| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2023-05-30 20:55:55 UTC |
|---|
| HMDB ID | HMDB0000168 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | L-Asparagine |
|---|
| Description | Asparagine (Asn) or L-asparagine is an alpha-amino acid. These are amino acids in which the amino group is attached to the carbon atom immediately adjacent to the carboxylate group (alpha carbon). Amino acids are organic compounds that contain amino (-NH2) and carboxyl (-COOH) functional groups, along with a side chain (R group) specific to each amino acid. L-asparagine is one of 20 proteinogenic amino acids, i.e., the amino acids used in the biosynthesis of proteins. Asparagine is found in all organisms ranging from bacteria to plants to animals. In humans, asparagine is not an essential amino acid, which means that it can be synthesized from central metabolic pathway intermediates in humans and is not required in the diet. The precursor to asparagine is oxaloacetate. Oxaloacetate is converted to aspartate using a transaminase enzyme. This enzyme transfers the amino group from glutamate to oxaloacetate producing alpha-ketoglutarate and aspartate. The enzyme asparagine synthetase produces asparagine, AMP, glutamate, and pyrophosphate from aspartate, glutamine, and ATP. In the asparagine synthetase reaction, ATP is used to activate aspartate, forming beta-aspartyl-AMP. Glutamine donates an ammonium group which reacts with beta-aspartyl-AMP to form asparagine and free AMP. Since the asparagine side chain can make efficient hydrogen bond interactions with the peptide backbone, asparagines are often found near the beginning and end of alpha-helices, and in turn motifs in beta sheets. Its role can be thought as "capping" the hydrogen bond interactions which would otherwise need to be satisfied by the polypeptide backbone. Asparagine also provides key sites for N-linked glycosylation, a modification of the protein chain that is characterized by the addition of carbohydrate chains. A reaction between asparagine and reducing sugars or reactive carbonyls produces acrylamide (acrylic amide) in food when heated to sufficient temperature (i.e. baking). These occur primarily in baked goods such as French fries, potato chips, and roasted coffee. Asparagine was first isolated in 1806 from asparagus juice --hence its name. Asparagine was the first amino acid to be isolated. The smell observed in the urine of some individuals after the consumption of asparagus is attributed to a byproduct of the metabolic breakdown of asparagine, asparagine-amino-succinic-acid monoamide. However, some scientists disagree and implicate other substances in the smell, especially methanethiol. | Read more...
|---|
| Structure | InChI=1S/C4H8N2O3/c5-2(4(8)9)1-3(6)7/h2H,1,5H2,(H2,6,7)(H,8,9)/t2-/m0/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| (2S)-2,4-Diamino-4-oxobutanoic acid | ChEBI | | (2S)-2-Amino-3-carbamoylpropanoic acid | ChEBI | | (S)-2-Amino-3-carbamoylpropanoic acid | ChEBI | | (S)-Asparagine | ChEBI | | 2-Aminosuccinamic acid | ChEBI | | alpha-Aminosuccinamic acid | ChEBI | | Asn | ChEBI | | ASPARAGINE | ChEBI | | Aspartamic acid | ChEBI | | L-2-Aminosuccinamic acid | ChEBI | | L-Asparagin | ChEBI | | L-Aspartic acid beta-amide | ChEBI | | N | ChEBI | | (2S)-2,4-Diamino-4-oxobutanoate | Generator | | (2S)-2-Amino-3-carbamoylpropanoate | Generator | | (S)-2-Amino-3-carbamoylpropanoate | Generator | | 2-Aminosuccinamate | Generator | | a-Aminosuccinamate | Generator | | a-Aminosuccinamic acid | Generator | | alpha-Aminosuccinamate | Generator | | Α-aminosuccinamate | Generator | | Α-aminosuccinamic acid | Generator | | Aspartamate | Generator | | L-2-Aminosuccinamate | Generator | | L-Aspartate b-amide | Generator | | L-Aspartate beta-amide | Generator | | L-Aspartate β-amide | Generator | | L-Aspartic acid b-amide | Generator | | L-Aspartic acid β-amide | Generator | | (-)-Asparagine | HMDB | | (S)-2,4-Diamino-4-oxobutanoate | HMDB | | (S)-2,4-Diamino-4-oxobutanoic acid | HMDB | | Agedoite | HMDB | | alpha Amminosuccinamate | HMDB | | alpha Amminosuccinamic acid | HMDB | | Altheine | HMDB | | Asparagine acid | HMDB | | Asparamide | HMDB | | Aspartic acid amide | HMDB | | Aspartic acid b-amide | HMDB | | Aspartic acid beta amide | HMDB | | b2,4-(S)-Diamino-4-oxo-utanoate | HMDB | | b2,4-(S)-Diamino-4-oxo-utanoic acid | HMDB | | Crystal VI | HMDB | | L-2,4-Diamino-4-oxobutanoate | HMDB | | L-2,4-Diamino-4-oxobutanoic acid | HMDB | | L-Aspartamine | HMDB | | L-b-Asparagine | HMDB | | L-beta-Asparagine | HMDB |
| Show more...
|---|
| Chemical Formula | C4H8N2O3 |
|---|
| Average Molecular Weight | 132.1179 |
|---|
| Monoisotopic Molecular Weight | 132.053492132 |
|---|
| IUPAC Name | (2S)-2-amino-3-carbamoylpropanoic acid |
|---|
| Traditional Name | L-asparagine |
|---|
| CAS Registry Number | 70-47-3 |
|---|
| SMILES | N[C@@H](CC(N)=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C4H8N2O3/c5-2(4(8)9)1-3(6)7/h2H,1,5H2,(H2,6,7)(H,8,9)/t2-/m0/s1 |
|---|
| InChI Key | DCXYFEDJOCDNAF-REOHCLBHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as asparagine and derivatives. Asparagine and derivatives are compounds containing asparagine or a derivative thereof resulting from reaction of asparagine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Asparagine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Asparagine or derivatives
- Alpha-amino acid
- L-alpha-amino acid
- Fatty amide
- Fatty acyl
- Fatty acid
- Carboxamide group
- Amino acid
- Primary carboxylic acid amide
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic nitrogen compound
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Hydrocarbon derivative
- Primary aliphatic amine
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Amine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | 234 - 235 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 29.4 mg/mL | Not Available | | LogP | -3.82 | CHMELIK,J ET AL. (1991) |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | | Show more...
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| L-Asparagine,1TMS,isomer #1 | C[Si](C)(C)OC(=O)[C@@H](N)CC(N)=O | 1480.3 | Semi standard non polar | 33892256 | | L-Asparagine,1TMS,isomer #2 | C[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O | 1554.1 | Semi standard non polar | 33892256 | | L-Asparagine,1TMS,isomer #3 | C[Si](C)(C)NC(=O)C[C@H](N)C(=O)O | 1575.0 | Semi standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #1 | C[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O[Si](C)(C)C | 1577.0 | Semi standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #1 | C[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O[Si](C)(C)C | 1579.7 | Standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #1 | C[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O[Si](C)(C)C | 2388.1 | Standard polar | 33892256 | | L-Asparagine,2TMS,isomer #2 | C[Si](C)(C)NC(=O)C[C@H](N)C(=O)O[Si](C)(C)C | 1552.7 | Semi standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #2 | C[Si](C)(C)NC(=O)C[C@H](N)C(=O)O[Si](C)(C)C | 1668.8 | Standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #2 | C[Si](C)(C)NC(=O)C[C@H](N)C(=O)O[Si](C)(C)C | 2300.9 | Standard polar | 33892256 | | L-Asparagine,2TMS,isomer #3 | C[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C)C(=O)O | 1647.5 | Semi standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #3 | C[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C)C(=O)O | 1676.0 | Standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #3 | C[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C)C(=O)O | 2314.9 | Standard polar | 33892256 | | L-Asparagine,2TMS,isomer #4 | C[Si](C)(C)N([C@@H](CC(N)=O)C(=O)O)[Si](C)(C)C | 1734.3 | Semi standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #4 | C[Si](C)(C)N([C@@H](CC(N)=O)C(=O)O)[Si](C)(C)C | 1633.9 | Standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #4 | C[Si](C)(C)N([C@@H](CC(N)=O)C(=O)O)[Si](C)(C)C | 2584.6 | Standard polar | 33892256 | | L-Asparagine,2TMS,isomer #5 | C[Si](C)(C)N(C(=O)C[C@H](N)C(=O)O)[Si](C)(C)C | 1715.3 | Semi standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #5 | C[Si](C)(C)N(C(=O)C[C@H](N)C(=O)O)[Si](C)(C)C | 1689.0 | Standard non polar | 33892256 | | L-Asparagine,2TMS,isomer #5 | C[Si](C)(C)N(C(=O)C[C@H](N)C(=O)O)[Si](C)(C)C | 2797.8 | Standard polar | 33892256 | | L-Asparagine,3TMS,isomer #1 | C[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1668.6 | Semi standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #1 | C[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1696.3 | Standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #1 | C[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1922.2 | Standard polar | 33892256 | | L-Asparagine,3TMS,isomer #2 | C[Si](C)(C)OC(=O)[C@H](CC(N)=O)N([Si](C)(C)C)[Si](C)(C)C | 1729.5 | Semi standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #2 | C[Si](C)(C)OC(=O)[C@H](CC(N)=O)N([Si](C)(C)C)[Si](C)(C)C | 1687.5 | Standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #2 | C[Si](C)(C)OC(=O)[C@H](CC(N)=O)N([Si](C)(C)C)[Si](C)(C)C | 2273.3 | Standard polar | 33892256 | | L-Asparagine,3TMS,isomer #3 | C[Si](C)(C)OC(=O)[C@@H](N)CC(=O)N([Si](C)(C)C)[Si](C)(C)C | 1670.2 | Semi standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #3 | C[Si](C)(C)OC(=O)[C@@H](N)CC(=O)N([Si](C)(C)C)[Si](C)(C)C | 1739.7 | Standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #3 | C[Si](C)(C)OC(=O)[C@@H](N)CC(=O)N([Si](C)(C)C)[Si](C)(C)C | 2159.8 | Standard polar | 33892256 | | L-Asparagine,3TMS,isomer #4 | C[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)O | 1749.3 | Semi standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #4 | C[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)O | 1717.5 | Standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #4 | C[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2099.9 | Standard polar | 33892256 | | L-Asparagine,3TMS,isomer #5 | C[Si](C)(C)NC(=O)C[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 1799.7 | Semi standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #5 | C[Si](C)(C)NC(=O)C[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 1755.2 | Standard non polar | 33892256 | | L-Asparagine,3TMS,isomer #5 | C[Si](C)(C)NC(=O)C[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2065.8 | Standard polar | 33892256 | | L-Asparagine,4TMS,isomer #1 | C[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1767.5 | Semi standard non polar | 33892256 | | L-Asparagine,4TMS,isomer #1 | C[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1789.1 | Standard non polar | 33892256 | | L-Asparagine,4TMS,isomer #1 | C[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1838.9 | Standard polar | 33892256 | | L-Asparagine,4TMS,isomer #2 | C[Si](C)(C)NC(=O)C[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 1813.2 | Semi standard non polar | 33892256 | | L-Asparagine,4TMS,isomer #2 | C[Si](C)(C)NC(=O)C[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 1800.9 | Standard non polar | 33892256 | | L-Asparagine,4TMS,isomer #2 | C[Si](C)(C)NC(=O)C[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 1870.6 | Standard polar | 33892256 | | L-Asparagine,4TMS,isomer #3 | C[Si](C)(C)N(C(=O)C[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 1878.6 | Semi standard non polar | 33892256 | | L-Asparagine,4TMS,isomer #3 | C[Si](C)(C)N(C(=O)C[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 1849.9 | Standard non polar | 33892256 | | L-Asparagine,4TMS,isomer #3 | C[Si](C)(C)N(C(=O)C[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 1960.8 | Standard polar | 33892256 | | L-Asparagine,5TMS,isomer #1 | C[Si](C)(C)OC(=O)[C@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 1952.7 | Semi standard non polar | 33892256 | | L-Asparagine,5TMS,isomer #1 | C[Si](C)(C)OC(=O)[C@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 1906.5 | Standard non polar | 33892256 | | L-Asparagine,5TMS,isomer #1 | C[Si](C)(C)OC(=O)[C@H](CC(=O)N([Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 1798.7 | Standard polar | 33892256 | | L-Asparagine,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CC(N)=O | 1705.2 | Semi standard non polar | 33892256 | | L-Asparagine,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O | 1796.6 | Semi standard non polar | 33892256 | | L-Asparagine,1TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N)C(=O)O | 1813.3 | Semi standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O[Si](C)(C)C(C)(C)C | 2018.7 | Semi standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O[Si](C)(C)C(C)(C)C | 1977.5 | Standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CC(N)=O)C(=O)O[Si](C)(C)C(C)(C)C | 2436.3 | Standard polar | 33892256 | | L-Asparagine,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C | 1995.6 | Semi standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C | 2045.0 | Standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C | 2360.4 | Standard polar | 33892256 | | L-Asparagine,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C(C)(C)C)C(=O)O | 2106.3 | Semi standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C(C)(C)C)C(=O)O | 2028.6 | Standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C(C)(C)C)C(=O)O | 2338.6 | Standard polar | 33892256 | | L-Asparagine,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)N([C@@H](CC(N)=O)C(=O)O)[Si](C)(C)C(C)(C)C | 2171.1 | Semi standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)N([C@@H](CC(N)=O)C(=O)O)[Si](C)(C)C(C)(C)C | 2037.3 | Standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)N([C@@H](CC(N)=O)C(=O)O)[Si](C)(C)C(C)(C)C | 2521.8 | Standard polar | 33892256 | | L-Asparagine,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N(C(=O)C[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 2124.3 | Semi standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N(C(=O)C[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 2100.7 | Standard non polar | 33892256 | | L-Asparagine,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N(C(=O)C[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 2609.6 | Standard polar | 33892256 | | L-Asparagine,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2297.4 | Semi standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2256.1 | Standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC(=O)C[C@H](N[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2252.1 | Standard polar | 33892256 | | L-Asparagine,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CC(N)=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2388.8 | Semi standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CC(N)=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2311.7 | Standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CC(N)=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2427.8 | Standard polar | 33892256 | | L-Asparagine,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2310.9 | Semi standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2364.2 | Standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2357.8 | Standard polar | 33892256 | | L-Asparagine,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 2394.3 | Semi standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 2325.7 | Standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 2330.3 | Standard polar | 33892256 | | L-Asparagine,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)NC(=O)C[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2435.4 | Semi standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)NC(=O)C[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2322.4 | Standard non polar | 33892256 | | L-Asparagine,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)NC(=O)C[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2314.0 | Standard polar | 33892256 | | L-Asparagine,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2591.2 | Semi standard non polar | 33892256 | | L-Asparagine,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2534.1 | Standard non polar | 33892256 | | L-Asparagine,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2302.0 | Standard polar | 33892256 | | L-Asparagine,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)C[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2641.7 | Semi standard non polar | 33892256 | | L-Asparagine,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)C[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2528.0 | Standard non polar | 33892256 | | L-Asparagine,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)C[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2315.2 | Standard polar | 33892256 | | L-Asparagine,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N(C(=O)C[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2730.7 | Semi standard non polar | 33892256 | | L-Asparagine,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N(C(=O)C[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2606.8 | Standard non polar | 33892256 | | L-Asparagine,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N(C(=O)C[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2339.9 | Standard polar | 33892256 | | L-Asparagine,5TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2942.3 | Semi standard non polar | 33892256 | | L-Asparagine,5TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2795.8 | Standard non polar | 33892256 | | L-Asparagine,5TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2349.5 | Standard polar | 33892256 |
| Show more...
|---|
| Spectra |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (3 TMS) | splash10-0159-0910000000-bedf57998656ab5ebc16 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (3 TMS) | splash10-0159-0910000000-4979c4d028d2dc931263 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-0159-0910000000-b7ea3fef61f3940cbdda | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (3 TMS) | splash10-00di-9710000000-6d0afcbcc003347e598e | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (2 TMS) | splash10-0uxr-0910000000-2be567239bd3229b1ca1 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-02t9-0790000000-b8141f48cbebb90f683e | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-02t9-1790000000-1d12ed9b4fb2799da766 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (4 TMS) | splash10-014u-0961000000-65a5c0999f17110c9939 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (4 TMS) | splash10-00kr-0940000000-0e0e5c7bdbac5ea0e49e | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (3 TMS) | splash10-00lr-1920000000-7864dbb1f685e64dd1cf | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (4 TMS) | splash10-000i-1940000000-964ea25da2789805985a | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine EI-B (Non-derivatized) | splash10-00lr-0930000000-79a089d9875e092dd7f2 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Non-derivatized) | splash10-0159-0910000000-bedf57998656ab5ebc16 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Non-derivatized) | splash10-0159-0910000000-4979c4d028d2dc931263 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Non-derivatized) | splash10-0159-0910000000-b7ea3fef61f3940cbdda | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-QQ (Non-derivatized) | splash10-00di-4931100000-17f149310ce3a0f748d1 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-EI-TOF (Non-derivatized) | splash10-00di-9710000000-6d0afcbcc003347e598e | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-0uxr-0910000000-2be567239bd3229b1ca1 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-02t9-0790000000-b8141f48cbebb90f683e | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-02t9-1790000000-1d12ed9b4fb2799da766 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-014u-0961000000-65a5c0999f17110c9939 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-00kr-0940000000-0e0e5c7bdbac5ea0e49e | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-00lr-1920000000-7864dbb1f685e64dd1cf | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) | splash10-000i-1940000000-964ea25da2789805985a | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - L-Asparagine GC-MS (Non-derivatized) - 70eV, Positive | splash10-000f-9100000000-5881591331bd059b7e7d | 2016-09-22 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine Quattro_QQQ 10V, Positive-QTOF (Annotated) | splash10-0080-9400000000-e5c7e19f427eea6d71b0 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine Quattro_QQQ 25V, Positive-QTOF (Annotated) | splash10-00di-9000000000-afc1214100db1168b095 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine Quattro_QQQ 40V, Positive-QTOF (Annotated) | splash10-00dl-9000000000-57e977cd87e9a86d482b | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-001i-0900000000-6fb96f5aa291359dba29 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-000i-9000000000-76f11e6fe5657c35d15f | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-000i-9000000000-e944486273dbfde4cae4 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-001i-0900000000-27ad91a86be0c4d86c3c | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-001i-0900000000-b8b7a3431b66246ad613 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-000i-9000000000-03fa294ec740e189dd99 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-000i-9000000000-cee2081406fc25ab6169 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-001i-0900000000-cd79afa0c27f65e54adc | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-01q9-0943200000-5a24322ba0ce2f410155 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-03di-0900000000-fc3086cc1bb0c06a4c11 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-03di-3900000000-2d74d8232e7523f3b1c9 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-001i-0900000000-fa61c6fa9a87ce4905bb | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-01q9-0942200000-757da8b3406485c997b0 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-014i-9000000000-eac18512f2d22ab7a7ef | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-03di-0900000000-8df6d5b6cf94bf081c89 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-001i-0900000000-18079004a95c252da208 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative-QTOF | splash10-001i-0900000000-e6b1f9b4982e6cdc863d | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative-QTOF | splash10-03di-5900000000-7b9c09b3b6de28972f97 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative-QTOF | splash10-00dl-9100000000-2ea95ead344d62e230a4 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative-QTOF | splash10-0006-9000000000-defacc365589bc4437d7 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative-QTOF | splash10-0006-9000000000-fd60db4d5e35794c45ae | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - L-Asparagine LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive-QTOF | splash10-001i-3900000000-fd0f1034b5c1e40435c0 | 2012-08-31 | HMDB team, MONA | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Experimental 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, experimental) | 2021-10-10 | Wishart Lab | View Spectrum | | Experimental 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | 2021-10-10 | Wishart Lab | View Spectrum | | Experimental 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | 2012-12-05 | Wishart Lab | View Spectrum |
IR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M-H]-) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+H]+) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+Na]+) | 2023-02-03 | FELIX lab | View Spectrum |
| Show more...
|---|
| Biological Properties |
|---|
| Cellular Locations | - Cytoplasm
- Extracellular
- Mitochondria
|
|---|
| Biospecimen Locations | - Blood
- Breast Milk
- Cerebrospinal Fluid (CSF)
- Feces
- Saliva
- Sweat
- Urine
|
|---|
| Tissue Locations | - All Tissues
- Placenta
- Prostate
|
|---|
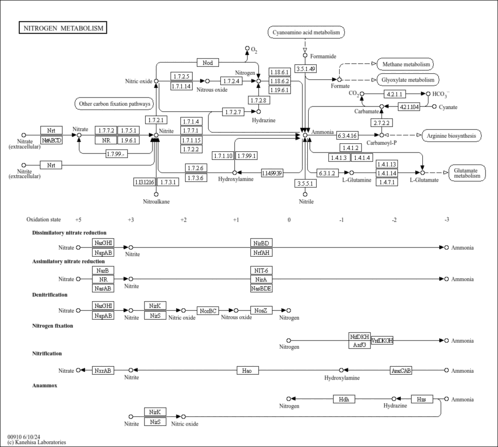
| Pathways | |
|---|
| Normal Concentrations |
|---|
| |
| Blood | Detected and Quantified | 36-87 uM | Newborn (0-30 days old) | Both | Normal | | details | | Blood | Detected and Quantified | 33-77 uM | Infant (1 - 3 months old) | Both | Normal | | details | | Blood | Detected and Quantified | 23-79 uM | Children (3 months - 6 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 25-75 uM | Children (6 - 18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 32.9 +/- 7.6 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 41.0 (31.0-51.0) uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 29-132 uM | Infant (0-1 year old) | Not Specified | Normal | | details | | Blood | Detected and Quantified | 61.7(12.09) uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 48.0 +/- 7.0 uM | Children (1-13 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 46.0 +/- 7.0 uM | Children (1-13 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 29.0-90.0 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 49.0 +/- 7.0 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 47.0 +/- 9.0 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 48.4 +/- 9.29 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 54.4(47.3-68.3) uM | Children (1-13 uears old) | Both | Normal | | details | | Blood | Detected and Quantified | 35.0 (16.4-57.2) uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 82.4 +/- 7.3 uM | Adult (>18 years old) | Both | Normal | | details | | Breast Milk | Detected and Quantified | 16.6 +/- 10.5 uM | Adult (>18 years old) | Female | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 4.43 (0.0-8.86) uM | Adult (>18 years old) | Both | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 19.0 +/- 11.1 uM | Children (1-13 years old) | Not Specified | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 8.1 +/- 4.9 uM | Adult (>18 years old) | Male | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 9.0 +/- 4.2 uM | Adult (>18 years old) | Female | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 5.4 +/- 1.4 uM | Adult (>18 years old) | Both | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 6.20 (5.90-6.50) uM | Adult (>18 years old) | Both | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 5.99 +/- 0.92 umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 4-10 uM | Children (0 - 10 years old) | Both | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 4-10 uM | Adolescent (>11 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Male | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Children (1-13 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Children (6 - 18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Children (6 - 18 years old) | Not Specified | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected and Quantified | 30 +/- 30 nmol/g wet feces | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Children (1-13 years old) | Not Specified | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Children (1-13 years old) | Not Specified | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Saliva | Detected and Quantified | 1.20 +/- 0.809 uM | Adult (>18 years old) | Not Specified | Normal | | details | | Saliva | Detected and Quantified | 1.24 +/- 0.768 uM | Adult (>18 years old) | Female | Normal | | details | | Saliva | Detected and Quantified | 1.70 +/- 1.69 uM | Adult (>18 years old) | Female | Normal | | details | | Saliva | Detected and Quantified | 1.74 +/- 0.972 uM | Adult (>18 years old) | Not Specified | Normal | | details | | Saliva | Detected and Quantified | 2.17 +/- 1.41 uM | Adult (>18 years old) | Both | Normal | | details | | Saliva | Detected and Quantified | 0.580 +/- 0.511 uM | Adult (>18 years old) | Female | Normal | | details | | Saliva | Detected and Quantified | 1.76 +/- 2.11 uM | Adult (>18 years old) | Female | Normal | | details | | Urine | Detected and Quantified | 8.75 umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 9.5 (3.0-26.0) umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 2.49-32.012 umol/mmol creatinine | Adolescent (13-18 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 4.66-16.53 umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 2.1-5.5 umol/mmol creatinine | Adult (>18 years old) | Female | Normal | | details | | Urine | Detected and Quantified | 2.83-26.92 umol/mmol creatinine | Adult (>18 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 15.15-53.01 umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0.96 +/- 0.65 umol/mmol creatinine | Infant (0-1 year old) | Both | Normal | | details | | Urine | Detected and Quantified | 2.83-113.12 umol/mmol creatinine | Infant (0-1 year old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 10.1 (4.6-17.8) umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 77.90 umol/mmol creatinine | Newborn (0-30 days old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 53.03 umol/mmol creatinine | Infant (1 - 6 months old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 32.00 umol/mmol creatinine | Infant (6 months - <1 year old) | Both | Normal | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 28.15 umol/mmol creatinine | Children (1 - 2 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 26.23 umol/mmol creatinine | Children (2 - 4 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 25.33 umol/mmol creatinine | Children (4 - 13 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 19.56 umol/mmol creatinine | Adolescent (13 - 21 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0 - 19.56 umol/mmol creatinine | Adult (>21 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 25.158 +/- 23.462 umol/mmol creatinine | Children (1 - 13 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | <3.0 umol/mmol creatinine | Infant (0-1 year old) | Both | Normal | | details | | Urine | Detected and Quantified | <4.0 umol/mmol creatinine | Infant (1 - 6 months old) | Both | Normal | | details | | Urine | Detected and Quantified | 7.013-100.00 umol/mmol creatinine | Children (1-13 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | <5.0 umol/mmol creatinine | Newborn (0-30 days old) | Both | Normal | | details | | Urine | Detected and Quantified | 4.296-155.231 umol/mmol creatinine | Infant (0-1 year old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 10.52 +/- 3.85 umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 2.5-3.7 umol/mmol creatinine | Adult (>18 years old) | Male | Normal | | details | | Urine | Detected and Quantified | 8.8 (4.6-17.7) umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 12.77 (7.96-24.28) umol/mmol creatinine | Newborn (0-30 days old) | Both | Normal | | details | | Urine | Detected and Quantified | 35.7098 +/- 12.6254 umol/mmol creatinine | Children (1 - 13 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 3.17-46.60 umol/mmol creatinine | Children (1-13 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 10.0 (4.6-16.32) umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 9.211 (3.289-15.1) umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 0.42-10.5 umol/mmol creatinine | Newborn (0-30 days old) | Both | Normal | | details | | Urine | Detected and Quantified | 2.81(1.84-5.77) umol/mmol creatinine | Newborn (0-30 days old) | Female | Normal | | details | | Urine | Detected and Quantified | 4.30-44.79 umol/mmol creatinine | Children (1-13 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 2.68(0.36-10.57) umol/mmol creatinine | Newborn (0-30 days old) | Male | Normal | | details |
| Show more...
|---|
| Abnormal Concentrations |
|---|
| |
| Blood | Detected and Quantified | 25.6 (10.3) uM | Adult (>18 years old) | Female | Pregnancy | | details | | Blood | Detected and Quantified | 54.1 +/- 21.7 uM | Adult (>18 years old) | Both | Heart Transplant | | details | | Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Colorectal cancer | | details | | Blood | Detected and Quantified | 39.13 +/- 16.35 uM | Adult (>18 years old) | Female | Pregnancy with fetuses with trisomy 18 | | details | | Blood | Detected and Quantified | 31.89 +/- 13.6 uM | Adult (>18 years old) | Female | Pregnancy | | details | | Blood | Detected and Quantified | 36.5 (17.2) uM | Adult (>18 years old) | Female | Early preeclampsia | | details | | Blood | Detected and Quantified | 34.5 (14.5) uM | Adult (>18 years old) | Female | Pregnancy | | details | | Blood | Detected and Quantified | 40.8 +/- 11.5 uM | Children (1-13 years old) | Both | Uremia | | details | | Blood | Detected and Quantified | 39.9 +/- 3.2 uM | Adult (>18 years old) | Both | Epilepsy | | details | | Blood | Detected and Quantified | 99 uM | Infant (0-1 year old) | Male | Fumaric aciduria | | details | | Blood | Detected and Quantified | 56.7(42.3-70.3) uM | Children (1-13 uears old) | Both | Environmental enteric dysfunction | | details | | Blood | Detected and Quantified | 69.29(13.01) uM | Adult (>18 years old) | Both | Heart failure with preserved ejection fraction | | details | | Blood | Detected and Quantified | 31.3 (11.3) uM | Adult (>18 years old) | Female | Late-onset preeclampsia | | details | | Blood | Detected and Quantified | 32.4 (13.7) uM | Adult (>18 years old) | Female | Pregnancy | | details | | Blood | Detected and Quantified | 27.3 (9.6) uM | Adult (>18 years old) | Female | Down syndrome pregnancy | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 7.9 +/- 6.4 uM | Children (1-13 years old) | Not Specified | Leukemia | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 11.1 +/- 6.7 uM | Children (1-13 years old) | Not Specified | leukemia | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 6.01 +/- 0.65 uM | Adult (>18 years old) | Both | Schizophrenia | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 1.77 +/- 0.16 umol/mmol creatinine | Adult (>18 years old) | Both | Alzheimer's disease | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Colorectal cancer | | details | | Feces | Detected but not Quantified | Not Quantified | Children (1-13 years old) | Both | celiac disease | | details | | Feces | Detected but not Quantified | Not Quantified | Children (6 - 18 years old) | Both | Crohns disease | | details | | Feces | Detected but not Quantified | Not Quantified | Children (6 - 18 years old) | Both | Ulcerative colitis | | details | | Feces | Detected but not Quantified | Not Quantified | Children (6 - 18 years old) | Not Specified | Crohns disease | | details | | Feces | Detected but not Quantified | Not Quantified | Children (6 - 18 years old) | Not Specified | Ulcerative colitis | | details | | Feces | Detected but not Quantified | Not Quantified | Children (6 - 18 years old) | Not Specified | Unclassified IBD | | details | | Feces | Detected but not Quantified | Not Quantified | Children (1-13 years old) | Not Specified | celiac disease | | details | | Feces | Detected but not Quantified | Not Quantified | Children (1-13 years old) | Not Specified | Treated celiac disease | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Crohn's disease | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Colorectal cancer | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Male | Gout | | details | | Saliva | Detected and Quantified | 1.68 +/- 2.90 uM | Adult (>18 years old) | Male | Alzheimer's disease | | details | | Saliva | Detected and Quantified | 0.97 +/- 1.12 uM | Adult (>18 years old) | Male | Frontotemporal lobe dementia | | details | | Saliva | Detected and Quantified | 3.02 +/- 0.83 uM | Adult (>18 years old) | Both | Lewy body disease | | details | | Sweat | Detected but not Quantified | Not Quantified | Infant (0-1 year old) | Not Specified | screen-positive CF | | details | | Urine | Detected and Quantified | 35.098 +/- 22.947 umol/mmol creatinine | Children (1 - 13 years old) | Not Specified | Eosinophilic esophagitis | | details | | Urine | Detected and Quantified | 20.700-447 umol/mmol creatinine | Infant (0-1 year old) | Not Specified | Fumaric aciduria | | details | | Urine | Detected and Quantified | 29.6797 +/- 15.0964 umol/mmol creatinine | Children (1 - 13 years old) | Not Specified | Eosinophilic esophagitis | | details | | Urine | Detected and Quantified | 28.0762 +/- 31.9291 umol/mmol creatinine | Children (1 - 13 years old) | Not Specified | Gastroesophageal reflux disease | | details |
| Show more...
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | | Epilepsy |
|---|
- Rainesalo S, Keranen T, Palmio J, Peltola J, Oja SS, Saransaari P: Plasma and cerebrospinal fluid amino acids in epileptic patients. Neurochem Res. 2004 Jan;29(1):319-24. [PubMed:14992292 ]
| | Uremia |
|---|
- Canepa A, Filho JC, Gutierrez A, Carrea A, Forsberg AM, Nilsson E, Verrina E, Perfumo F, Bergstrom J: Free amino acids in plasma, red blood cells, polymorphonuclear leukocytes, and muscle in normal and uraemic children. Nephrol Dial Transplant. 2002 Mar;17(3):413-21. [PubMed:11865086 ]
| | Colorectal cancer |
|---|
- Ni Y, Xie G, Jia W: Metabonomics of human colorectal cancer: new approaches for early diagnosis and biomarker discovery. J Proteome Res. 2014 Sep 5;13(9):3857-70. doi: 10.1021/pr500443c. Epub 2014 Aug 14. [PubMed:25105552 ]
- Sinha R, Ahn J, Sampson JN, Shi J, Yu G, Xiong X, Hayes RB, Goedert JJ: Fecal Microbiota, Fecal Metabolome, and Colorectal Cancer Interrelations. PLoS One. 2016 Mar 25;11(3):e0152126. doi: 10.1371/journal.pone.0152126. eCollection 2016. [PubMed:27015276 ]
- Goedert JJ, Sampson JN, Moore SC, Xiao Q, Xiong X, Hayes RB, Ahn J, Shi J, Sinha R: Fecal metabolomics: assay performance and association with colorectal cancer. Carcinogenesis. 2014 Sep;35(9):2089-96. doi: 10.1093/carcin/bgu131. Epub 2014 Jul 18. [PubMed:25037050 ]
| | Early preeclampsia |
|---|
- Bahado-Singh RO, Akolekar R, Mandal R, Dong E, Xia J, Kruger M, Wishart DS, Nicolaides K: Metabolomics and first-trimester prediction of early-onset preeclampsia. J Matern Fetal Neonatal Med. 2012 Oct;25(10):1840-7. doi: 10.3109/14767058.2012.680254. Epub 2012 Apr 28. [PubMed:22494326 ]
| | Pregnancy |
|---|
- Bahado-Singh RO, Akolekar R, Mandal R, Dong E, Xia J, Kruger M, Wishart DS, Nicolaides K: Metabolomics and first-trimester prediction of early-onset preeclampsia. J Matern Fetal Neonatal Med. 2012 Oct;25(10):1840-7. doi: 10.3109/14767058.2012.680254. Epub 2012 Apr 28. [PubMed:22494326 ]
- Bahado-Singh RO, Akolekar R, Mandal R, Dong E, Xia J, Kruger M, Wishart DS, Nicolaides K: First-trimester metabolomic detection of late-onset preeclampsia. Am J Obstet Gynecol. 2013 Jan;208(1):58.e1-7. doi: 10.1016/j.ajog.2012.11.003. Epub 2012 Nov 13. [PubMed:23159745 ]
- Bahado-Singh RO, Akolekar R, Mandal R, Dong E, Xia J, Kruger M, Wishart DS, Nicolaides K: Metabolomic analysis for first-trimester Down syndrome prediction. Am J Obstet Gynecol. 2013 May;208(5):371.e1-8. doi: 10.1016/j.ajog.2012.12.035. Epub 2013 Jan 8. [PubMed:23313728 ]
- Bahado-Singh RO, Akolekar R, Chelliah A, Mandal R, Dong E, Kruger M, Wishart DS, Nicolaides K: Metabolomic analysis for first-trimester trisomy 18 detection. Am J Obstet Gynecol. 2013 Jul;209(1):65.e1-9. doi: 10.1016/j.ajog.2013.03.028. Epub 2013 Mar 25. [PubMed:23535240 ]
| | Late-onset preeclampsia |
|---|
- Bahado-Singh RO, Akolekar R, Mandal R, Dong E, Xia J, Kruger M, Wishart DS, Nicolaides K: First-trimester metabolomic detection of late-onset preeclampsia. Am J Obstet Gynecol. 2013 Jan;208(1):58.e1-7. doi: 10.1016/j.ajog.2012.11.003. Epub 2012 Nov 13. [PubMed:23159745 ]
| | Fumarase deficiency |
|---|
- Allegri G, Fernandes MJ, Scalco FB, Correia P, Simoni RE, Llerena JC Jr, de Oliveira ML: Fumaric aciduria: an overview and the first Brazilian case report. J Inherit Metab Dis. 2010 Aug;33(4):411-9. doi: 10.1007/s10545-010-9134-2. Epub 2010 Jun 15. [PubMed:20549362 ]
| | Leukemia |
|---|
- Peng CT, Wu KH, Lan SJ, Tsai JJ, Tsai FJ, Tsai CH: Amino acid concentrations in cerebrospinal fluid in children with acute lymphoblastic leukemia undergoing chemotherapy. Eur J Cancer. 2005 May;41(8):1158-63. Epub 2005 Apr 14. [PubMed:15911239 ]
| | Schizophrenia |
|---|
- Do KQ, Lauer CJ, Schreiber W, Zollinger M, Gutteck-Amsler U, Cuenod M, Holsboer F: gamma-Glutamylglutamine and taurine concentrations are decreased in the cerebrospinal fluid of drug-naive patients with schizophrenic disorders. J Neurochem. 1995 Dec;65(6):2652-62. [PubMed:7595563 ]
| | Alzheimer's disease |
|---|
- Fonteh AN, Harrington RJ, Tsai A, Liao P, Harrington MG: Free amino acid and dipeptide changes in the body fluids from Alzheimer's disease subjects. Amino Acids. 2007 Feb;32(2):213-24. Epub 2006 Oct 10. [PubMed:17031479 ]
- Tsuruoka M, Hara J, Hirayama A, Sugimoto M, Soga T, Shankle WR, Tomita M: Capillary electrophoresis-mass spectrometry-based metabolome analysis of serum and saliva from neurodegenerative dementia patients. Electrophoresis. 2013 Oct;34(19):2865-72. doi: 10.1002/elps.201300019. Epub 2013 Sep 6. [PubMed:23857558 ]
| | Celiac disease |
|---|
- Di Cagno R, De Angelis M, De Pasquale I, Ndagijimana M, Vernocchi P, Ricciuti P, Gagliardi F, Laghi L, Crecchio C, Guerzoni ME, Gobbetti M, Francavilla R: Duodenal and faecal microbiota of celiac children: molecular, phenotype and metabolome characterization. BMC Microbiol. 2011 Oct 4;11:219. doi: 10.1186/1471-2180-11-219. [PubMed:21970810 ]
- De Angelis M, Vannini L, Di Cagno R, Cavallo N, Minervini F, Francavilla R, Ercolini D, Gobbetti M: Salivary and fecal microbiota and metabolome of celiac children under gluten-free diet. Int J Food Microbiol. 2016 Dec 19;239:125-132. doi: 10.1016/j.ijfoodmicro.2016.07.025. Epub 2016 Jul 19. [PubMed:27452636 ]
| | Crohn's disease |
|---|
- Marchesi JR, Holmes E, Khan F, Kochhar S, Scanlan P, Shanahan F, Wilson ID, Wang Y: Rapid and noninvasive metabonomic characterization of inflammatory bowel disease. J Proteome Res. 2007 Feb;6(2):546-51. [PubMed:17269711 ]
- Kolho KL, Pessia A, Jaakkola T, de Vos WM, Velagapudi V: Faecal and Serum Metabolomics in Paediatric Inflammatory Bowel Disease. J Crohns Colitis. 2017 Mar 1;11(3):321-334. doi: 10.1093/ecco-jcc/jjw158. [PubMed:27609529 ]
| | Ulcerative colitis |
|---|
- Kolho KL, Pessia A, Jaakkola T, de Vos WM, Velagapudi V: Faecal and Serum Metabolomics in Paediatric Inflammatory Bowel Disease. J Crohns Colitis. 2017 Mar 1;11(3):321-334. doi: 10.1093/ecco-jcc/jjw158. [PubMed:27609529 ]
| | Gout |
|---|
- Shao T, Shao L, Li H, Xie Z, He Z, Wen C: Combined Signature of the Fecal Microbiome and Metabolome in Patients with Gout. Front Microbiol. 2017 Feb 21;8:268. doi: 10.3389/fmicb.2017.00268. eCollection 2017. [PubMed:28270806 ]
| | Frontotemporal dementia |
|---|
- Tsuruoka M, Hara J, Hirayama A, Sugimoto M, Soga T, Shankle WR, Tomita M: Capillary electrophoresis-mass spectrometry-based metabolome analysis of serum and saliva from neurodegenerative dementia patients. Electrophoresis. 2013 Oct;34(19):2865-72. doi: 10.1002/elps.201300019. Epub 2013 Sep 6. [PubMed:23857558 ]
| | Lewy body disease |
|---|
- Tsuruoka M, Hara J, Hirayama A, Sugimoto M, Soga T, Shankle WR, Tomita M: Capillary electrophoresis-mass spectrometry-based metabolome analysis of serum and saliva from neurodegenerative dementia patients. Electrophoresis. 2013 Oct;34(19):2865-72. doi: 10.1002/elps.201300019. Epub 2013 Sep 6. [PubMed:23857558 ]
| | Cystic fibrosis |
|---|
- Adriana Nori de Macedo. Robust capillary electrophoresis methods for biomarker discovery and routine measurements in clinical and epidemiological applications. March 2017 [Link]
| | Eosinophilic esophagitis |
|---|
- Slae, M., Huynh, H., Wishart, D.S. (2014). Analysis of 30 normal pediatric urine samples via NMR spectroscopy (unpublished work). NA.
|
|
|---|
| Associated OMIM IDs | |
|---|
| External Links |
|---|
| DrugBank ID | DB00174 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | FDB000787 |
|---|
| KNApSAcK ID | C00001341 |
|---|
| Chemspider ID | 6031 |
|---|
| KEGG Compound ID | C00152 |
|---|
| BioCyc ID | ASN |
|---|
| BiGG ID | 34055 |
|---|
| Wikipedia Link | Asparagine |
|---|
| METLIN ID | 14 |
|---|
| PubChem Compound | 6267 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17196 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | ASN_L |
|---|
| MarkerDB ID | MDB00000082 |
|---|
| Good Scents ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Wang, Fangda. Preparation of L-b-asparagine. Faming Zhuanli Shenqing Gongkai Shuomingshu (2005), 8 pp. |
|---|
| Material Safety Data Sheet (MSDS) | Not Available |
|---|
| General References | - Rip JW, Coulter-Mackie MB, Rupar CA, Gordon BA: Purification and structure of human liver aspartylglucosaminidase. Biochem J. 1992 Dec 15;288 ( Pt 3):1005-10. [PubMed:1281977 ]
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [PubMed:19212411 ]
- Hagenfeldt L, Bjerkenstedt L, Edman G, Sedvall G, Wiesel FA: Amino acids in plasma and CSF and monoamine metabolites in CSF: interrelationship in healthy subjects. J Neurochem. 1984 Mar;42(3):833-7. [PubMed:6198473 ]
- Peng CT, Wu KH, Lan SJ, Tsai JJ, Tsai FJ, Tsai CH: Amino acid concentrations in cerebrospinal fluid in children with acute lymphoblastic leukemia undergoing chemotherapy. Eur J Cancer. 2005 May;41(8):1158-63. Epub 2005 Apr 14. [PubMed:15911239 ]
- Cynober LA: Plasma amino acid levels with a note on membrane transport: characteristics, regulation, and metabolic significance. Nutrition. 2002 Sep;18(9):761-6. [PubMed:12297216 ]
- Rainesalo S, Keranen T, Palmio J, Peltola J, Oja SS, Saransaari P: Plasma and cerebrospinal fluid amino acids in epileptic patients. Neurochem Res. 2004 Jan;29(1):319-24. [PubMed:14992292 ]
- Starczynowski DT, Reynolds JG, Gilmore TD: Mutations of tumor necrosis factor alpha-responsive serine residues within the C-terminal transactivation domain of human transcription factor REL enhance its in vitro transforming ability. Oncogene. 2005 Nov 10;24(49):7355-68. [PubMed:16027730 ]
- Fischer D, Schroers A, Blumcke I, Urbach H, Zerres K, Mortier W, Vorgerd M, Schroder R: Consequences of a novel caveolin-3 mutation in a large German family. Ann Neurol. 2003 Feb;53(2):233-41. [PubMed:12557291 ]
- Filho JC, Bergstrom J, Stehle P, Furst P: Simultaneous measurements of free amino acid patterns of plasma, muscle and erythrocytes in healthy human subjects. Clin Nutr. 1997 Dec;16(6):299-305. [PubMed:16844612 ]
- Sun S, Han J, Ralph WM Jr, Chandrasekaran A, Liu K, Auborn KJ, Carter TH: Endoplasmic reticulum stress as a correlate of cytotoxicity in human tumor cells exposed to diindolylmethane in vitro. Cell Stress Chaperones. 2004 Mar;9(1):76-87. [PubMed:15270080 ]
- Takamatsu S, Inoue N, Katsumata T, Nakamura K, Fujibayashi Y, Takeuchi M: The relationship between the branch-forming glycosyltransferases and cell surface sugar chain structures. Biochemistry. 2005 Apr 26;44(16):6343-9. [PubMed:15835923 ]
- Chiara F, Goumans MJ, Forsberg H, Ahgren A, Rasola A, Aspenstrom P, Wernstedt C, Hellberg C, Heldin CH, Heuchel R: A gain of function mutation in the activation loop of platelet-derived growth factor beta-receptor deregulates its kinase activity. J Biol Chem. 2004 Oct 8;279(41):42516-27. Epub 2004 Jul 28. [PubMed:15284236 ]
- Xu L, Wang Y, Gillespie D, Meissner G: Two rings of negative charges in the cytosolic vestibule of type-1 ryanodine receptor modulate ion fluxes. Biophys J. 2006 Jan 15;90(2):443-53. Epub 2005 Oct 20. [PubMed:16239337 ]
- Poon CJ, Plaas AH, Keene DR, McQuillan DJ, Last K, Fosang AJ: N-linked keratan sulfate in the aggrecan interglobular domain potentiates aggrecanase activity. J Biol Chem. 2005 Jun 24;280(25):23615-21. Epub 2005 Apr 22. [PubMed:15849197 ]
- Ahlman B, Andersson K, Leijonmarck CE, Ljungqvist O, Hedenborg L, Wernerman J: Short-term starvation alters the free amino acid content of the human intestinal mucosa. Clin Sci (Lond). 1994 Jun;86(6):653-62. [PubMed:7914846 ]
- Avramis VI, Panosyan EH: Pharmacokinetic/pharmacodynamic relationships of asparaginase formulations: the past, the present and recommendations for the future. Clin Pharmacokinet. 2005;44(4):367-93. [PubMed:15828851 ]
- Mohrmann K, van Eijndhoven MA, Schinkel AH, Schellens JH: Absence of N-linked glycosylation does not affect plasma membrane localization of breast cancer resistance protein (BCRP/ABCG2). Cancer Chemother Pharmacol. 2005 Oct;56(4):344-50. Epub 2005 May 5. [PubMed:15875186 ]
- Elshenawy S, Pinney SE, Stuart T, Doulias PT, Zura G, Parry S, Elovitz MA, Bennett MJ, Bansal A, Strauss JF 3rd, Ischiropoulos H, Simmons RA: The Metabolomic Signature of the Placenta in Spontaneous Preterm Birth. Int J Mol Sci. 2020 Feb 4;21(3). pii: ijms21031043. doi: 10.3390/ijms21031043. [PubMed:32033212 ]
| Show more...
|---|