0010278)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010289)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010300)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0010309)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0010314)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0010319)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0010334)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0010351)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0010366)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010377)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010388)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0010397)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0010402)
- Cardiolipin Biosynthesis CL(14:1(9Z)/16:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0010407)
- Cardiolipin Biosynthesis CL(14:1(9Z)/18:0/18:0/22:1(11Z)) (PathBank: SMP0010418)
- Cardiolipin Biosynthesis CL(14:1(9Z)/18:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010426)
- Cardiolipin Biosynthesis CL(14:1(9Z)/18:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010434)
- Cardiolipin Biosynthesis CL(14:1(9Z)/18:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010453)
- Cardiolipin Biosynthesis CL(14:1(9Z)/18:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010461)
- Cardiolipin Biosynthesis CL(14:1(9Z)/18:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010480)
- Cardiolipin Biosynthesis CL(14:1(9Z)/18:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010488)
- Cardiolipin Biosynthesis CL(14:1(9Z)/22:1(11Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0010509)
- Cardiolipin Biosynthesis CL(14:1(9Z)/22:1(11Z)/14:1(11Z)/22:1(9Z)) (PathBank: SMP0010510)
- Cardiolipin Biosynthesis CL(14:1(9Z)/22:1(11Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0010511)
- Cardiolipin Biosynthesis CL(14:1(9Z)/22:1(11Z)/14:1(9Z)/22:1(9Z)) (PathBank: SMP0010512)
- Cardiolipin Biosynthesis CL(14:1(9Z)/22:1(9Z)/14:1(11Z)/22:1(11Z)) (PathBank: SMP0010513)
- Cardiolipin Biosynthesis CL(14:1(9Z)/22:1(9Z)/14:1(9Z)/22:1(11Z)) (PathBank: SMP0010515)
- Cardiolipin Biosynthesis CL(15:0/15:0/16:0/22:1(11Z)) (PathBank: SMP0010553)
- Cardiolipin Biosynthesis CL(15:0/15:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0010568)
- Cardiolipin Biosynthesis CL(15:0/15:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0010583)
- Cardiolipin Biosynthesis CL(15:0/15:0/18:0/22:1(11Z)) (PathBank: SMP0010597)
- Cardiolipin Biosynthesis CL(15:0/15:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010606)
- Cardiolipin Biosynthesis CL(15:0/15:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010615)
- Cardiolipin Biosynthesis CL(15:0/15:0/20:0/22:1(11Z)) (PathBank: SMP0010623)
- Cardiolipin Biosynthesis CL(15:0/15:0/20:1(11Z)/22:1(11Z)) (PathBank: SMP0010626)
- Cardiolipin Biosynthesis CL(15:0/15:0/20:1(13Z)/22:1(11Z)) (PathBank: SMP0010629)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/16:0/16:0) (PathBank: SMP0010653)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0010663)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0010680)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0010697)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0010712)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010723)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010734)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0010743)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0010748)
- Cardiolipin Biosynthesis CL(15:0/15:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0010753)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/16:0/16:0) (PathBank: SMP0010777)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0010787)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0010804)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0010821)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0010836)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0010847)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0010858)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0010867)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0010872)
- Cardiolipin Biosynthesis CL(15:0/15:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0010877)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0010976)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0010991)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011006)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0011020)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011029)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011038)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0011046)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0011049)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0011052)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0011083)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011098)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011113)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0011127)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011136)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011145)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0011153)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0011156)
- Cardiolipin Biosynthesis CL(15:1(11Z)/15:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0011159)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/16:0/22:1(11Z)) (PathBank: SMP0011273)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011288)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011303)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0011317)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011326)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011335)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/20:0/22:1(11Z)) (PathBank: SMP0011343)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0011346)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(11Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0011349)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/16:0/22:1(11Z)) (PathBank: SMP0011380)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011395)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011410)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0011424)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011433)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011442)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/20:0/22:1(11Z)) (PathBank: SMP0011450)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/20:1(11Z)/22:1(11Z)) (PathBank: SMP0011453)
- Cardiolipin Biosynthesis CL(15:1(9Z)/15:1(9Z)/20:1(13Z)/22:1(11Z)) (PathBank: SMP0011456)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/16:1(11Z)) (PathBank: SMP0011542)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/22:1(11Z)) (PathBank: SMP0011551)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/22:1(9Z)) (PathBank: SMP0011552)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/24:0) (PathBank: SMP0011553)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/24:1(11Z)) (PathBank: SMP0011554)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/24:1(9Z)) (PathBank: SMP0011555)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/18:0) (PathBank: SMP0011556)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/18:1(11Z)) (PathBank: SMP0011557)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/18:1(9Z)) (PathBank: SMP0011558)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/20:0) (PathBank: SMP0011559)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/20:1(11Z)) (PathBank: SMP0011560)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/20:1(13Z)) (PathBank: SMP0011561)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/22:0) (PathBank: SMP0011562)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011563)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/22:1(9Z)) (PathBank: SMP0011564)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/24:0) (PathBank: SMP0011565)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/24:1(11Z)) (PathBank: SMP0011566)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(11Z)/24:1(9Z)) (PathBank: SMP0011567)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:1(13Z)) (PathBank: SMP0011573)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011575)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/22:1(9Z)) (PathBank: SMP0011576)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/24:0) (PathBank: SMP0011577)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/24:1(11Z)) (PathBank: SMP0011578)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/24:1(9Z)) (PathBank: SMP0011579)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/22:1(11Z)) (PathBank: SMP0011586)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/22:1(9Z)) (PathBank: SMP0011587)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011592)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/22:1(9Z)) (PathBank: SMP0011593)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011598)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/22:1(9Z)) (PathBank: SMP0011599)
- Cardiolipin Biosynthesis CL(16:0/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011613)
- Cardiolipin Biosynthesis CL(16:0/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011627)
- Cardiolipin Biosynthesis CL(16:0/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0011639)
- Cardiolipin Biosynthesis CL(16:0/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011647)
- Cardiolipin Biosynthesis CL(16:0/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011655)
- Cardiolipin Biosynthesis CL(16:0/16:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011675)
- Cardiolipin Biosynthesis CL(16:0/16:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011689)
- Cardiolipin Biosynthesis CL(16:0/16:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0011701)
- Cardiolipin Biosynthesis CL(16:0/16:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011709)
- Cardiolipin Biosynthesis CL(16:0/16:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011717)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011781)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011795)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0011806)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011812)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011818)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(9Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011831)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(9Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011845)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(9Z)/18:0/22:1(11Z)) (PathBank: SMP0011856)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(9Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011862)
- Cardiolipin Biosynthesis CL(16:1(11Z)/16:1(9Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011868)
- Cardiolipin Biosynthesis CL(16:1(9Z)/16:1(11Z)/16:1(11Z)/22:1(11Z)) (PathBank: SMP0011937)
- Cardiolipin Biosynthesis CL(16:1(9Z)/16:1(11Z)/16:1(9Z)/22:1(11Z)) (PathBank: SMP0011951)
- Cardiolipin Biosynthesis CL(16:1(9Z)/16:1(11Z)/18:0/22:1(11Z)) (PathBank: SMP0011962)
- Cardiolipin Biosynthesis CL(16:1(9Z)/16:1(11Z)/18:1(11Z)/22:1(11Z)) (PathBank: SMP0011968)
- Cardiolipin Biosynthesis CL(16:1(9Z)/16:1(11Z)/18:1(9Z)/22:1(11Z)) (PathBank: SMP0011974)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:1(13Z)) (PathBank: SMP0080914)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:1(11Z)) (PathBank: SMP0080920)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:1(13Z)) (PathBank: SMP0080921)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/22:0) (PathBank: SMP0080922)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/22:1(13Z)) (PathBank: SMP0080923)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:1(13Z)) (PathBank: SMP0080927)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(11Z)/20:1(13Z)) (PathBank: SMP0080936)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(13Z)/20:1(13Z)) (PathBank: SMP0080939)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(13Z)/22:0) (PathBank: SMP0080940)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(13Z)/22:1(13Z)) (PathBank: SMP0080941)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0083366)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0083367)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083370)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0083376)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0083379)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083380)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083383)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0083391)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0083392)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083395)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0083402)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083403)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083406)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0083412)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083413)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:1(11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083416)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0083421)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083422)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:2(9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083425)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0083429)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083430)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0083431)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0083432)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(6Z,9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083433)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0083436)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083437)
- Cardiolipin Biosynthesis CL(16:0/16:0/18:3(9Z,12Z,15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083440)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0083442)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0083443)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083446)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(5Z,8Z,11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0083447)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(5Z,8Z,11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083448)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(5Z,8Z,11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0083449)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(5Z,8Z,11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0083450)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(5Z,8Z,11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083451)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(8Z,11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0083452)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0083453)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0083454)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:3(8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083455)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083458)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083460)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:5(5Z,8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0083461)
- Cardiolipin Biosynthesis CL(14:0/15:0/16:0/16:0) (PathBank: SMP0096432)
- Cardiolipin Biosynthesis CL(14:1(9Z)/14:1(9Z)/16:0/16:0) (PathBank: SMP0097007)
- Cardiolipin Biosynthesis CL(14:1(9Z)/15:0/16:0/16:0) (PathBank: SMP0097118)
- Cardiolipin Biosynthesis CL(15:0/15:0/16:0/16:0) (PathBank: SMP0097678)
- Cardiolipin Biosynthesis CL(15:0/16:0/16:0/16:0) (PathBank: SMP0097769)
- Cardiolipin Biosynthesis CL(16:0/16:0/16:1(9Z)/22:1(13Z)) (PathBank: SMP0098248)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0098301)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:1(11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0098302)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/22:0) (PathBank: SMP0098307)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(5Z,8Z,11Z,14Z)/22:1(13Z)) (PathBank: SMP0098308)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(8Z,11Z,14Z,17Z)/22:0) (PathBank: SMP0098310)
- Cardiolipin Biosynthesis CL(16:0/16:0/20:4(8Z,11Z,14Z,17Z)/22:1(13Z)) (PathBank: SMP0098311)
- Lipid metabolism pathway (HMDB: HMDB0006968)
Cellular processBiochemical process |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | | Show more...
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesNot Available | Show more...
|---|