| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2023-05-30 20:55:56 UTC |
|---|
| HMDB ID | HMDB0000707 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 4-Hydroxyphenylpyruvic acid |
|---|
| Description | 4-Hydroxyphenylpyruvic acid (4-HPPA) is a keto acid that is involved in the tyrosine catabolism pathway. It is a product of the enzyme (R)-4-hydroxyphenyllactate dehydrogenase (EC 1.1.1.222) and is formed during tyrosine metabolism. The conversion from tyrosine to 4-HPPA is catalyzed by tyrosine aminotransferase. Additionally, 4-HPPA can be converted to homogentisic acid which is one of the precursors to ochronotic pigment. The enzyme 4-hydroxyphenylpyruvic acid dioxygenase (HPD) catalyzes the reaction that converts 4-hydroxyphenylpyruvic acid to homogentisic acid. A deficiency in the catalytic activity of HPD is known to lead to tyrosinemia type III, an autosomal recessive disorder characterized by elevated levels of blood tyrosine and massive excretion of tyrosine derivatives into urine. It has been shown that hawkinsinuria, an autosomal dominant disorder characterized by the excretion of 'hawkinsin,' may also be a result of HPD deficiency (PMID: 11073718 ). Moreover, 4-hydroxyphenylpyruvic acid is also found to be associated in phenylketonuria, which is also an inborn error of metabolism. There are two isomers of HPPA, specifically 4HPPA and 3HPPA, of which 4HPPA is the most common. 4-HPPA has been found to be a microbial metabolite in Escherichia (ECMDB). |
|---|
| Structure | OC(=O)C(=O)CC1=CC=C(O)C=C1 InChI=1S/C9H8O4/c10-7-3-1-6(2-4-7)5-8(11)9(12)13/h1-4,10H,5H2,(H,12,13) |
|---|
| Synonyms | | Value | Source |
|---|
| (4-Hydroxyphenyl)pyruvic acid | ChEBI | | (p-Hydroxyphenyl)pyruvic acid | ChEBI | | 3-(4-HYDROXY-phenyl)pyruvIC ACID | ChEBI | | 3-(4-Hydroxyphenyl)pyruvic acid | ChEBI | | 3-(p-Hydroxyphenyl)-2-oxopropanoic acid | ChEBI | | 4-Hydroxy alpha-oxobenzenepropanoic acid | ChEBI | | p-Hydroxyphenylpyruvic acid | ChEBI | | 4-Hydroxyphenylpyruvate | Kegg | | (4-Hydroxyphenyl)pyruvate | Generator | | (p-Hydroxyphenyl)pyruvate | Generator | | 3-(4-HYDROXY-phenyl)pyruvate | Generator | | 3-(4-Hydroxyphenyl)pyruvate | Generator | | 3-(p-Hydroxyphenyl)-2-oxopropanoate | Generator | | 4-Hydroxy a-oxobenzenepropanoate | Generator | | 4-Hydroxy a-oxobenzenepropanoic acid | Generator | | 4-Hydroxy alpha-oxobenzenepropanoate | Generator | | 4-Hydroxy α-oxobenzenepropanoate | Generator | | 4-Hydroxy α-oxobenzenepropanoic acid | Generator | | p-Hydroxyphenylpyruvate | Generator | | (p-Hydroxyphenyl)-pyruvate | HMDB | | (p-Hydroxyphenyl)-pyruvic acid | HMDB | | 3-(4-Hydroxyphenyl)-2-oxo-propanoate | HMDB | | 3-(4-Hydroxyphenyl)-2-oxo-propanoic acid | HMDB | | 3-(4-Hydroxyphenyl)-2-oxopropionate | HMDB | | 3-(4-Hydroxyphenyl)-2-oxopropionic acid | HMDB | | 3-(p-Hydroxyphenyl)-2-oxopropionate | HMDB | | 3-(p-Hydroxyphenyl)-2-oxopropionic acid | HMDB | | 3-(p-Hydroxyphenyl)pyruvate | HMDB | | 3-(p-Hydroxyphenyl)pyruvic acid | HMDB | | 4-Hydroxy-a-oxobenzenepropanoate | HMDB | | 4-Hydroxy-a-oxobenzenepropanoic acid | HMDB | | 4-Hydroxy-alpha-oxobenzenepropanoate | HMDB | | 4-Hydroxy-alpha-oxobenzenepropanoic acid | HMDB | | 4HPPA | HMDB | | HPPA | HMDB | | Hydroxyphenylpyruvate | HMDB | | Hydroxyphenylpyruvic acid | HMDB | | p-Hydroxyphenylpyruvic | HMDB | | Testacid | HMDB | | 4-Hydroxyphenylpyruvic acid, ion | HMDB | | 4-Hydroxyphenylpyruvic acid, sodium salt | HMDB | | Para-hydroxyphenylpyruvic acid | HMDB |
|
|---|
| Chemical Formula | C9H8O4 |
|---|
| Average Molecular Weight | 180.1574 |
|---|
| Monoisotopic Molecular Weight | 180.042258744 |
|---|
| IUPAC Name | 3-(4-hydroxyphenyl)-2-oxopropanoic acid |
|---|
| Traditional Name | 4-hydroxyphenylpyruvic acid |
|---|
| CAS Registry Number | 156-39-8 |
|---|
| SMILES | OC(=O)C(=O)CC1=CC=C(O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C9H8O4/c10-7-3-1-6(2-4-7)5-8(11)9(12)13/h1-4,10H,5H2,(H,12,13) |
|---|
| InChI Key | KKADPXVIOXHVKN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as phenylpyruvic acid derivatives. Phenylpyruvic acid derivatives are compounds containing a phenylpyruvic acid moiety, which consists of a phenyl group substituted at the second position by an pyruvic acid. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Phenylpyruvic acid derivatives |
|---|
| Direct Parent | Phenylpyruvic acid derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenylpyruvate
- 3-phenylpropanoic-acid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Alpha-keto acid
- Keto acid
- Alpha-hydroxy ketone
- Ketone
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Hydrocarbon derivative
- Organooxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | Not Available |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | 219 - 220 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 3.52 minutes | 32390414 | | Predicted by Siyang on May 30, 2022 | 9.9809 minutes | 33406817 | | Predicted by Siyang using ReTip algorithm on June 8, 2022 | 2.56 minutes | 32390414 | | AjsUoB = Accucore 150 Amide HILIC with 10mM Ammonium Formate, 0.1% Formic Acid | 42.1 seconds | 40023050 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 1275.0 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 315.7 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 111.3 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 182.9 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 93.2 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 368.1 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 347.5 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 125.8 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 784.3 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 317.0 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 1012.2 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 236.9 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 293.6 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 478.0 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 244.5 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 154.0 seconds | 40023050 |
Predicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 4-Hydroxyphenylpyruvic acid,1TMS,isomer #1 | C[Si](C)(C)OC(=O)C(=O)CC1=CC=C(O)C=C1 | 1824.8 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,1TMS,isomer #2 | C[Si](C)(C)OC1=CC=C(CC(=O)C(=O)O)C=C1 | 1839.9 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,1TMS,isomer #3 | C[Si](C)(C)OC(=CC1=CC=C(O)C=C1)C(=O)O | 2025.2 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,2TMS,isomer #1 | C[Si](C)(C)OC(=O)C(=O)CC1=CC=C(O[Si](C)(C)C)C=C1 | 1882.9 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,2TMS,isomer #2 | C[Si](C)(C)OC(=O)C(=CC1=CC=C(O)C=C1)O[Si](C)(C)C | 2053.6 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,2TMS,isomer #3 | C[Si](C)(C)OC(=CC1=CC=C(O[Si](C)(C)C)C=C1)C(=O)O | 2088.7 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,3TMS,isomer #1 | C[Si](C)(C)OC(=O)C(=CC1=CC=C(O[Si](C)(C)C)C=C1)O[Si](C)(C)C | 2071.8 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,3TMS,isomer #1 | C[Si](C)(C)OC(=O)C(=CC1=CC=C(O[Si](C)(C)C)C=C1)O[Si](C)(C)C | 2014.1 | Standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,3TMS,isomer #1 | C[Si](C)(C)OC(=O)C(=CC1=CC=C(O[Si](C)(C)C)C=C1)O[Si](C)(C)C | 2015.1 | Standard polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C(=O)CC1=CC=C(O)C=C1 | 2094.5 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC1=CC=C(CC(=O)C(=O)O)C=C1 | 2121.6 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,1TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=CC1=CC=C(O)C=C1)C(=O)O | 2278.9 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C(=O)CC1=CC=C(O[Si](C)(C)C(C)(C)C)C=C1 | 2382.7 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)C(=CC1=CC=C(O)C=C1)O[Si](C)(C)C(C)(C)C | 2529.5 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=CC1=CC=C(O[Si](C)(C)C(C)(C)C)C=C1)C(=O)O | 2596.6 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C(=CC1=CC=C(O[Si](C)(C)C(C)(C)C)C=C1)O[Si](C)(C)C(C)(C)C | 2764.6 | Semi standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C(=CC1=CC=C(O[Si](C)(C)C(C)(C)C)C=C1)O[Si](C)(C)C(C)(C)C | 2665.0 | Standard non polar | 33892256 | | 4-Hydroxyphenylpyruvic acid,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)C(=CC1=CC=C(O[Si](C)(C)C(C)(C)C)C=C1)O[Si](C)(C)C(C)(C)C | 2386.9 | Standard polar | 33892256 |
|
|---|
| Spectra |
|---|
| |
|---|
| Biological Properties |
|---|
| Cellular Locations | |
|---|
| Biospecimen Locations | |
|---|
| Tissue Locations | |
|---|
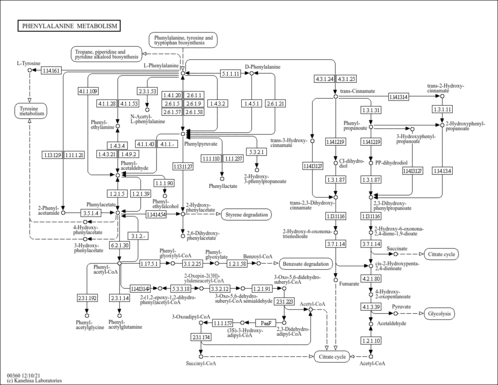
| Pathways | |
|---|
| Normal Concentrations |
|---|
| |
| Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.37 +/- 0.23 uM | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 10.669 +/- 16.944 umol/mmol creatinine | Infant (0-1 year old) | Both | Normal | | details | | Urine | Detected and Quantified | 150.31(60.04-507.20) umol/mmol creatinine | Newborn (0-30 days old) | Both | Normal | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 9.413 +/- 18.199 umol/mmol creatinine | Infant (0-1 year old) | Both | Normal | | details | | Urine | Detected and Quantified | 20.0820 +/- 49.577 umol/mmol creatinine | Infant (0-1 year old) | Both | Normal | | details | | Urine | Detected and Quantified | 1.65 (0.15-8.74) umol/mmol creatinine | Adult (>18 years old) | Male | Normal | | details | | Urine | Detected and Quantified | 4.9 umol/mmol creatinine | Infant (0-1 year old) | Both | Normal | | details | | Urine | Detected and Quantified | 1.8 (1.2-4.3) umol/mmol creatinine | Adolescent (13-18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 1.5 (0.6-3.4) umol/mmol creatinine | Children (1-13 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 1.8 (0.1-4.5) umol/mmol creatinine | Children (1-13 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 4.6 (0.1-21.3) umol/mmol creatinine | Newborn (0-30 days old) | Both | Normal | | details | | Urine | Detected and Quantified | 0.66 (0.23-2.50) umol/mmol creatinine | Adult (>18 years old) | Female | Normal | | details |
|
|---|
| Abnormal Concentrations |
|---|
| |
| Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Colorectal cancer | | details | | Saliva | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Male | Attachment loss | | details | | Saliva | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Male | Periodontal Probing Depth | | details | | Urine | Detected and Quantified | 24.00 +/- 30.5 umol/mmol creatinine | Newborn (0-30 days old) | Both | Phenylketonuria | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Cardiosvacular risk | | details | | Urine | Detected and Quantified | 0 - 3.4 umol/mmol creatinine | Newborn (0-30 days old) | Both | Hawkinsinuria | | details | | Urine | Detected and Quantified | 102.8 -537.4 umol/mmol creatinine | Newborn (0-30 days old) | Both | Hawkinsinuria | | details | | Urine | Detected and Quantified | 2.447 +/- 20.710 umol/mmol creatinine | Children (1-13 years old) | Both | Phenylketonuria | | details | | Urine | Detected and Quantified | 33.261 +/- 20.0820 umol/mmol creatinine | Children (1-13 years old) | Both | Phenylketonuria | | details | | Urine | Detected and Quantified | 33.261 +/- 26.985 umol/mmol creatinine | Children (1-13 years old) | Both | Phenylketonuria | | details | | Urine | Detected and Quantified | 12 umol/mmol creatinine | Newborn (0-30 days old) | Female | Deafness, Onychodystrophy, Osteodystrophy, Mental Retardation, and Seizures Syndrome | | details |
|
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | | Colorectal cancer |
|---|
- Goedert JJ, Sampson JN, Moore SC, Xiao Q, Xiong X, Hayes RB, Ahn J, Shi J, Sinha R: Fecal metabolomics: assay performance and association with colorectal cancer. Carcinogenesis. 2014 Sep;35(9):2089-96. doi: 10.1093/carcin/bgu131. Epub 2014 Jul 18. [PubMed:25037050 ]
| | Attachment loss |
|---|
- Liebsch C, Pitchika V, Pink C, Samietz S, Kastenmuller G, Artati A, Suhre K, Adamski J, Nauck M, Volzke H, Friedrich N, Kocher T, Holtfreter B, Pietzner M: The Saliva Metabolome in Association to Oral Health Status. J Dent Res. 2019 Jun;98(6):642-651. doi: 10.1177/0022034519842853. Epub 2019 Apr 26. [PubMed:31026179 ]
| | Periodontal Probing Depth |
|---|
- Liebsch C, Pitchika V, Pink C, Samietz S, Kastenmuller G, Artati A, Suhre K, Adamski J, Nauck M, Volzke H, Friedrich N, Kocher T, Holtfreter B, Pietzner M: The Saliva Metabolome in Association to Oral Health Status. J Dent Res. 2019 Jun;98(6):642-651. doi: 10.1177/0022034519842853. Epub 2019 Apr 26. [PubMed:31026179 ]
| | Phenylketonuria |
|---|
- Monch E, Kneer J, Jakobs C, Arnold M, Diehl H, Batzler U: Examination of urine metabolites in the newborn period and during protein loading tests at 6 months of age--Part 1. Eur J Pediatr. 1990;149 Suppl 1:S17-24. [PubMed:2091926 ]
- Rampini S, Vollmin JA, Bosshard HR, Muller M, Curtius HC: Aromatic acids in urine of healthy infants, persistent hyperphenylalaninemia, and phenylketonuria, before and after phenylalanine load. Pediatr Res. 1974 Jul;8(7):704-9. [PubMed:4837567 ]
| | Hawkinsinuria |
|---|
- Thodi G, Schulpis KH, Dotsikas Y, Pavlides C, Molou E, Chatzidaki M, Triantafylli O, Loukas YL: Hawkinsinuria in two unrelated Greek newborns: identification of a novel variant, biochemical findings and treatment. J Pediatr Endocrinol Metab. 2016 Jan;29(1):15-20. doi: 10.1515/jpem-2015-0132. [PubMed:26226126 ]
- G.Frauendienst-Egger, Friedrich K. Trefz (2017). MetaGene: Metabolic & Genetic Information Center (MIC: http://www.metagene.de). METAGENE consortium.
| | Deafness, Onychodystrophy, Osteodystrophy, Mental Retardation, and Seizures Syndrome |
|---|
- James AW, Miranda SG, Culver K, Hall BD, Golabi M: DOOR syndrome: clinical report, literature review and discussion of natural history. Am J Med Genet A. 2007 Dec 1;143A(23):2821-31. doi: 10.1002/ajmg.a.32054. [PubMed:17994565 ]
|
|
|---|
| Associated OMIM IDs | - 114500 (Colorectal cancer)
- 261600 (Phenylketonuria)
- 140350 (Hawkinsinuria)
- 220500 (Deafness, Onychodystrophy, Osteodystrophy, Mental Retardation, and Seizures Syndrome)
|
|---|
| External Links |
|---|
| DrugBank ID | DB07718 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | FDB030506 |
|---|
| KNApSAcK ID | C00007512 |
|---|
| Chemspider ID | 954 |
|---|
| KEGG Compound ID | C01179 |
|---|
| BioCyc ID | P-HYDROXY-PHENYLPYRUVATE |
|---|
| BiGG ID | 37006 |
|---|
| Wikipedia Link | 4-Hydroxyphenylpyruvic_acid |
|---|
| METLIN ID | 5675 |
|---|
| PubChem Compound | 979 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 15999 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | 34HPP |
|---|
| MarkerDB ID | MDB00000229 |
|---|
| Good Scents ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Billek, Gerhard. p-Hydroxyphenylpyruvic acid. Organic Syntheses (1963), 43 49-54. |
|---|
| Material Safety Data Sheet (MSDS) | Download (PDF) |
|---|
| General References | - Tomoeda K, Awata H, Matsuura T, Matsuda I, Ploechl E, Milovac T, Boneh A, Scott CR, Danks DM, Endo F: Mutations in the 4-hydroxyphenylpyruvic acid dioxygenase gene are responsible for tyrosinemia type III and hawkinsinuria. Mol Genet Metab. 2000 Nov;71(3):506-10. [PubMed:11073718 ]
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [PubMed:19212411 ]
- Guneral F, Bachmann C: Age-related reference values for urinary organic acids in a healthy Turkish pediatric population. Clin Chem. 1994 Jun;40(6):862-6. [PubMed:8087979 ]
- Endo F, Katoh H, Yamamoto S, Matsuda I: A murine model for type III tyrosinemia: lack of immunologically detectable 4-hydroxyphenylpyruvic acid dioxygenase enzyme protein in a novel mouse strain with hypertyrosinemia. Am J Hum Genet. 1991 Apr;48(4):704-9. [PubMed:2014797 ]
- Shoda J, Tanaka N, Osuga T, Matsuura K, Miyazaki H: Altered bile acid metabolism in liver disease: concurrent occurrence of C-1 and C-6 hydroxylated bile acid metabolites and their preferential excretion into urine. J Lipid Res. 1990 Feb;31(2):249-59. [PubMed:2324645 ]
- Wolff JA, Barshop B, Nyhan WL, Leslie J, Seegmiller JE, Gruber H, Garst M, Winter S, Michals K, Matalon R: Effects of ascorbic acid in alkaptonuria: alterations in benzoquinone acetic acid and an ontogenic effect in infancy. Pediatr Res. 1989 Aug;26(2):140-4. [PubMed:2771520 ]
- Deutsch JC: Determination of p-hydroxyphenylpyruvate, p-hydroxyphenyllactate and tyrosine in normal human plasma by gas chromatography-mass spectrometry isotope-dilution assay. J Chromatogr B Biomed Sci Appl. 1997 Mar 7;690(1-2):1-6. [PubMed:9106023 ]
- Muskiet FA, Fremouw-Ottevangers DC, Nagel GT, Wolthers BG, de Vries JA: Determination of 3-methoxy-4-hydroxyphenylpyruvic acid, 3,4-dihydroxyphenylethylene glycol, and 3,4-dihydroxyphenylmandelic acid in urine by mass fragmentography, with use of deuterium-labeled internal standards. Clin Chem. 1978 Nov;24(11):2001-8. [PubMed:709835 ]
- Elshenawy S, Pinney SE, Stuart T, Doulias PT, Zura G, Parry S, Elovitz MA, Bennett MJ, Bansal A, Strauss JF 3rd, Ischiropoulos H, Simmons RA: The Metabolomic Signature of the Placenta in Spontaneous Preterm Birth. Int J Mol Sci. 2020 Feb 4;21(3). pii: ijms21031043. doi: 10.3390/ijms21031043. [PubMed:32033212 ]
|
|---|