| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2006-05-22 14:17:41 UTC |
|---|
| Update Date | 2022-03-07 02:49:13 UTC |
|---|
| HMDB ID | HMDB0002197 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 7a,12a-Dihydroxy-cholestene-3-one |
|---|
| Description | 7a,12a-Dihydroxy-cholestene-3-one belongs to the class of organic compounds known as cholesterols and derivatives. Cholesterols and derivatives are compounds containing a 3-hydroxylated cholestane core. Based on a literature review very few articles have been published on 7a,12a-Dihydroxy-cholestene-3-one. |
|---|
| Structure | [H][C@@]12CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)[C@@H](O)C[C@@]1([H])[C@@]2([H])[C@H](O)CC2CC(=O)C=C[C@]12C InChI=1S/C27H44O3/c1-16(2)7-6-8-17(3)20-9-10-21-25-22(15-24(30)27(20,21)5)26(4)12-11-19(28)13-18(26)14-23(25)29/h11-12,16-18,20-25,29-30H,6-10,13-15H2,1-5H3/t17-,18?,20-,21+,22+,23-,24+,25+,26+,27-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 7-a,12-a-Dihydroxy-4-cholesten-3-one | HMDB | | 7-a,12-a-Dihydroxycholest-4-en-3-one | HMDB | | 7-a,12-alpha-Dihydroxy-4-cholesten-3-one | HMDB | | 7-a,12-alpha-Dihydroxycholest-4-en-3-one | HMDB | | 7-alpha,12-alpha-Dihydroxy-4-cholesten-3-one | HMDB | | 7-alpha,12-alpha-Dihydroxycholest-4-en-3-one | HMDB | | 7alpha,12alpha-Dihydroxy-cholestene-3-one | HMDB |
|
|---|
| Chemical Formula | C27H44O3 |
|---|
| Average Molecular Weight | 416.6365 |
|---|
| Monoisotopic Molecular Weight | 416.329045274 |
|---|
| IUPAC Name | (1S,2R,9R,10R,11S,14R,15R,16S)-9,16-dihydroxy-2,15-dimethyl-14-[(2R)-6-methylheptan-2-yl]tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-3-en-5-one |
|---|
| Traditional Name | (1S,2R,9R,10R,11S,14R,15R,16S)-9,16-dihydroxy-2,15-dimethyl-14-[(2R)-6-methylheptan-2-yl]tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-3-en-5-one |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H][C@@]12CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)[C@@H](O)C[C@@]1([H])[C@@]2([H])[C@H](O)CC2CC(=O)C=C[C@]12C |
|---|
| InChI Identifier | InChI=1S/C27H44O3/c1-16(2)7-6-8-17(3)20-9-10-21-25-22(15-24(30)27(20,21)5)26(4)12-11-19(28)13-18(26)14-23(25)29/h11-12,16-18,20-25,29-30H,6-10,13-15H2,1-5H3/t17-,18?,20-,21+,22+,23-,24+,25+,26+,27-/m1/s1 |
|---|
| InChI Key | VJGNBLOGSXHTLR-JFXJTJEYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as cholesterols and derivatives. Cholesterols and derivatives are compounds containing a 3-hydroxylated cholestane core. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Cholestane steroids |
|---|
| Direct Parent | Cholesterols and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cholesterol-skeleton
- 3-oxo-delta-1-steroid
- 3-oxosteroid
- 7-hydroxysteroid
- Oxosteroid
- 12-hydroxysteroid
- Hydroxysteroid
- Delta-1-steroid
- Cyclohexenone
- Cyclic alcohol
- Cyclic ketone
- Secondary alcohol
- Ketone
- Organic oxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organic oxide
- Organooxygen compound
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | | Show more...
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 7a,12a-Dihydroxy-cholestene-3-one,1TMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C)[C@@]21C)[C@@]1(C)C=CC(=O)CC1C[C@H]3O | 3441.3 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,1TMS,isomer #2 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O)[C@@]21C)[C@@]1(C)C=CC(=O)CC1C[C@H]3O[Si](C)(C)C | 3401.9 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,1TMS,isomer #3 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C)=CC1C[C@H]3O | 3308.5 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,2TMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C)[C@@]21C)[C@@]1(C)C=CC(=O)CC1C[C@H]3O[Si](C)(C)C | 3353.1 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,2TMS,isomer #2 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C)=CC1C[C@H]3O | 3289.9 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,2TMS,isomer #3 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C)=CC1C[C@H]3O[Si](C)(C)C | 3232.7 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,3TMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C)=CC1C[C@H]3O[Si](C)(C)C | 3242.6 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,3TMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C)=CC1C[C@H]3O[Si](C)(C)C | 3251.3 | Standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,3TMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C)=CC1C[C@H]3O[Si](C)(C)C | 3370.3 | Standard polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,1TBDMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C)[C@@]1(C)C=CC(=O)CC1C[C@H]3O | 3660.7 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,1TBDMS,isomer #2 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O)[C@@]21C)[C@@]1(C)C=CC(=O)CC1C[C@H]3O[Si](C)(C)C(C)(C)C | 3610.9 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,1TBDMS,isomer #3 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C(C)(C)C)=CC1C[C@H]3O | 3552.6 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,2TBDMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C)[C@@]1(C)C=CC(=O)CC1C[C@H]3O[Si](C)(C)C(C)(C)C | 3778.5 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,2TBDMS,isomer #2 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C(C)(C)C)=CC1C[C@H]3O | 3745.1 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,2TBDMS,isomer #3 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C(C)(C)C)=CC1C[C@H]3O[Si](C)(C)C(C)(C)C | 3678.9 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,3TBDMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C(C)(C)C)=CC1C[C@H]3O[Si](C)(C)C(C)(C)C | 3900.7 | Semi standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,3TBDMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C(C)(C)C)=CC1C[C@H]3O[Si](C)(C)C(C)(C)C | 3830.7 | Standard non polar | 33892256 | | 7a,12a-Dihydroxy-cholestene-3-one,3TBDMS,isomer #1 | CC(C)CCC[C@@H](C)[C@H]1CC[C@H]2[C@H]3[C@H](C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C)[C@@]1(C)C=CC(O[Si](C)(C)C(C)(C)C)=CC1C[C@H]3O[Si](C)(C)C(C)(C)C | 3596.9 | Standard polar | 33892256 |
| Show more...
|---|
| Spectra |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-0957100000-f9ca360b20be30aee5cd | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one GC-MS (2 TMS) - 70eV, Positive | splash10-0002-6421590000-060d19b9863c108036ea | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 10V, Positive-QTOF | splash10-00kb-0009200000-ca011752a678df923d91 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 20V, Positive-QTOF | splash10-007k-2109000000-fe2eac67f433d8e5ffd6 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 40V, Positive-QTOF | splash10-0avi-7209000000-dd9edd2da2f8a7cfa3b7 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 10V, Negative-QTOF | splash10-014i-0004900000-9a4838827d7cc0003b0c | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 20V, Negative-QTOF | splash10-014j-0009800000-d99dd5a2761313bbd19c | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 40V, Negative-QTOF | splash10-0012-1109000000-12f6e81d23b56a07105c | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 10V, Positive-QTOF | splash10-014i-0005900000-6e10002b2e4a4ff63704 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 20V, Positive-QTOF | splash10-052r-6369100000-1f4ae070beee131b40ce | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 40V, Positive-QTOF | splash10-0a6u-9530000000-59dfe5c229199b50616d | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 10V, Negative-QTOF | splash10-014i-0000900000-67ea14fb7a26b9b2d929 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 20V, Negative-QTOF | splash10-014i-0001900000-949821f2e2f9dbbe8632 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 7a,12a-Dihydroxy-cholestene-3-one 40V, Negative-QTOF | splash10-0udr-0079200000-cad453714d3d5275e24b | 2021-09-22 | Wishart Lab | View Spectrum |
| Show more...
|---|
| Biological Properties |
|---|
| Cellular Locations | - Extracellular
- Membrane
- Mitochondria
|
|---|
| Biospecimen Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
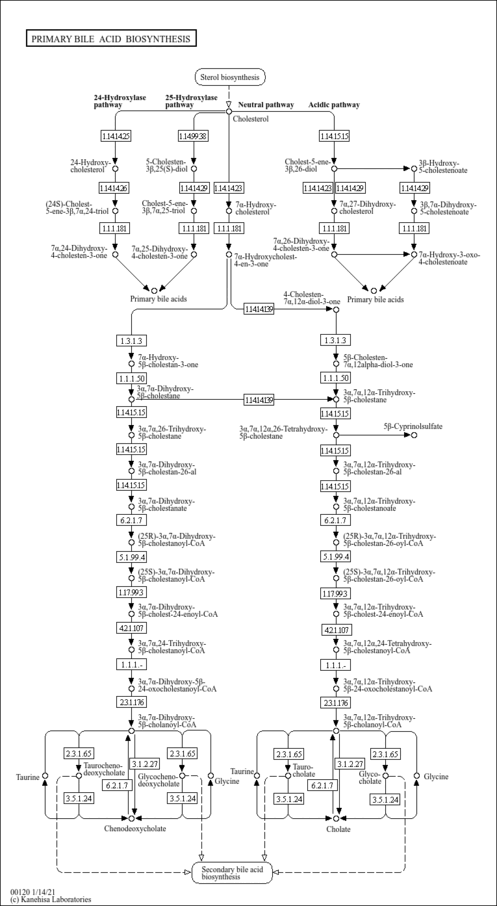
| Pathways | |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | None |
|---|
| Associated OMIM IDs | None |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | FDB022899 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 13628315 |
|---|
| KEGG Compound ID | C05457 |
|---|
| BioCyc ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| METLIN ID | 6539 |
|---|
| PubChem Compound | 21252277 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 28477 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | XOLDIOLONE |
|---|
| MarkerDB ID | Not Available |
|---|
| Good Scents ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| Material Safety Data Sheet (MSDS) | Not Available |
|---|
| General References | - Bjorkhem I, Oftebro H, Skrede S, Pedersen JI: Assay of intermediates in bile acid biosynthesis using isotope dilution--mass spectrometry: hepatic levels in the normal state and in cerebrotendinous xanthomatosis. J Lipid Res. 1981 Feb;22(2):191-200. [PubMed:7017048 ]
|
|---|