| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected but not Quantified |
|---|
| Creation Date | 2007-05-22 21:09:59 UTC |
|---|
| Update Date | 2022-09-22 18:34:19 UTC |
|---|
| HMDB ID | HMDB0006281 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
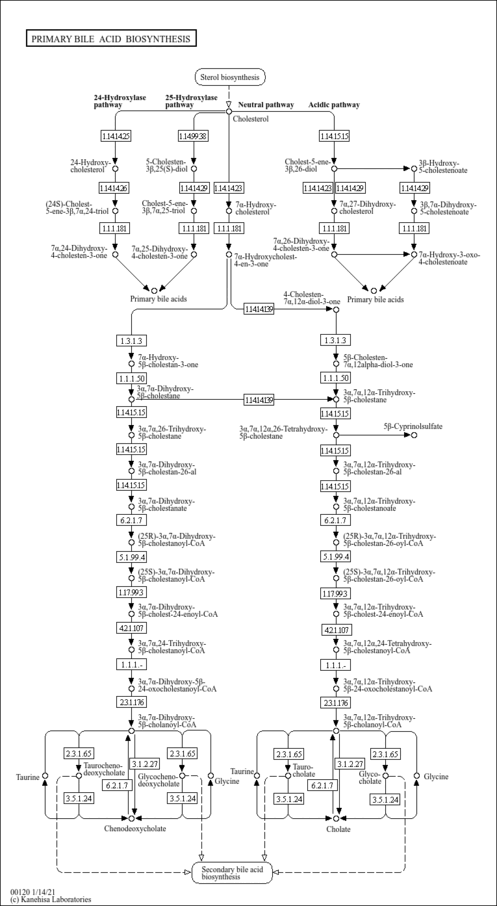
| Common Name | 7-a,27-Dihydroxycholesterol |
|---|
| Description | 7-a,27-Dihydroxycholesterol, also known as cholest-5-ene-3b,7a,26-triol, belongs to the class of organic compounds known as trihydroxy bile acids, alcohols and derivatives. These are prenol lipids structurally characterized by a bile acid or alcohol which bears three hydroxyl groups. Thus, 7-a,27-dihydroxycholesterol is considered to be a bile acid. Based on a literature review very few articles have been published on 7-a,27-Dihydroxycholesterol. |
|---|
| Structure | [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCCC(C)CO InChI=1S/C27H46O3/c1-17(16-28)6-5-7-18(2)21-8-9-22-25-23(11-13-27(21,22)4)26(3)12-10-20(29)14-19(26)15-24(25)30/h15,17-18,20-25,28-30H,5-14,16H2,1-4H3/t17?,18-,20+,21-,22+,23+,24-,25+,26+,27-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 5-Cholestene-3beta,7alpha,26-triol | ChEBI | | 7-alpha,27-Dihydroxycholesterol | ChEBI | | Cholest-5-ene-3beta,7alpha,26-triol | ChEBI | | Cholest-5-ene-3beta,7alpha,27-triol | ChEBI | | 5-Cholestene-3b,7a,26-triol | Generator | | 5-Cholestene-3β,7α,26-triol | Generator | | 7-Α,27-dihydroxycholesterol | Generator | | Cholest-5-ene-3b,7a,26-triol | Generator | | Cholest-5-ene-3β,7α,26-triol | Generator | | Cholest-5-ene-3b,7a,27-triol | Generator | | Cholest-5-ene-3β,7α,27-triol | Generator | | Cholest-5-ene-3,7,27-triol | MeSH | | 7alpha,26-Dihydroxycholesterol | HMDB | | 7alpha,27-Dihydroxycholesterol | HMDB | | Cholest-5-ene-3- b,7-a,27-triol | HMDB | | Cholest-5-ene-3-b,7-a,27-triol | HMDB | | Cholest-5-ene-3-beta,7-alpha,27-triol | HMDB |
| Show more...
|---|
| Chemical Formula | C27H46O3 |
|---|
| Average Molecular Weight | 418.6523 |
|---|
| Monoisotopic Molecular Weight | 418.344695338 |
|---|
| IUPAC Name | (1S,2R,5S,9S,10S,11S,14R,15R)-14-[(2R)-7-hydroxy-6-methylheptan-2-yl]-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-7-ene-5,9-diol |
|---|
| Traditional Name | 7α,27-dihydroxycholesterol |
|---|
| CAS Registry Number | 4725-24-0 |
|---|
| SMILES | [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCCC(C)CO |
|---|
| InChI Identifier | InChI=1S/C27H46O3/c1-17(16-28)6-5-7-18(2)21-8-9-22-25-23(11-13-27(21,22)4)26(3)12-10-20(29)14-19(26)15-24(25)30/h15,17-18,20-25,28-30H,5-14,16H2,1-4H3/t17?,18-,20+,21-,22+,23+,24-,25+,26+,27-/m1/s1 |
|---|
| InChI Key | RXMHNAKZMGJANZ-DTTSCKGMSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as trihydroxy bile acids, alcohols and derivatives. These are prenol lipids structurally characterized by a bile acid or alcohol which bears three hydroxyl groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Bile acids, alcohols and derivatives |
|---|
| Direct Parent | Trihydroxy bile acids, alcohols and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - 26-hydroxysteroid
- Trihydroxy bile acid, alcohol, or derivatives
- 3-hydroxy-delta-5-steroid
- 3-hydroxysteroid
- 7-hydroxysteroid
- 3-beta-hydroxysteroid
- 3-beta-hydroxy-delta-5-steroid
- Hydroxysteroid
- Delta-5-steroid
- Fatty alcohol
- Fatty acyl
- Cyclic alcohol
- Secondary alcohol
- Primary alcohol
- Organic oxygen compound
- Alcohol
- Hydrocarbon derivative
- Organooxygen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | | Show more...
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 7-a,27-Dihydroxycholesterol,1TMS,isomer #1 | CC(CO)CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C | 3496.1 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,1TMS,isomer #2 | CC(CO)CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C | 3501.4 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,1TMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C | 3505.3 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,2TMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C | 3467.5 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,2TMS,isomer #2 | CC(CO)CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C | 3415.3 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,2TMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C | 3502.9 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,3TMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C | 3399.3 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,1TBDMS,isomer #1 | CC(CO)CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C | 3730.5 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,1TBDMS,isomer #2 | CC(CO)CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C | 3723.0 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,1TBDMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C(C)(C)C | 3765.5 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,2TBDMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C(C)(C)C | 3957.7 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,2TBDMS,isomer #2 | CC(CO)CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C | 3878.7 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,2TBDMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C(C)(C)C | 3983.9 | Semi standard non polar | 33892256 | | 7-a,27-Dihydroxycholesterol,3TBDMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)CO[Si](C)(C)C(C)(C)C | 4106.7 | Semi standard non polar | 33892256 |
| Show more...
|---|