| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2008-09-12 01:33:06 UTC |
|---|
| Update Date | 2022-03-07 02:50:04 UTC |
|---|
| HMDB ID | HMDB0008204 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | PC(18:3(9Z,12Z,15Z)/18:2(9Z,12Z)) |
|---|
| Description | PC(18:3(9Z,12Z,15Z)/18:2(9Z,12Z)) is a phosphatidylcholine (PC or GPCho). It is a glycerophospholipid in which a phosphorylcholine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphocholines can have many different combinations of fatty acids of varying lengths and saturation attached at the C-1 and C-2 positions. Fatty acids containing 16, 18 and 20 carbons are the most common. PC(18:3(9Z,12Z,15Z)/18:2(9Z,12Z)), in particular, consists of one chain of a-linolenic acid at the C-1 position and one chain of linoleic acid at the C-2 position. The a-linolenic acid moiety is derived from seed oils, especially canola and soybean oil, while the linoleic acid moiety is derived from seed oils. Phospholipids, are ubiquitous in nature and are key components of the lipid bilayer of cells, as well as being involved in metabolism and signaling.While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PCs can be synthesized via three different routes. In one route, choline is activated first by phosphorylation and then by coupling to CDP prior to attachment to phosphatidic acid. PCs can also synthesized by the addition of choline to CDP-activated 1,2-diacylglycerol. A third route to PC synthesis involves the conversion of either PS or PE to PC. |
|---|
| Structure | CCCCC\C=C/C\C=C/CCCCCCCC(=O)O[C@]([H])(COC(=O)CCCCCCC\C=C/C\C=C/C\C=C/CC)COP([O-])(=O)OCC[N+](C)(C)C InChI=1S/C44H78NO8P/c1-6-8-10-12-14-16-18-20-22-24-26-28-30-32-34-36-43(46)50-40-42(41-52-54(48,49)51-39-38-45(3,4)5)53-44(47)37-35-33-31-29-27-25-23-21-19-17-15-13-11-9-7-2/h8,10,14-17,20-23,42H,6-7,9,11-13,18-19,24-41H2,1-5H3/b10-8-,16-14-,17-15-,22-20-,23-21-/t42-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 1-a-linolenoyl-2-linoleoyl-sn-glycero-3-phosphocholine | Lipid Annotator, HMDB | | GPCho(18:3/18:2) | Lipid Annotator, HMDB | | GPCho(36:5) | Lipid Annotator, HMDB | | PC(18:3/18:2) | Lipid Annotator, HMDB | | Phosphatidylcholine(36:5) | Lipid Annotator, HMDB | | Lecithin | Lipid Annotator, HMDB | | PC(36:5) | Lipid Annotator, HMDB | | Phosphatidylcholine(18:3/18:2) | Lipid Annotator, HMDB | | 1-(9Z,12Z,15Z-octadeatrienoyl)-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine | Lipid Annotator, HMDB | | PC(18:3(9Z,12Z,15Z)/18:2(9Z,12Z)) | Lipid Annotator |
|
|---|
| Chemical Formula | C44H78NO8P |
|---|
| Average Molecular Weight | 780.0658 |
|---|
| Monoisotopic Molecular Weight | 779.546504989 |
|---|
| IUPAC Name | trimethyl(2-{[(2R)-3-[(9Z,12Z,15Z)-octadeca-9,12,15-trienoyloxy]-2-[(9Z,12Z)-octadeca-9,12-dienoyloxy]propyl phosphonato]oxy}ethyl)azanium |
|---|
| Traditional Name | lecithin |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | CCCCC\C=C/C\C=C/CCCCCCCC(=O)O[C@]([H])(COC(=O)CCCCCCC\C=C/C\C=C/C\C=C/CC)COP([O-])(=O)OCC[N+](C)(C)C |
|---|
| InChI Identifier | InChI=1S/C44H78NO8P/c1-6-8-10-12-14-16-18-20-22-24-26-28-30-32-34-36-43(46)50-40-42(41-52-54(48,49)51-39-38-45(3,4)5)53-44(47)37-35-33-31-29-27-25-23-21-19-17-15-13-11-9-7-2/h8,10,14-17,20-23,42H,6-7,9,11-13,18-19,24-41H2,1-5H3/b10-8-,16-14-,17-15-,22-20-,23-21-/t42-/m1/s1 |
|---|
| InChI Key | QFDYIDGUKXRPKH-VSLGLSMXSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as phosphatidylcholines. These are glycerophosphocholines in which the two free -OH are attached to one fatty acid each through an ester linkage. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerophospholipids |
|---|
| Sub Class | Glycerophosphocholines |
|---|
| Direct Parent | Phosphatidylcholines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diacylglycero-3-phosphocholine
- Phosphocholine
- Fatty acid ester
- Dialkyl phosphate
- Dicarboxylic acid or derivatives
- Organic phosphoric acid derivative
- Phosphoric acid ester
- Alkyl phosphate
- Fatty acyl
- Quaternary ammonium salt
- Tetraalkylammonium salt
- Carboxylic acid ester
- Carboxylic acid derivative
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Amine
- Organic salt
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | |
|---|
| Disposition | Biological locationSourceExogenousFoodMilk and milk productUnfermented milk- Milk (Cow) (FooDB: FOOD00618)
- Cow milk, pasteurized, vitamin A + D added, 0% fat (FooDB: FOOD00889)
- Cow milk, pasteurized, vitamin A + D added, 1% fat (FooDB: FOOD00890)
- Cow milk, pasteurized, vitamin A + D added, 2% fat (FooDB: FOOD00891)
- Cow milk, pasteurized, vitamin D added, 3.25% fat (FooDB: FOOD00892)
|
|---|
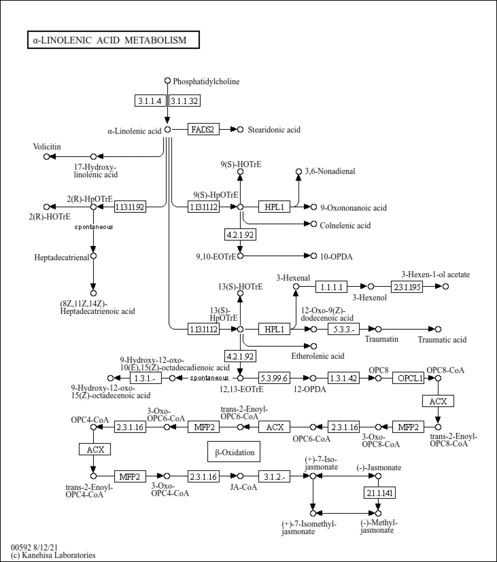
| Process | Naturally occurring processBiological processBiochemical pathwayMetabolic pathway- Alpha Linolenic Acid and Linoleic Acid Metabolism (HMDB: HMDB0008204)
- Phosphatidylcholine Biosynthesis (HMDB: HMDB0008204)
- Arachidonic Acid Metabolism (PathBank: SMP0000075)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:2(9Z,12Z)) (PathBank: SMP0014513)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/18:2(9Z,12Z)) (PathBank: SMP0015353)
- Phospholipid Biosynthesis (PathBank: SMP0000025)
- Sphingolipid Metabolism (PathBank: SMP0000034)
- Retinol Metabolism (PathBank: SMP0000074)
- Sphingolipid Metabolism (PathBank: SMP0002378)
- Choline Metabolism (PathBank: SMP0002393)
- Lysolipid Incorporation into Mitochondria (PathBank: SMP0002424)
- Lysolipid Incorporation into ER (PathBank: SMP0002425)
- Lysolipid Incorporation into Mitochondria PC(16:0/16:0) (PathBank: SMP0002492)
- Lysolipid Incorporation into ER PC(14:0/14:0) (PathBank: SMP0002513)
- Lysolipid Incorporation into ER PC(16:0/16:0) (PathBank: SMP0002514)
- Lysolipid Incorporation into ER PC(16:1(9Z)/16:1(9Z)) (PathBank: SMP0002515)
- Lysolipid Incorporation into ER PC(18:0/18:0) (PathBank: SMP0002517)
- Lysolipid Incorporation into ER PC(18:1(9Z)/18:1(9Z)) (PathBank: SMP0002518)
- Lysolipid Incorporation into ER PC(20:4(5Z,8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0002520)
- Lysolipid Incorporation into Mitochondria PC(14:0/14:0) (PathBank: SMP0002522)
- Lysolipid Incorporation into Mitochondria PC(16:1(9Z)/16:1(9Z)) (PathBank: SMP0002523)
- Lysolipid Incorporation into Mitochondria PC(18:0/18:0) (PathBank: SMP0002525)
- Lysolipid Incorporation into Mitochondria PC(18:1(9Z)/18:1(9Z)) (PathBank: SMP0002526)
- Lysolipid Incorporation into Mitochondria PC(20:4(5Z,8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0002528)
- Phosphatidylcholine Biosynthesis PC(10:0/12:0) (PathBank: SMP0002530)
- Phosphatidylcholine Biosynthesis PC(10:0/14:0) (PathBank: SMP0002531)
- Phosphatidylcholine Biosynthesis PC(10:0/15:0) (PathBank: SMP0002532)
- Phosphatidylcholine Biosynthesis PC(10:0/16:0) (PathBank: SMP0002533)
- Phosphatidylcholine Biosynthesis PC(12:0/12:0) (PathBank: SMP0002534)
- Phosphatidylcholine Biosynthesis PC(12:0/14:0) (PathBank: SMP0002535)
- Phosphatidylcholine Biosynthesis PC(12:0/15:0) (PathBank: SMP0002536)
- Phosphatidylcholine Biosynthesis PC(12:0/16:0) (PathBank: SMP0002538)
- Phosphatidylcholine Biosynthesis PC(14:0/15:0) (PathBank: SMP0002539)
- Phosphatidylcholine Biosynthesis PC(10:0/14:1(9Z)) (PathBank: SMP0002540)
- Phosphatidylcholine Biosynthesis PC(10:0/14:1(11Z)) (PathBank: SMP0002541)
- Phosphatidylcholine Biosynthesis PC(12:0/14:1(9Z)) (PathBank: SMP0002542)
- Phosphatidylcholine Biosynthesis PC(12:0/14:1(11Z)) (PathBank: SMP0002543)
- Phosphatidylcholine Biosynthesis PC(14:0/14:1(9Z)) (PathBank: SMP0002544)
- Phosphatidylcholine Biosynthesis PC(14:0/14:1(11Z)) (PathBank: SMP0002545)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/15:0) (PathBank: SMP0002546)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/15:0) (PathBank: SMP0002547)
- Phosphatidylcholine Biosynthesis PC(15:0/15:0) (PathBank: SMP0002548)
- Phosphatidylcholine Biosynthesis PC(10:0/20:1(13Z)) (PathBank: SMP0002549)
- Phosphatidylcholine Biosynthesis PC(10:0/20:1(11Z)) (PathBank: SMP0002550)
- Phosphatidylcholine Biosynthesis PC(10:0/15:1(9Z)) (PathBank: SMP0002551)
- Phosphatidylcholine Biosynthesis PC(10:0/15:1(11Z)) (PathBank: SMP0002552)
- Phosphatidylcholine Biosynthesis PC(10:0/16:1(9Z)) (PathBank: SMP0002553)
- Phosphatidylcholine Biosynthesis PC(10:0/16:1(11Z)) (PathBank: SMP0002554)
- Phosphatidylcholine Biosynthesis PC(12:0/18:1(9Z)) (PathBank: SMP0002555)
- Phosphatidylcholine Biosynthesis PC(12:0/18:1(11Z)) (PathBank: SMP0002556)
- Phosphatidylcholine Biosynthesis PC(12:0/15:1(9Z)) (PathBank: SMP0002557)
- Phosphatidylcholine Biosynthesis PC(12:0/15:1(11Z)) (PathBank: SMP0002558)
- Phosphatidylcholine Biosynthesis PC(14:0/16:1(9Z)) (PathBank: SMP0002559)
- Phosphatidylcholine Biosynthesis PC(14:0/16:1(11Z)) (PathBank: SMP0002560)
- Phosphatidylcholine Biosynthesis PC(14:0/15:1(9Z)) (PathBank: SMP0002561)
- Phosphatidylcholine Biosynthesis PC(14:0/15:1(11Z)) (PathBank: SMP0002562)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/16:0) (PathBank: SMP0002563)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/16:0) (PathBank: SMP0002564)
- Phosphatidylcholine Biosynthesis PC(15:0/15:1(9Z)) (PathBank: SMP0002565)
- Phosphatidylcholine Biosynthesis PC(15:0/15:1(11Z)) (PathBank: SMP0002566)
- Phosphatidylcholine Biosynthesis PC(12:0/16:1(9Z)) (PathBank: SMP0002567)
- Phosphatidylcholine Biosynthesis PC(12:0/16:1(11Z)) (PathBank: SMP0002568)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/14:1(9Z)) (PathBank: SMP0002569)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/14:1(11Z)) (PathBank: SMP0002570)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/14:1(9Z)) (PathBank: SMP0002571)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/14:1(11Z)) (PathBank: SMP0002572)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/15:1(9Z)) (PathBank: SMP0002573)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/15:1(11Z)) (PathBank: SMP0002574)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/15:1(9Z)) (PathBank: SMP0002575)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/15:1(11Z)) (PathBank: SMP0002576)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/16:1(9Z)) (PathBank: SMP0002577)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/16:1(11Z)) (PathBank: SMP0002578)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/16:1(9Z)) (PathBank: SMP0002579)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/16:1(11Z)) (PathBank: SMP0002580)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/15:1(9Z)) (PathBank: SMP0002581)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/15:1(11Z)) (PathBank: SMP0002582)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/15:1(9Z)) (PathBank: SMP0002583)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/15:1(11Z)) (PathBank: SMP0002584)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/16:1(9Z)) (PathBank: SMP0002585)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/16:1(11Z)) (PathBank: SMP0002586)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/16:1(9Z)) (PathBank: SMP0002587)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/16:1(11Z)) (PathBank: SMP0002588)
- Phosphatidylcholine Biosynthesis PC(14:0/16:0) (PathBank: SMP0002589)
- Phosphatidylcholine Biosynthesis PC(15:0/16:1(9Z)) (PathBank: SMP0002590)
- Phosphatidylcholine Biosynthesis PC(15:0/16:1(11Z)) (PathBank: SMP0002591)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/16:0) (PathBank: SMP0002592)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/16:0) (PathBank: SMP0002593)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/18:1(9Z)) (PathBank: SMP0002594)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/18:1(11Z)) (PathBank: SMP0002595)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/18:1(9Z)) (PathBank: SMP0002596)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/18:1(11Z)) (PathBank: SMP0002597)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/16:1(9Z)) (PathBank: SMP0002598)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/16:1(11Z)) (PathBank: SMP0002599)
- Phosphatidylcholine Biosynthesis PC(16:1(11Z)/16:1(9Z)) (PathBank: SMP0002600)
- Phosphatidylcholine Biosynthesis PC(10:0/18:1(9Z)) (PathBank: SMP0002602)
- Phosphatidylcholine Biosynthesis PC(10:0/18:1(11Z)) (PathBank: SMP0002603)
- Phosphatidylcholine Biosynthesis PC(16:0/16:1(9Z)) (PathBank: SMP0002604)
- Phosphatidylcholine Biosynthesis PC(16:0/16:1(11Z)) (PathBank: SMP0002605)
- Phosphatidylcholine Biosynthesis PC(10:0/22:1(9Z)) (PathBank: SMP0002606)
- Phosphatidylcholine Biosynthesis PC(10:0/22:1(11Z)) (PathBank: SMP0002607)
- Phosphatidylcholine Biosynthesis PC(10:0/18:0) (PathBank: SMP0002608)
- Phosphatidylcholine Biosynthesis PC(12:0/20:1(13Z)) (PathBank: SMP0002609)
- Phosphatidylcholine Biosynthesis PC(12:0/20:1(11Z)) (PathBank: SMP0002610)
- Phosphatidylcholine Biosynthesis PC(14:0/18:1(9Z)) (PathBank: SMP0002611)
- Phosphatidylcholine Biosynthesis PC(14:0/18:1(11Z)) (PathBank: SMP0002612)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/18:0) (PathBank: SMP0002613)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/18:0) (PathBank: SMP0002614)
- Phosphatidylcholine Biosynthesis PC(16:0/16:0) (PathBank: SMP0002615)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/18:1(9Z)) (PathBank: SMP0002616)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/18:1(11Z)) (PathBank: SMP0002617)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/18:1(9Z)) (PathBank: SMP0002618)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/18:1(11Z)) (PathBank: SMP0002619)
- Phosphatidylcholine Biosynthesis PC(10:0/23:1(9Z)) (PathBank: SMP0002620)
- Phosphatidylcholine Biosynthesis PC(10:0/23:1(11Z)) (PathBank: SMP0002621)
- Phosphatidylcholine Biosynthesis PC(12:0/18:0) (PathBank: SMP0002622)
- Phosphatidylcholine Biosynthesis PC(15:0/18:1(9Z)) (PathBank: SMP0002623)
- Phosphatidylcholine Biosynthesis PC(15:0/18:1(11Z)) (PathBank: SMP0002624)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/18:0) (PathBank: SMP0002625)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/18:0) (PathBank: SMP0002626)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:1(13Z)) (PathBank: SMP0002627)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:1(11Z)) (PathBank: SMP0002628)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/20:1(13Z)) (PathBank: SMP0002629)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/20:1(11Z)) (PathBank: SMP0002630)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/18:1(9Z)) (PathBank: SMP0002631)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/18:1(11Z)) (PathBank: SMP0002632)
- Phosphatidylcholine Biosynthesis PC(16:1(11Z)/18:1(9Z)) (PathBank: SMP0002633)
- Phosphatidylcholine Biosynthesis PC(16:1(11Z)/18:1(11Z)) (PathBank: SMP0002634)
- Phosphatidylcholine Biosynthesis PC(10:0/24:1(9Z)) (PathBank: SMP0002635)
- Phosphatidylcholine Biosynthesis PC(10:0/24:1(11Z)) (PathBank: SMP0002636)
- Phosphatidylcholine Biosynthesis PC(12:0/22:1(9Z)) (PathBank: SMP0002637)
- Phosphatidylcholine Biosynthesis PC(12:0/22:1(11Z)) (PathBank: SMP0002638)
- Phosphatidylcholine Biosynthesis PC(14:0/20:1(13Z)) (PathBank: SMP0002639)
- Phosphatidylcholine Biosynthesis PC(14:0/20:1(11Z)) (PathBank: SMP0002640)
- Phosphatidylcholine Biosynthesis PC(14:0/18:0) (PathBank: SMP0002641)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:0) (PathBank: SMP0002642)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/20:0) (PathBank: SMP0002643)
- Phosphatidylcholine Biosynthesis PC(16:0/18:1(9Z)) (PathBank: SMP0002644)
- Phosphatidylcholine Biosynthesis PC(16:0/18:1(11Z)) (PathBank: SMP0002645)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/18:0) (PathBank: SMP0002646)
- Phosphatidylcholine Biosynthesis PC(16:1(11Z)/18:0) (PathBank: SMP0002647)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/20:1(13Z)) (PathBank: SMP0002648)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/20:1(11Z)) (PathBank: SMP0002649)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/20:1(13Z)) (PathBank: SMP0002650)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/20:1(11Z)) (PathBank: SMP0002651)
- Phosphatidylcholine Biosynthesis PC(10:0/25:1(9Z)) (PathBank: SMP0002652)
- Phosphatidylcholine Biosynthesis PC(10:0/25:1(11Z)) (PathBank: SMP0002653)
- Phosphatidylcholine Biosynthesis PC(10:0/20:0) (PathBank: SMP0002654)
- Phosphatidylcholine Biosynthesis PC(12:0/23:1(9Z)) (PathBank: SMP0002655)
- Phosphatidylcholine Biosynthesis PC(12:0/23:1(11Z)) (PathBank: SMP0002656)
- Phosphatidylcholine Biosynthesis PC(15:0/20:1(13Z)) (PathBank: SMP0002657)
- Phosphatidylcholine Biosynthesis PC(15:0/20:1(11Z)) (PathBank: SMP0002658)
- Phosphatidylcholine Biosynthesis PC(15:0/16:0) (PathBank: SMP0002659)
- Phosphatidylcholine Biosynthesis PC(15:1(9Z)/20:0) (PathBank: SMP0002660)
- Phosphatidylcholine Biosynthesis PC(15:1(11Z)/20:0) (PathBank: SMP0002661)
- Phosphatidylcholine Biosynthesis PC(16:0/18:0) (PathBank: SMP0002662)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:1(9Z)) (PathBank: SMP0002663)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:1(11Z)) (PathBank: SMP0002664)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/22:1(9Z)) (PathBank: SMP0002665)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/22:1(11Z)) (PathBank: SMP0002666)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:1(13Z)) (PathBank: SMP0002667)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:1(11Z)) (PathBank: SMP0002668)
- Phosphatidylcholine Biosynthesis PC(16:1(11Z)/20:1(13Z)) (PathBank: SMP0002669)
- Phosphatidylcholine Biosynthesis PC(16:1(11Z)/20:1(11Z)) (PathBank: SMP0002670)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/18:1(9Z)) (PathBank: SMP0002671)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/18:1(11Z)) (PathBank: SMP0002672)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/18:1(9Z)) (PathBank: SMP0002673)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/18:1(11Z)) (PathBank: SMP0002674)
- Phosphatidylcholine Biosynthesis PC(18:0/18:1(9Z)) (PathBank: SMP0002675)
- Phosphatidylcholine Biosynthesis PC(18:0/18:1(11Z)) (PathBank: SMP0002676)
- Phosphatidylcholine Biosynthesis PC(10:0/26:1(9Z)) (PathBank: SMP0002677)
- Phosphatidylcholine Biosynthesis PC(10:0/26:1(11Z)) (PathBank: SMP0002678)
- Phosphatidylcholine Biosynthesis PC(12:0/24:1(9Z)) (PathBank: SMP0002679)
- Phosphatidylcholine Biosynthesis PC(12:0/24:1(11Z)) (PathBank: SMP0002680)
- Phosphatidylcholine Biosynthesis PC(14:0/22:1(9Z)) (PathBank: SMP0002682)
- Phosphatidylcholine Biosynthesis PC(14:0/22:1(11Z)) (PathBank: SMP0002683)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:0) (PathBank: SMP0002684)
- Phosphatidylcholine Biosynthesis PC(14:1(11Z)/22:0) (PathBank: SMP0002685)
- Phosphatidylcholine Biosynthesis PC(16:0/20:1(13Z)) (PathBank: SMP0002686)
- Phosphatidylcholine Biosynthesis PC(16:0/20:1(11Z)) (PathBank: SMP0002687)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:0) (PathBank: SMP0002688)
- Phosphatidylcholine Biosynthesis PC(16:1(11Z)/20:0) (PathBank: SMP0002689)
- Phosphatidylcholine Biosynthesis PC(18:0/18:0) (PathBank: SMP0002690)
- Phosphatidylcholine Biosynthesis PC(14:0/14:0) (PathBank: SMP0014216)
- Phosphatidylcholine Biosynthesis PC(14:0/18:2(9Z,12Z)) (PathBank: SMP0014224)
- Phosphatidylcholine Biosynthesis PC(14:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0014225)
- Phosphatidylcholine Biosynthesis PC(14:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0014226)
- Phosphatidylcholine Biosynthesis PC(14:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014227)
- Phosphatidylcholine Biosynthesis PC(14:0/20:0) (PathBank: SMP0014228)
- Phosphatidylcholine Biosynthesis PC(14:0/20:2(11Z,14Z)) (PathBank: SMP0014230)
- Phosphatidylcholine Biosynthesis PC(14:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0014231)
- Phosphatidylcholine Biosynthesis PC(14:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0014232)
- Phosphatidylcholine Biosynthesis PC(14:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014233)
- Phosphatidylcholine Biosynthesis PC(14:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014234)
- Phosphatidylcholine Biosynthesis PC(14:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014235)
- Phosphatidylcholine Biosynthesis PC(14:0/22:0) (PathBank: SMP0014236)
- Phosphatidylcholine Biosynthesis PC(14:0/22:1(13Z)) (PathBank: SMP0014237)
- Phosphatidylcholine Biosynthesis PC(14:0/22:2(13Z,16Z)) (PathBank: SMP0014238)
- Phosphatidylcholine Biosynthesis PC(14:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014239)
- Phosphatidylcholine Biosynthesis PC(14:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014240)
- Phosphatidylcholine Biosynthesis PC(14:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014241)
- Phosphatidylcholine Biosynthesis PC(14:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014242)
- Phosphatidylcholine Biosynthesis PC(14:0/24:0) (PathBank: SMP0014243)
- Phosphatidylcholine Biosynthesis PC(14:0/24:1(15Z)) (PathBank: SMP0014244)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/14:0) (PathBank: SMP0014245)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0014253)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014254)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014255)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014256)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:2(11Z,14Z)) (PathBank: SMP0014259)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014260)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014261)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014262)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014263)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014264)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:1(13Z)) (PathBank: SMP0014266)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:2(13Z,16Z)) (PathBank: SMP0014267)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014268)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014269)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014270)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014271)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/24:0) (PathBank: SMP0014272)
- Phosphatidylcholine Biosynthesis PC(14:1(9Z)/24:1(15Z)) (PathBank: SMP0014273)
- Phosphatidylcholine Biosynthesis PC(15:0/14:0) (PathBank: SMP0014274)
- Phosphatidylcholine Biosynthesis PC(15:0/14:1(9Z)) (PathBank: SMP0014275)
- Phosphatidylcholine Biosynthesis PC(15:0/18:0) (PathBank: SMP0014279)
- Phosphatidylcholine Biosynthesis PC(15:0/18:2(9Z,12Z)) (PathBank: SMP0014282)
- Phosphatidylcholine Biosynthesis PC(15:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0014283)
- Phosphatidylcholine Biosynthesis PC(15:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0014284)
- Phosphatidylcholine Biosynthesis PC(15:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014285)
- Phosphatidylcholine Biosynthesis PC(15:0/20:0) (PathBank: SMP0014286)
- Phosphatidylcholine Biosynthesis PC(15:0/20:2(11Z,14Z)) (PathBank: SMP0014288)
- Phosphatidylcholine Biosynthesis PC(15:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0014289)
- Phosphatidylcholine Biosynthesis PC(15:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0014290)
- Phosphatidylcholine Biosynthesis PC(15:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014291)
- Phosphatidylcholine Biosynthesis PC(15:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014292)
- Phosphatidylcholine Biosynthesis PC(15:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014293)
- Phosphatidylcholine Biosynthesis PC(15:0/22:0) (PathBank: SMP0014294)
- Phosphatidylcholine Biosynthesis PC(15:0/22:1(13Z)) (PathBank: SMP0014295)
- Phosphatidylcholine Biosynthesis PC(15:0/22:2(13Z,16Z)) (PathBank: SMP0014296)
- Phosphatidylcholine Biosynthesis PC(15:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014297)
- Phosphatidylcholine Biosynthesis PC(15:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014298)
- Phosphatidylcholine Biosynthesis PC(15:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014299)
- Phosphatidylcholine Biosynthesis PC(15:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014300)
- Phosphatidylcholine Biosynthesis PC(15:0/24:0) (PathBank: SMP0014301)
- Phosphatidylcholine Biosynthesis PC(15:0/24:1(15Z)) (PathBank: SMP0014302)
- Phosphatidylcholine Biosynthesis PC(16:0/14:0) (PathBank: SMP0014303)
- Phosphatidylcholine Biosynthesis PC(16:0/14:1(9Z)) (PathBank: SMP0014304)
- Phosphatidylcholine Biosynthesis PC(16:0/15:0) (PathBank: SMP0014305)
- Phosphatidylcholine Biosynthesis PC(16:0/18:2(9Z,12Z)) (PathBank: SMP0014311)
- Phosphatidylcholine Biosynthesis PC(16:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0014312)
- Phosphatidylcholine Biosynthesis PC(16:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0014313)
- Phosphatidylcholine Biosynthesis PC(16:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014314)
- Phosphatidylcholine Biosynthesis PC(16:0/20:0) (PathBank: SMP0014315)
- Phosphatidylcholine Biosynthesis PC(16:0/20:2(11Z,14Z)) (PathBank: SMP0014317)
- Phosphatidylcholine Biosynthesis PC(16:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0014318)
- Phosphatidylcholine Biosynthesis PC(16:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0014319)
- Phosphatidylcholine Biosynthesis PC(16:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014320)
- Phosphatidylcholine Biosynthesis PC(16:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014321)
- Phosphatidylcholine Biosynthesis PC(16:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014322)
- Phosphatidylcholine Biosynthesis PC(16:0/22:0) (PathBank: SMP0014323)
- Phosphatidylcholine Biosynthesis PC(16:0/22:1(13Z)) (PathBank: SMP0014324)
- Phosphatidylcholine Biosynthesis PC(16:0/22:2(13Z,16Z)) (PathBank: SMP0014325)
- Phosphatidylcholine Biosynthesis PC(16:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014326)
- Phosphatidylcholine Biosynthesis PC(16:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014327)
- Phosphatidylcholine Biosynthesis PC(16:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014328)
- Phosphatidylcholine Biosynthesis PC(16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014329)
- Phosphatidylcholine Biosynthesis PC(16:0/24:0) (PathBank: SMP0014330)
- Phosphatidylcholine Biosynthesis PC(16:0/24:1(15Z)) (PathBank: SMP0014331)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/14:0) (PathBank: SMP0014332)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/14:1(9Z)) (PathBank: SMP0014333)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/15:0) (PathBank: SMP0014334)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/16:0) (PathBank: SMP0014335)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0014340)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014341)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014342)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014343)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:2(11Z,14Z)) (PathBank: SMP0014346)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014347)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014348)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014349)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014350)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014351)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/22:0) (PathBank: SMP0014352)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/22:1(13Z)) (PathBank: SMP0014353)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/22:2(13Z,16Z)) (PathBank: SMP0014354)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014355)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014356)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014357)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014358)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/24:0) (PathBank: SMP0014359)
- Phosphatidylcholine Biosynthesis PC(16:1(9Z)/24:1(15Z)) (PathBank: SMP0014360)
- Phosphatidylcholine Biosynthesis PC(18:0/14:0) (PathBank: SMP0014361)
- Phosphatidylcholine Biosynthesis PC(18:0/14:1(9Z)) (PathBank: SMP0014362)
- Phosphatidylcholine Biosynthesis PC(18:0/15:0) (PathBank: SMP0014363)
- Phosphatidylcholine Biosynthesis PC(18:0/16:0) (PathBank: SMP0014364)
- Phosphatidylcholine Biosynthesis PC(18:0/16:1(9Z)) (PathBank: SMP0014365)
- Phosphatidylcholine Biosynthesis PC(18:0/18:2(9Z,12Z)) (PathBank: SMP0014369)
- Phosphatidylcholine Biosynthesis PC(18:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0014370)
- Phosphatidylcholine Biosynthesis PC(18:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0014371)
- Phosphatidylcholine Biosynthesis PC(18:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014372)
- Phosphatidylcholine Biosynthesis PC(18:0/20:0) (PathBank: SMP0014373)
- Phosphatidylcholine Biosynthesis PC(18:0/20:1(11Z)) (PathBank: SMP0014374)
- Phosphatidylcholine Biosynthesis PC(18:0/20:2(11Z,14Z)) (PathBank: SMP0014375)
- Phosphatidylcholine Biosynthesis PC(18:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0014376)
- Phosphatidylcholine Biosynthesis PC(18:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0014377)
- Phosphatidylcholine Biosynthesis PC(18:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014378)
- Phosphatidylcholine Biosynthesis PC(18:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014379)
- Phosphatidylcholine Biosynthesis PC(18:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014380)
- Phosphatidylcholine Biosynthesis PC(18:0/22:0) (PathBank: SMP0014381)
- Phosphatidylcholine Biosynthesis PC(18:0/22:1(13Z)) (PathBank: SMP0014382)
- Phosphatidylcholine Biosynthesis PC(18:0/22:2(13Z,16Z)) (PathBank: SMP0014383)
- Phosphatidylcholine Biosynthesis PC(18:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014384)
- Phosphatidylcholine Biosynthesis PC(18:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014385)
- Phosphatidylcholine Biosynthesis PC(18:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014386)
- Phosphatidylcholine Biosynthesis PC(18:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014387)
- Phosphatidylcholine Biosynthesis PC(18:0/24:0) (PathBank: SMP0014388)
- Phosphatidylcholine Biosynthesis PC(18:0/24:1(15Z)) (PathBank: SMP0014389)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/14:0) (PathBank: SMP0014390)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/14:1(9Z)) (PathBank: SMP0014391)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/15:0) (PathBank: SMP0014392)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/16:0) (PathBank: SMP0014393)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/16:1(9Z)) (PathBank: SMP0014394)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/18:0) (PathBank: SMP0014395)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/18:2(9Z,12Z)) (PathBank: SMP0014398)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014399)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014400)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014401)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:0) (PathBank: SMP0014402)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:1(11Z)) (PathBank: SMP0014403)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:2(11Z,14Z)) (PathBank: SMP0014404)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014405)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014406)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014407)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014408)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014409)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/22:0) (PathBank: SMP0014410)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/22:1(13Z)) (PathBank: SMP0014411)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/22:2(13Z,16Z)) (PathBank: SMP0014412)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014413)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014414)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014415)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014416)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/24:0) (PathBank: SMP0014417)
- Phosphatidylcholine Biosynthesis PC(18:1(11Z)/24:1(15Z)) (PathBank: SMP0014418)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/14:0) (PathBank: SMP0014419)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/14:1(9Z)) (PathBank: SMP0014420)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/15:0) (PathBank: SMP0014421)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/16:0) (PathBank: SMP0014422)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/16:1(9Z)) (PathBank: SMP0014423)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/18:0) (PathBank: SMP0014424)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0014427)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014428)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014429)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014430)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:0) (PathBank: SMP0014431)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:1(11Z)) (PathBank: SMP0014432)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:2(11Z,14Z)) (PathBank: SMP0014433)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014434)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014435)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014436)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014437)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014438)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/22:0) (PathBank: SMP0014439)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/22:1(13Z)) (PathBank: SMP0014440)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/22:2(13Z,16Z)) (PathBank: SMP0014441)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014442)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014443)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014444)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014445)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/24:0) (PathBank: SMP0014446)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/14:0) (PathBank: SMP0014447)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/14:1(9Z)) (PathBank: SMP0014448)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/15:0) (PathBank: SMP0014449)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/16:0) (PathBank: SMP0014450)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/16:1(9Z)) (PathBank: SMP0014451)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/18:0) (PathBank: SMP0014452)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/18:1(11Z)) (PathBank: SMP0014453)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/18:1(9Z)) (PathBank: SMP0014454)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/18:2(9Z,12Z)) (PathBank: SMP0014455)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014456)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014457)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014458)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:0) (PathBank: SMP0014459)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:1(11Z)) (PathBank: SMP0014460)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:2(11Z,14Z)) (PathBank: SMP0014461)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014462)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014463)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014464)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014465)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014466)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/22:0) (PathBank: SMP0014467)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/22:1(13Z)) (PathBank: SMP0014468)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/22:2(13Z,16Z)) (PathBank: SMP0014469)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014470)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014471)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014472)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014473)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/24:0) (PathBank: SMP0014474)
- Phosphatidylcholine Biosynthesis PC(18:2(9Z,12Z)/24:1(15Z)) (PathBank: SMP0014475)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/14:0) (PathBank: SMP0014476)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/14:1(9Z)) (PathBank: SMP0014477)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/15:0) (PathBank: SMP0014478)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/16:0) (PathBank: SMP0014479)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/16:1(9Z)) (PathBank: SMP0014480)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:0) (PathBank: SMP0014481)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:1(11Z)) (PathBank: SMP0014482)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:1(9Z)) (PathBank: SMP0014483)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:2(9Z,12Z)) (PathBank: SMP0014484)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014485)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014486)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014487)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:0) (PathBank: SMP0014488)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:1(11Z)) (PathBank: SMP0014489)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:2(11Z,14Z)) (PathBank: SMP0014490)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014491)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014492)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014493)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014494)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014495)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/22:0) (PathBank: SMP0014496)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/22:1(13Z)) (PathBank: SMP0014497)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/22:2(13Z,16Z)) (PathBank: SMP0014498)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014499)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014500)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014501)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014502)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/24:0) (PathBank: SMP0014503)
- Phosphatidylcholine Biosynthesis PC(18:3(6Z,9Z,12Z)/24:1(15Z)) (PathBank: SMP0014504)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/14:0) (PathBank: SMP0014505)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/14:1(9Z)) (PathBank: SMP0014506)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/15:0) (PathBank: SMP0014507)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/16:0) (PathBank: SMP0014508)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/16:1(9Z)) (PathBank: SMP0014509)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:0) (PathBank: SMP0014510)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:1(11Z)) (PathBank: SMP0014511)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:1(9Z)) (PathBank: SMP0014512)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014514)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014515)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014516)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:0) (PathBank: SMP0014517)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:1(11Z)) (PathBank: SMP0014518)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:2(11Z,14Z)) (PathBank: SMP0014519)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014520)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014521)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014522)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014523)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014524)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:0) (PathBank: SMP0014525)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:1(13Z)) (PathBank: SMP0014526)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:2(13Z,16Z)) (PathBank: SMP0014527)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014528)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014529)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014530)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014531)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/24:0) (PathBank: SMP0014532)
- Phosphatidylcholine Biosynthesis PC(18:3(9Z,12Z,15Z)/24:1(15Z)) (PathBank: SMP0014533)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/14:0) (PathBank: SMP0014534)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/14:1(9Z)) (PathBank: SMP0014535)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/15:0) (PathBank: SMP0014536)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/16:0) (PathBank: SMP0014537)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/16:1(9Z)) (PathBank: SMP0014538)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/18:0) (PathBank: SMP0014539)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/18:1(11Z)) (PathBank: SMP0014540)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/18:1(9Z)) (PathBank: SMP0014541)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/18:2(9Z,12Z)) (PathBank: SMP0014542)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014543)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014544)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014545)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:0) (PathBank: SMP0014546)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:1(11Z)) (PathBank: SMP0014547)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:2(11Z,14Z)) (PathBank: SMP0014548)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014549)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014550)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014551)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014552)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014553)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/22:0) (PathBank: SMP0014554)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/22:1(13Z)) (PathBank: SMP0014555)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/22:2(13Z,16Z)) (PathBank: SMP0014556)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014557)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014558)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014559)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014560)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/24:0) (PathBank: SMP0014561)
- Phosphatidylcholine Biosynthesis PC(18:4(6Z,9Z,12Z,15Z)/24:1(15Z)) (PathBank: SMP0014562)
- Phosphatidylcholine Biosynthesis PC(20:0/14:0) (PathBank: SMP0014563)
- Phosphatidylcholine Biosynthesis PC(20:0/14:1(9Z)) (PathBank: SMP0014564)
- Phosphatidylcholine Biosynthesis PC(20:0/15:0) (PathBank: SMP0014565)
- Phosphatidylcholine Biosynthesis PC(20:0/16:0) (PathBank: SMP0014566)
- Phosphatidylcholine Biosynthesis PC(20:0/16:1(9Z)) (PathBank: SMP0014567)
- Phosphatidylcholine Biosynthesis PC(20:0/18:0) (PathBank: SMP0014568)
- Phosphatidylcholine Biosynthesis PC(20:0/18:1(11Z)) (PathBank: SMP0014569)
- Phosphatidylcholine Biosynthesis PC(20:0/18:1(9Z)) (PathBank: SMP0014570)
- Phosphatidylcholine Biosynthesis PC(20:0/18:2(9Z,12Z)) (PathBank: SMP0014571)
- Phosphatidylcholine Biosynthesis PC(20:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0014572)
- Phosphatidylcholine Biosynthesis PC(20:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0014573)
- Phosphatidylcholine Biosynthesis PC(20:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014574)
- Phosphatidylcholine Biosynthesis PC(20:0/20:0) (PathBank: SMP0014575)
- Phosphatidylcholine Biosynthesis PC(20:0/20:1(11Z)) (PathBank: SMP0014576)
- Phosphatidylcholine Biosynthesis PC(20:0/20:2(11Z,14Z)) (PathBank: SMP0014577)
- Phosphatidylcholine Biosynthesis PC(20:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0014578)
- Phosphatidylcholine Biosynthesis PC(20:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0014579)
- Phosphatidylcholine Biosynthesis PC(20:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014580)
- Phosphatidylcholine Biosynthesis PC(20:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014581)
- Phosphatidylcholine Biosynthesis PC(20:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014582)
- Phosphatidylcholine Biosynthesis PC(20:0/22:0) (PathBank: SMP0014583)
- Phosphatidylcholine Biosynthesis PC(20:0/22:1(13Z)) (PathBank: SMP0014584)
- Phosphatidylcholine Biosynthesis PC(20:0/22:2(13Z,16Z)) (PathBank: SMP0014585)
- Phosphatidylcholine Biosynthesis PC(20:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014586)
- Phosphatidylcholine Biosynthesis PC(20:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014587)
- Phosphatidylcholine Biosynthesis PC(20:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014588)
- Phosphatidylcholine Biosynthesis PC(20:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014589)
- Phosphatidylcholine Biosynthesis PC(20:0/24:0) (PathBank: SMP0014590)
- Phosphatidylcholine Biosynthesis PC(20:0/24:1(15Z)) (PathBank: SMP0014591)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/14:0) (PathBank: SMP0014592)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/14:1(9Z)) (PathBank: SMP0014593)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/15:0) (PathBank: SMP0014594)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/16:0) (PathBank: SMP0014595)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/16:1(9Z)) (PathBank: SMP0014596)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/18:0) (PathBank: SMP0014597)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/18:1(11Z)) (PathBank: SMP0014598)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/18:1(9Z)) (PathBank: SMP0014599)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/18:2(9Z,12Z)) (PathBank: SMP0014600)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014601)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014602)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014603)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:0) (PathBank: SMP0014604)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:1(11Z)) (PathBank: SMP0014605)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:2(11Z,14Z)) (PathBank: SMP0014606)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014607)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014608)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014609)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014610)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014611)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/22:0) (PathBank: SMP0014612)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/22:1(13Z)) (PathBank: SMP0014613)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/22:2(13Z,16Z)) (PathBank: SMP0014614)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014615)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014616)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014617)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014618)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/24:0) (PathBank: SMP0014619)
- Phosphatidylcholine Biosynthesis PC(20:1(11Z)/24:1(15Z)) (PathBank: SMP0014620)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/14:0) (PathBank: SMP0014621)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/14:1(9Z)) (PathBank: SMP0014622)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/15:0) (PathBank: SMP0014623)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/16:0) (PathBank: SMP0014624)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/16:1(9Z)) (PathBank: SMP0014625)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/18:0) (PathBank: SMP0014626)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/18:1(11Z)) (PathBank: SMP0014627)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/18:1(9Z)) (PathBank: SMP0014628)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/18:2(9Z,12Z)) (PathBank: SMP0014629)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014630)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014631)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014632)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:0) (PathBank: SMP0014633)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:1(11Z)) (PathBank: SMP0014634)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:2(11Z,14Z)) (PathBank: SMP0014635)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014636)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014637)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014638)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014639)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014640)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/22:0) (PathBank: SMP0014641)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/22:1(13Z)) (PathBank: SMP0014642)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/22:2(13Z,16Z)) (PathBank: SMP0014643)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014644)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014645)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014646)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014647)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/24:0) (PathBank: SMP0014648)
- Phosphatidylcholine Biosynthesis PC(20:2(11Z,14Z)/24:1(15Z)) (PathBank: SMP0014649)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/14:0) (PathBank: SMP0014650)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/14:1(9Z)) (PathBank: SMP0014651)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/15:0) (PathBank: SMP0014652)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/16:0) (PathBank: SMP0014653)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/16:1(9Z)) (PathBank: SMP0014654)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/18:0) (PathBank: SMP0014655)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/18:1(11Z)) (PathBank: SMP0014656)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/18:1(9Z)) (PathBank: SMP0014657)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/18:2(9Z,12Z)) (PathBank: SMP0014658)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014659)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014660)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014661)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:0) (PathBank: SMP0014662)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:1(11Z)) (PathBank: SMP0014663)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:2(11Z,14Z)) (PathBank: SMP0014664)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014665)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014666)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014667)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014668)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014669)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/22:0) (PathBank: SMP0014670)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/22:1(13Z)) (PathBank: SMP0014671)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/22:2(13Z,16Z)) (PathBank: SMP0014672)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014673)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014674)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014675)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014676)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/24:0) (PathBank: SMP0014677)
- Phosphatidylcholine Biosynthesis PC(20:3(5Z,8Z,11Z)/24:1(15Z)) (PathBank: SMP0014678)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/14:0) (PathBank: SMP0014679)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/14:1(9Z)) (PathBank: SMP0014680)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/15:0) (PathBank: SMP0014681)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/16:0) (PathBank: SMP0014682)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/16:1(9Z)) (PathBank: SMP0014683)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/18:0) (PathBank: SMP0014684)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/18:1(11Z)) (PathBank: SMP0014685)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/18:1(9Z)) (PathBank: SMP0014686)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/18:2(9Z,12Z)) (PathBank: SMP0014687)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014688)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014689)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014690)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:0) (PathBank: SMP0014691)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:2(11Z,14Z)) (PathBank: SMP0014693)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014694)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014695)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014696)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014697)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014698)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/22:0) (PathBank: SMP0014699)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/22:1(13Z)) (PathBank: SMP0014700)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/22:2(13Z,16Z)) (PathBank: SMP0014701)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014702)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014703)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014704)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014705)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/24:0) (PathBank: SMP0014706)
- Phosphatidylcholine Biosynthesis PC(20:3(8Z,11Z,14Z)/24:1(15Z)) (PathBank: SMP0014707)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/14:0) (PathBank: SMP0014708)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/14:1(9Z)) (PathBank: SMP0014709)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/15:0) (PathBank: SMP0014710)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/16:0) (PathBank: SMP0014711)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/16:1(9Z)) (PathBank: SMP0014712)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/18:0) (PathBank: SMP0014713)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/18:1(11Z)) (PathBank: SMP0014714)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/18:1(9Z)) (PathBank: SMP0014715)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/18:2(9Z,12Z)) (PathBank: SMP0014716)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014717)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014718)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014719)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:0) (PathBank: SMP0014720)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:1(11Z)) (PathBank: SMP0014721)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:2(11Z,14Z)) (PathBank: SMP0014722)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014723)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014724)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014725)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014726)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014727)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:0) (PathBank: SMP0014728)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:1(13Z)) (PathBank: SMP0014729)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:2(13Z,16Z)) (PathBank: SMP0014730)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014731)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014732)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014733)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014734)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/24:0) (PathBank: SMP0014735)
- Phosphatidylcholine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/24:1(15Z)) (PathBank: SMP0014736)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/14:0) (PathBank: SMP0014737)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/14:1(9Z)) (PathBank: SMP0014738)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/15:0) (PathBank: SMP0014739)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/16:0) (PathBank: SMP0014740)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/16:1(9Z)) (PathBank: SMP0014741)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/18:0) (PathBank: SMP0014742)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/18:1(11Z)) (PathBank: SMP0014743)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/18:1(9Z)) (PathBank: SMP0014744)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/18:2(9Z,12Z)) (PathBank: SMP0014745)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014746)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014747)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014748)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:0) (PathBank: SMP0014749)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:1(11Z)) (PathBank: SMP0014750)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:2(11Z,14Z)) (PathBank: SMP0014751)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014752)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014753)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014754)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014755)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014756)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/22:0) (PathBank: SMP0014757)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/22:1(13Z)) (PathBank: SMP0014758)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/22:2(13Z,16Z)) (PathBank: SMP0014759)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014760)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014761)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014762)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014763)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/24:0) (PathBank: SMP0014764)
- Phosphatidylcholine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/24:1(15Z)) (PathBank: SMP0014765)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/14:0) (PathBank: SMP0014766)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/14:1(9Z)) (PathBank: SMP0014767)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/15:0) (PathBank: SMP0014768)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/16:0) (PathBank: SMP0014769)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/16:1(9Z)) (PathBank: SMP0014770)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/18:0) (PathBank: SMP0014771)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/18:1(11Z)) (PathBank: SMP0014772)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/18:1(9Z)) (PathBank: SMP0014773)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/18:2(9Z,12Z)) (PathBank: SMP0014774)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014775)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014776)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014777)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:0) (PathBank: SMP0014778)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:1(11Z)) (PathBank: SMP0014779)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:2(11Z,14Z)) (PathBank: SMP0014780)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014781)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014782)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014783)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014784)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014785)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/22:0) (PathBank: SMP0014786)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/22:1(13Z)) (PathBank: SMP0014787)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/22:2(13Z,16Z)) (PathBank: SMP0014788)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014789)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014790)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014791)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014792)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/24:0) (PathBank: SMP0014793)
- Phosphatidylcholine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/24:1(15Z)) (PathBank: SMP0014794)
- Phosphatidylcholine Biosynthesis PC(22:0/14:0) (PathBank: SMP0014795)
- Phosphatidylcholine Biosynthesis PC(22:0/14:1(9Z)) (PathBank: SMP0014796)
- Phosphatidylcholine Biosynthesis PC(22:0/15:0) (PathBank: SMP0014797)
- Phosphatidylcholine Biosynthesis PC(22:0/16:0) (PathBank: SMP0014798)
- Phosphatidylcholine Biosynthesis PC(22:0/16:1(9Z)) (PathBank: SMP0014799)
- Phosphatidylcholine Biosynthesis PC(22:0/18:0) (PathBank: SMP0014800)
- Phosphatidylcholine Biosynthesis PC(22:0/18:1(11Z)) (PathBank: SMP0014801)
- Phosphatidylcholine Biosynthesis PC(22:0/18:1(9Z)) (PathBank: SMP0014802)
- Phosphatidylcholine Biosynthesis PC(22:0/18:2(9Z,12Z)) (PathBank: SMP0014803)
- Phosphatidylcholine Biosynthesis PC(22:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0014804)
- Phosphatidylcholine Biosynthesis PC(22:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0014805)
- Phosphatidylcholine Biosynthesis PC(22:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014806)
- Phosphatidylcholine Biosynthesis PC(22:0/20:0) (PathBank: SMP0014807)
- Phosphatidylcholine Biosynthesis PC(22:0/20:1(11Z)) (PathBank: SMP0014808)
- Phosphatidylcholine Biosynthesis PC(22:0/20:2(11Z,14Z)) (PathBank: SMP0014809)
- Phosphatidylcholine Biosynthesis PC(22:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0014810)
- Phosphatidylcholine Biosynthesis PC(22:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0014811)
- Phosphatidylcholine Biosynthesis PC(22:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014812)
- Phosphatidylcholine Biosynthesis PC(22:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014813)
- Phosphatidylcholine Biosynthesis PC(22:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014814)
- Phosphatidylcholine Biosynthesis PC(22:0/22:0) (PathBank: SMP0014815)
- Phosphatidylcholine Biosynthesis PC(22:0/22:1(13Z)) (PathBank: SMP0014816)
- Phosphatidylcholine Biosynthesis PC(22:0/22:2(13Z,16Z)) (PathBank: SMP0014817)
- Phosphatidylcholine Biosynthesis PC(22:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014818)
- Phosphatidylcholine Biosynthesis PC(22:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014819)
- Phosphatidylcholine Biosynthesis PC(22:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014820)
- Phosphatidylcholine Biosynthesis PC(22:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014821)
- Phosphatidylcholine Biosynthesis PC(22:0/24:0) (PathBank: SMP0014822)
- Phosphatidylcholine Biosynthesis PC(22:0/24:1(15Z)) (PathBank: SMP0014823)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/14:0) (PathBank: SMP0014824)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/14:1(9Z)) (PathBank: SMP0014825)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/15:0) (PathBank: SMP0014826)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/16:0) (PathBank: SMP0014827)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/16:1(9Z)) (PathBank: SMP0014828)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/18:0) (PathBank: SMP0014829)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/18:1(9Z)) (PathBank: SMP0014831)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/18:2(9Z,12Z)) (PathBank: SMP0014832)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014833)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014834)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014835)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:0) (PathBank: SMP0014836)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:1(11Z)) (PathBank: SMP0014837)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:2(11Z,14Z)) (PathBank: SMP0014838)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014839)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014840)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014841)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014842)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014843)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/22:0) (PathBank: SMP0014844)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/22:1(13Z)) (PathBank: SMP0014845)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/22:2(13Z,16Z)) (PathBank: SMP0014846)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014847)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014848)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014849)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014850)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/24:0) (PathBank: SMP0014851)
- Phosphatidylcholine Biosynthesis PC(22:1(13Z)/24:1(15Z)) (PathBank: SMP0014852)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/14:0) (PathBank: SMP0014853)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/14:1(9Z)) (PathBank: SMP0014854)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/15:0) (PathBank: SMP0014855)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/16:0) (PathBank: SMP0014856)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/16:1(9Z)) (PathBank: SMP0014857)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/18:0) (PathBank: SMP0014858)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/18:1(11Z)) (PathBank: SMP0014859)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/18:1(9Z)) (PathBank: SMP0014860)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/18:2(9Z,12Z)) (PathBank: SMP0014861)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014862)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014863)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014864)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:0) (PathBank: SMP0014865)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:1(11Z)) (PathBank: SMP0014866)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:2(11Z,14Z)) (PathBank: SMP0014867)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014868)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014869)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014870)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014871)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014872)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/22:0) (PathBank: SMP0014873)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/22:1(13Z)) (PathBank: SMP0014874)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/22:2(13Z,16Z)) (PathBank: SMP0014875)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014876)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014877)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014878)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014879)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/24:0) (PathBank: SMP0014880)
- Phosphatidylcholine Biosynthesis PC(22:2(13Z,16Z)/24:1(15Z)) (PathBank: SMP0014881)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/14:0) (PathBank: SMP0014882)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/14:1(9Z)) (PathBank: SMP0014883)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/15:0) (PathBank: SMP0014884)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/16:0) (PathBank: SMP0014885)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/16:1(9Z)) (PathBank: SMP0014886)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/18:0) (PathBank: SMP0014887)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/18:1(11Z)) (PathBank: SMP0014888)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/18:1(9Z)) (PathBank: SMP0014889)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/18:2(9Z,12Z)) (PathBank: SMP0014890)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014891)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014892)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014893)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:0) (PathBank: SMP0014894)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:1(11Z)) (PathBank: SMP0014895)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:2(11Z,14Z)) (PathBank: SMP0014896)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014897)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014898)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014899)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014900)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014901)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/22:0) (PathBank: SMP0014902)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/22:1(13Z)) (PathBank: SMP0014903)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/22:2(13Z,16Z)) (PathBank: SMP0014904)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014905)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014906)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014907)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014908)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/24:0) (PathBank: SMP0014909)
- Phosphatidylcholine Biosynthesis PC(22:4(7Z,10Z,13Z,16Z)/24:1(15Z)) (PathBank: SMP0014910)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/14:0) (PathBank: SMP0014911)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/14:1(9Z)) (PathBank: SMP0014912)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/15:0) (PathBank: SMP0014913)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/16:0) (PathBank: SMP0014914)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/16:1(9Z)) (PathBank: SMP0014915)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/18:0) (PathBank: SMP0014916)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/18:1(11Z)) (PathBank: SMP0014917)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/18:1(9Z)) (PathBank: SMP0014918)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/18:2(9Z,12Z)) (PathBank: SMP0014919)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014920)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014921)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014922)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:0) (PathBank: SMP0014923)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:1(11Z)) (PathBank: SMP0014924)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:2(11Z,14Z)) (PathBank: SMP0014925)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014926)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014927)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014928)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014929)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014930)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/22:0) (PathBank: SMP0014931)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/22:1(13Z)) (PathBank: SMP0014932)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/22:2(13Z,16Z)) (PathBank: SMP0014933)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014934)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014935)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014936)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014937)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/24:0) (PathBank: SMP0014938)
- Phosphatidylcholine Biosynthesis PC(22:5(4Z,7Z,10Z,13Z,16Z)/24:1(15Z)) (PathBank: SMP0014939)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/14:0) (PathBank: SMP0014940)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/14:1(9Z)) (PathBank: SMP0014941)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/15:0) (PathBank: SMP0014942)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/16:0) (PathBank: SMP0014943)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/16:1(9Z)) (PathBank: SMP0014944)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/18:0) (PathBank: SMP0014945)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/18:1(11Z)) (PathBank: SMP0014946)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/18:1(9Z)) (PathBank: SMP0014947)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/18:2(9Z,12Z)) (PathBank: SMP0014948)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014949)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014950)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014951)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:0) (PathBank: SMP0014952)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:1(11Z)) (PathBank: SMP0014953)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:2(11Z,14Z)) (PathBank: SMP0014954)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014955)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014956)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014957)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014958)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014959)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/22:0) (PathBank: SMP0014960)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/22:1(13Z)) (PathBank: SMP0014961)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/22:2(13Z,16Z)) (PathBank: SMP0014962)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014963)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014964)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014965)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014966)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/24:0) (PathBank: SMP0014967)
- Phosphatidylcholine Biosynthesis PC(22:5(7Z,10Z,13Z,16Z,19Z)/24:1(15Z)) (PathBank: SMP0014968)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/14:0) (PathBank: SMP0014969)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/14:1(9Z)) (PathBank: SMP0014970)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/15:0) (PathBank: SMP0014971)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/16:0) (PathBank: SMP0014972)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/16:1(9Z)) (PathBank: SMP0014973)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:0) (PathBank: SMP0014974)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:1(11Z)) (PathBank: SMP0014975)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:1(9Z)) (PathBank: SMP0014976)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:2(9Z,12Z)) (PathBank: SMP0014977)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0014978)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0014979)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0014980)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:0) (PathBank: SMP0014981)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:1(11Z)) (PathBank: SMP0014982)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:2(11Z,14Z)) (PathBank: SMP0014983)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0014984)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0014985)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0014986)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0014987)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0014988)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:0) (PathBank: SMP0014989)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:1(13Z)) (PathBank: SMP0014990)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:2(13Z,16Z)) (PathBank: SMP0014991)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0014992)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0014993)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014994)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0014995)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/24:0) (PathBank: SMP0014996)
- Phosphatidylcholine Biosynthesis PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/24:1(15Z)) (PathBank: SMP0014997)
- Phosphatidylcholine Biosynthesis PC(24:0/14:0) (PathBank: SMP0014998)
- Phosphatidylcholine Biosynthesis PC(24:0/14:1(9Z)) (PathBank: SMP0014999)
- Phosphatidylcholine Biosynthesis PC(24:0/15:0) (PathBank: SMP0015000)
- Phosphatidylcholine Biosynthesis PC(24:0/16:0) (PathBank: SMP0015001)
- Phosphatidylcholine Biosynthesis PC(24:0/16:1(9Z)) (PathBank: SMP0015002)
- Phosphatidylcholine Biosynthesis PC(24:0/18:0) (PathBank: SMP0015003)
- Phosphatidylcholine Biosynthesis PC(24:0/18:1(11Z)) (PathBank: SMP0015004)
- Phosphatidylcholine Biosynthesis PC(24:0/18:1(9Z)) (PathBank: SMP0015005)
- Phosphatidylcholine Biosynthesis PC(24:0/18:2(9Z,12Z)) (PathBank: SMP0015006)
- Phosphatidylcholine Biosynthesis PC(24:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015007)
- Phosphatidylcholine Biosynthesis PC(24:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015008)
- Phosphatidylcholine Biosynthesis PC(24:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015009)
- Phosphatidylcholine Biosynthesis PC(24:0/20:0) (PathBank: SMP0015010)
- Phosphatidylcholine Biosynthesis PC(24:0/20:1(11Z)) (PathBank: SMP0015011)
- Phosphatidylcholine Biosynthesis PC(24:0/20:2(11Z,14Z)) (PathBank: SMP0015012)
- Phosphatidylcholine Biosynthesis PC(24:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015013)
- Phosphatidylcholine Biosynthesis PC(24:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015014)
- Phosphatidylcholine Biosynthesis PC(24:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015015)
- Phosphatidylcholine Biosynthesis PC(24:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015016)
- Phosphatidylcholine Biosynthesis PC(24:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015017)
- Phosphatidylcholine Biosynthesis PC(24:0/22:0) (PathBank: SMP0015018)
- Phosphatidylcholine Biosynthesis PC(24:0/22:1(13Z)) (PathBank: SMP0015019)
- Phosphatidylcholine Biosynthesis PC(24:0/22:2(13Z,16Z)) (PathBank: SMP0015020)
- Phosphatidylcholine Biosynthesis PC(24:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015021)
- Phosphatidylcholine Biosynthesis PC(24:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015022)
- Phosphatidylcholine Biosynthesis PC(24:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015023)
- Phosphatidylcholine Biosynthesis PC(24:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015024)
- Phosphatidylcholine Biosynthesis PC(24:0/24:0) (PathBank: SMP0015025)
- Phosphatidylcholine Biosynthesis PC(24:0/24:1(15Z)) (PathBank: SMP0015026)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/14:0) (PathBank: SMP0015027)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/14:1(9Z)) (PathBank: SMP0015028)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/15:0) (PathBank: SMP0015029)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/16:0) (PathBank: SMP0015030)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/16:1(9Z)) (PathBank: SMP0015031)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/18:0) (PathBank: SMP0015032)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/18:1(11Z)) (PathBank: SMP0015033)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/18:1(9Z)) (PathBank: SMP0015034)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/18:2(9Z,12Z)) (PathBank: SMP0015035)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015036)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015037)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015038)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:0) (PathBank: SMP0015039)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:1(11Z)) (PathBank: SMP0015040)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:2(11Z,14Z)) (PathBank: SMP0015041)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015042)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015043)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015044)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015045)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015046)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/22:0) (PathBank: SMP0015047)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/22:1(13Z)) (PathBank: SMP0015048)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/22:2(13Z,16Z)) (PathBank: SMP0015049)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015050)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015051)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015052)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015053)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/24:0) (PathBank: SMP0015054)
- Phosphatidylcholine Biosynthesis PC(24:1(15Z)/24:1(15Z)) (PathBank: SMP0015055)
- Phosphatidylethanolamine Biosynthesis PE(14:0/14:0) (PathBank: SMP0015056)
- Phosphatidylethanolamine Biosynthesis PE(14:0/14:1(9Z)) (PathBank: SMP0015057)
- Phosphatidylethanolamine Biosynthesis PE(14:0/15:0) (PathBank: SMP0015058)
- Phosphatidylethanolamine Biosynthesis PE(14:0/16:0) (PathBank: SMP0015059)
- Phosphatidylethanolamine Biosynthesis PE(14:0/16:1(9Z)) (PathBank: SMP0015060)
- Phosphatidylethanolamine Biosynthesis PE(14:0/18:0) (PathBank: SMP0015061)
- Phosphatidylethanolamine Biosynthesis PE(14:0/18:1(11Z)) (PathBank: SMP0015062)
- Phosphatidylethanolamine Biosynthesis PE(14:0/18:1(9Z)) (PathBank: SMP0015063)
- Phosphatidylethanolamine Biosynthesis PE(14:0/18:2(9Z,12Z)) (PathBank: SMP0015064)
- Phosphatidylethanolamine Biosynthesis PE(14:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015065)
- Phosphatidylethanolamine Biosynthesis PE(14:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015066)
- Phosphatidylethanolamine Biosynthesis PE(14:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015067)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:0) (PathBank: SMP0015068)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:1(11Z)) (PathBank: SMP0015069)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:2(11Z,14Z)) (PathBank: SMP0015070)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015071)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015072)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015073)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015074)
- Phosphatidylethanolamine Biosynthesis PE(14:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015075)
- Phosphatidylethanolamine Biosynthesis PE(14:0/22:0) (PathBank: SMP0015076)
- Phosphatidylethanolamine Biosynthesis PE(14:0/22:1(13Z)) (PathBank: SMP0015077)
- Phosphatidylethanolamine Biosynthesis PE(14:0/22:2(13Z,16Z)) (PathBank: SMP0015078)
- Phosphatidylethanolamine Biosynthesis PE(14:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015079)
- Phosphatidylethanolamine Biosynthesis PE(14:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015080)
- Phosphatidylethanolamine Biosynthesis PE(14:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015081)
- Phosphatidylethanolamine Biosynthesis PE(14:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015082)
- Phosphatidylethanolamine Biosynthesis PE(14:0/24:0) (PathBank: SMP0015083)
- Phosphatidylethanolamine Biosynthesis PE(14:0/24:1(15Z)) (PathBank: SMP0015084)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/14:0) (PathBank: SMP0015085)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/14:1(9Z)) (PathBank: SMP0015086)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/15:0) (PathBank: SMP0015087)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/16:0) (PathBank: SMP0015088)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/16:1(9Z)) (PathBank: SMP0015089)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/18:0) (PathBank: SMP0015090)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/18:1(11Z)) (PathBank: SMP0015091)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/18:1(9Z)) (PathBank: SMP0015092)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0015093)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015094)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015095)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015096)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:0) (PathBank: SMP0015097)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:1(11Z)) (PathBank: SMP0015098)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:2(11Z,14Z)) (PathBank: SMP0015099)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015100)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015101)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015102)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015103)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015104)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/22:0) (PathBank: SMP0015105)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/22:1(13Z)) (PathBank: SMP0015106)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/22:2(13Z,16Z)) (PathBank: SMP0015107)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015108)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015109)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015110)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015111)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/24:0) (PathBank: SMP0015112)
- Phosphatidylethanolamine Biosynthesis PE(14:1(9Z)/24:1(15Z)) (PathBank: SMP0015113)
- Phosphatidylethanolamine Biosynthesis PE(15:0/14:0) (PathBank: SMP0015114)
- Phosphatidylethanolamine Biosynthesis PE(15:0/14:1(9Z)) (PathBank: SMP0015115)
- Phosphatidylethanolamine Biosynthesis PE(15:0/15:0) (PathBank: SMP0015116)
- Phosphatidylethanolamine Biosynthesis PE(15:0/16:0) (PathBank: SMP0015117)
- Phosphatidylethanolamine Biosynthesis PE(15:0/16:1(9Z)) (PathBank: SMP0015118)
- Phosphatidylethanolamine Biosynthesis PE(15:0/18:0) (PathBank: SMP0015119)
- Phosphatidylethanolamine Biosynthesis PE(15:0/18:1(11Z)) (PathBank: SMP0015120)
- Phosphatidylethanolamine Biosynthesis PE(15:0/18:1(9Z)) (PathBank: SMP0015121)
- Phosphatidylethanolamine Biosynthesis PE(15:0/18:2(9Z,12Z)) (PathBank: SMP0015122)
- Phosphatidylethanolamine Biosynthesis PE(15:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015123)
- Phosphatidylethanolamine Biosynthesis PE(15:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015124)
- Phosphatidylethanolamine Biosynthesis PE(15:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015125)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:0) (PathBank: SMP0015126)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:1(11Z)) (PathBank: SMP0015127)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:2(11Z,14Z)) (PathBank: SMP0015128)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015129)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015130)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015131)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015132)
- Phosphatidylethanolamine Biosynthesis PE(15:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015133)
- Phosphatidylethanolamine Biosynthesis PE(15:0/22:0) (PathBank: SMP0015134)
- Phosphatidylethanolamine Biosynthesis PE(15:0/22:1(13Z)) (PathBank: SMP0015135)
- Phosphatidylethanolamine Biosynthesis PE(15:0/22:2(13Z,16Z)) (PathBank: SMP0015136)
- Phosphatidylethanolamine Biosynthesis PE(15:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015137)
- Phosphatidylethanolamine Biosynthesis PE(15:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015138)
- Phosphatidylethanolamine Biosynthesis PE(15:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015139)
- Phosphatidylethanolamine Biosynthesis PE(15:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015140)
- Phosphatidylethanolamine Biosynthesis PE(15:0/24:0) (PathBank: SMP0015141)
- Phosphatidylethanolamine Biosynthesis PE(15:0/24:1(15Z)) (PathBank: SMP0015142)
- Phosphatidylethanolamine Biosynthesis PE(16:0/14:0) (PathBank: SMP0015143)
- Phosphatidylethanolamine Biosynthesis PE(16:0/14:1(9Z)) (PathBank: SMP0015144)
- Phosphatidylethanolamine Biosynthesis PE(16:0/15:0) (PathBank: SMP0015145)
- Phosphatidylethanolamine Biosynthesis PE(16:0/16:0) (PathBank: SMP0015146)
- Phosphatidylethanolamine Biosynthesis PE(16:0/16:1(9Z)) (PathBank: SMP0015147)
- Phosphatidylethanolamine Biosynthesis PE(16:0/18:0) (PathBank: SMP0015148)
- Phosphatidylethanolamine Biosynthesis PE(16:0/18:1(11Z)) (PathBank: SMP0015149)
- Phosphatidylethanolamine Biosynthesis PE(16:0/18:1(9Z)) (PathBank: SMP0015150)
- Phosphatidylethanolamine Biosynthesis PE(16:0/18:2(9Z,12Z)) (PathBank: SMP0015151)
- Phosphatidylethanolamine Biosynthesis PE(16:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015152)
- Phosphatidylethanolamine Biosynthesis PE(16:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015153)
- Phosphatidylethanolamine Biosynthesis PE(16:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015154)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:0) (PathBank: SMP0015155)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:1(11Z)) (PathBank: SMP0015156)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:2(11Z,14Z)) (PathBank: SMP0015157)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015158)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015159)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015160)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015161)
- Phosphatidylethanolamine Biosynthesis PE(16:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015162)
- Phosphatidylethanolamine Biosynthesis PE(16:0/22:0) (PathBank: SMP0015163)
- Phosphatidylethanolamine Biosynthesis PE(16:0/22:1(13Z)) (PathBank: SMP0015164)
- Phosphatidylethanolamine Biosynthesis PE(16:0/22:2(13Z,16Z)) (PathBank: SMP0015165)
- Phosphatidylethanolamine Biosynthesis PE(16:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015166)
- Phosphatidylethanolamine Biosynthesis PE(16:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015167)
- Phosphatidylethanolamine Biosynthesis PE(16:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015168)
- Phosphatidylethanolamine Biosynthesis PE(16:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015169)
- Phosphatidylethanolamine Biosynthesis PE(16:0/24:0) (PathBank: SMP0015170)
- Phosphatidylethanolamine Biosynthesis PE(16:0/24:1(15Z)) (PathBank: SMP0015171)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/14:0) (PathBank: SMP0015172)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/14:1(9Z)) (PathBank: SMP0015173)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/15:0) (PathBank: SMP0015174)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/16:0) (PathBank: SMP0015175)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/16:1(9Z)) (PathBank: SMP0015176)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/18:1(11Z)) (PathBank: SMP0015177)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/18:1(9Z)) (PathBank: SMP0015178)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0015179)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015180)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015181)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015182)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:0) (PathBank: SMP0015183)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:1(11Z)) (PathBank: SMP0015184)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:2(11Z,14Z)) (PathBank: SMP0015185)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015186)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015187)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015188)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015189)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015190)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/22:0) (PathBank: SMP0015191)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/22:1(13Z)) (PathBank: SMP0015192)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/22:2(13Z,16Z)) (PathBank: SMP0015193)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015194)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015195)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015196)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015197)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/24:0) (PathBank: SMP0015198)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/24:1(15Z)) (PathBank: SMP0015199)
- Phosphatidylethanolamine Biosynthesis PE(18:0/14:0) (PathBank: SMP0015200)
- Phosphatidylethanolamine Biosynthesis PE(18:0/14:1(9Z)) (PathBank: SMP0015201)
- Phosphatidylethanolamine Biosynthesis PE(18:0/15:0) (PathBank: SMP0015202)
- Phosphatidylethanolamine Biosynthesis PE(18:0/16:0) (PathBank: SMP0015203)
- Phosphatidylethanolamine Biosynthesis PE(18:0/16:1(9Z)) (PathBank: SMP0015204)
- Phosphatidylethanolamine Biosynthesis PE(18:0/18:0) (PathBank: SMP0015205)
- Phosphatidylethanolamine Biosynthesis PE(18:0/18:1(11Z)) (PathBank: SMP0015206)
- Phosphatidylethanolamine Biosynthesis PE(18:0/18:1(9Z)) (PathBank: SMP0015207)
- Phosphatidylethanolamine Biosynthesis PE(18:0/18:2(9Z,12Z)) (PathBank: SMP0015208)
- Phosphatidylethanolamine Biosynthesis PE(18:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015209)
- Phosphatidylethanolamine Biosynthesis PE(18:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015210)
- Phosphatidylethanolamine Biosynthesis PE(18:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015211)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:0) (PathBank: SMP0015212)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:1(11Z)) (PathBank: SMP0015213)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:2(11Z,14Z)) (PathBank: SMP0015214)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015215)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015216)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015217)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015218)
- Phosphatidylethanolamine Biosynthesis PE(18:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015219)
- Phosphatidylethanolamine Biosynthesis PE(18:0/22:0) (PathBank: SMP0015220)
- Phosphatidylethanolamine Biosynthesis PE(18:0/22:1(13Z)) (PathBank: SMP0015221)
- Phosphatidylethanolamine Biosynthesis PE(18:0/22:2(13Z,16Z)) (PathBank: SMP0015222)
- Phosphatidylethanolamine Biosynthesis PE(18:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015223)
- Phosphatidylethanolamine Biosynthesis PE(18:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015224)
- Phosphatidylethanolamine Biosynthesis PE(18:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015225)
- Phosphatidylethanolamine Biosynthesis PE(18:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015226)
- Phosphatidylethanolamine Biosynthesis PE(18:0/24:0) (PathBank: SMP0015227)
- Phosphatidylethanolamine Biosynthesis PE(18:0/24:1(15Z)) (PathBank: SMP0015228)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/14:0) (PathBank: SMP0015229)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/14:1(9Z)) (PathBank: SMP0015230)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/15:0) (PathBank: SMP0015231)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/16:0) (PathBank: SMP0015232)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/16:1(9Z)) (PathBank: SMP0015233)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/18:0) (PathBank: SMP0015234)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/18:1(11Z)) (PathBank: SMP0015235)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/18:1(9Z)) (PathBank: SMP0015236)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/18:2(9Z,12Z)) (PathBank: SMP0015237)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015238)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015239)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015240)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:0) (PathBank: SMP0015241)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:1(11Z)) (PathBank: SMP0015242)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:2(11Z,14Z)) (PathBank: SMP0015243)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015244)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015245)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015246)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015247)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015248)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/22:0) (PathBank: SMP0015249)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/22:1(13Z)) (PathBank: SMP0015250)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/22:2(13Z,16Z)) (PathBank: SMP0015251)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015252)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015253)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015254)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015255)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/24:0) (PathBank: SMP0015256)
- Phosphatidylethanolamine Biosynthesis PE(18:1(11Z)/24:1(15Z)) (PathBank: SMP0015257)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/14:0) (PathBank: SMP0015258)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/14:1(9Z)) (PathBank: SMP0015259)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/15:0) (PathBank: SMP0015260)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/16:0) (PathBank: SMP0015261)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/16:1(9Z)) (PathBank: SMP0015262)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/18:0) (PathBank: SMP0015263)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/18:1(11Z)) (PathBank: SMP0015264)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/18:1(9Z)) (PathBank: SMP0015265)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0015266)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015267)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015268)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015269)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:0) (PathBank: SMP0015270)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:1(11Z)) (PathBank: SMP0015271)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:2(11Z,14Z)) (PathBank: SMP0015272)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015273)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015274)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015275)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015276)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015277)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/22:0) (PathBank: SMP0015278)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/22:1(13Z)) (PathBank: SMP0015279)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/22:2(13Z,16Z)) (PathBank: SMP0015280)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015281)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015282)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015283)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015284)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/24:0) (PathBank: SMP0015285)
- Phosphatidylethanolamine Biosynthesis PE(18:1(9Z)/24:1(15Z)) (PathBank: SMP0015286)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/14:0) (PathBank: SMP0015287)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/14:1(9Z)) (PathBank: SMP0015288)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/15:0) (PathBank: SMP0015289)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/16:0) (PathBank: SMP0015290)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/16:1(9Z)) (PathBank: SMP0015291)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/18:0) (PathBank: SMP0015292)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/18:1(11Z)) (PathBank: SMP0015293)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/18:1(9Z)) (PathBank: SMP0015294)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/18:2(9Z,12Z)) (PathBank: SMP0015295)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015296)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015297)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015298)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:0) (PathBank: SMP0015299)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:1(11Z)) (PathBank: SMP0015300)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:2(11Z,14Z)) (PathBank: SMP0015301)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015302)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015303)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015304)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015305)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015306)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/22:0) (PathBank: SMP0015307)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/22:1(13Z)) (PathBank: SMP0015308)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/22:2(13Z,16Z)) (PathBank: SMP0015309)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015310)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015311)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015312)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015313)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/24:0) (PathBank: SMP0015314)
- Phosphatidylethanolamine Biosynthesis PE(18:2(9Z,12Z)/24:1(15Z)) (PathBank: SMP0015315)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/14:0) (PathBank: SMP0015316)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/14:1(9Z)) (PathBank: SMP0015317)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/15:0) (PathBank: SMP0015318)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/16:0) (PathBank: SMP0015319)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/16:1(9Z)) (PathBank: SMP0015320)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/18:0) (PathBank: SMP0015321)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/18:1(11Z)) (PathBank: SMP0015322)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/18:1(9Z)) (PathBank: SMP0015323)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/18:2(9Z,12Z)) (PathBank: SMP0015324)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015325)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015326)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015327)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:0) (PathBank: SMP0015328)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:1(11Z)) (PathBank: SMP0015329)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:2(11Z,14Z)) (PathBank: SMP0015330)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015331)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015332)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015333)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015334)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015335)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/22:0) (PathBank: SMP0015336)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/22:1(13Z)) (PathBank: SMP0015337)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/22:2(13Z,16Z)) (PathBank: SMP0015338)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015339)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015340)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015341)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015342)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/24:0) (PathBank: SMP0015343)
- Phosphatidylethanolamine Biosynthesis PE(18:3(6Z,9Z,12Z)/24:1(15Z)) (PathBank: SMP0015344)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/14:0) (PathBank: SMP0015345)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/14:1(9Z)) (PathBank: SMP0015346)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/15:0) (PathBank: SMP0015347)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/16:0) (PathBank: SMP0015348)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/16:1(9Z)) (PathBank: SMP0015349)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/18:0) (PathBank: SMP0015350)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/18:1(11Z)) (PathBank: SMP0015351)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/18:1(9Z)) (PathBank: SMP0015352)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015354)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015355)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015356)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:0) (PathBank: SMP0015357)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:1(11Z)) (PathBank: SMP0015358)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:2(11Z,14Z)) (PathBank: SMP0015359)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015360)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015361)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015362)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015363)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015364)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/22:0) (PathBank: SMP0015365)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/22:1(13Z)) (PathBank: SMP0015366)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/22:2(13Z,16Z)) (PathBank: SMP0015367)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015368)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015369)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015370)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015371)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/24:0) (PathBank: SMP0015372)
- Phosphatidylethanolamine Biosynthesis PE(18:3(9Z,12Z,15Z)/24:1(15Z)) (PathBank: SMP0015373)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/14:0) (PathBank: SMP0015374)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/14:1(9Z)) (PathBank: SMP0015375)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/15:0) (PathBank: SMP0015376)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/16:0) (PathBank: SMP0015377)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/16:1(9Z)) (PathBank: SMP0015378)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/18:0) (PathBank: SMP0015379)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/18:1(11Z)) (PathBank: SMP0015380)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/18:1(9Z)) (PathBank: SMP0015381)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/18:2(9Z,12Z)) (PathBank: SMP0015382)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015383)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015384)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015385)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:0) (PathBank: SMP0015386)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:1(11Z)) (PathBank: SMP0015387)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:2(11Z,14Z)) (PathBank: SMP0015388)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015389)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015390)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015391)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015392)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015393)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/22:0) (PathBank: SMP0015394)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/22:1(13Z)) (PathBank: SMP0015395)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/22:2(13Z,16Z)) (PathBank: SMP0015396)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015397)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015398)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015399)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015400)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/24:0) (PathBank: SMP0015401)
- Phosphatidylethanolamine Biosynthesis PE(18:4(6Z,9Z,12Z,15Z)/24:1(15Z)) (PathBank: SMP0015402)
- Phosphatidylethanolamine Biosynthesis PE(20:0/14:0) (PathBank: SMP0015403)
- Phosphatidylethanolamine Biosynthesis PE(20:0/14:1(9Z)) (PathBank: SMP0015404)
- Phosphatidylethanolamine Biosynthesis PE(20:0/15:0) (PathBank: SMP0015405)
- Phosphatidylethanolamine Biosynthesis PE(20:0/16:0) (PathBank: SMP0015406)
- Phosphatidylethanolamine Biosynthesis PE(20:0/16:1(9Z)) (PathBank: SMP0015407)
- Phosphatidylethanolamine Biosynthesis PE(20:0/18:0) (PathBank: SMP0015408)
- Phosphatidylethanolamine Biosynthesis PE(20:0/18:1(11Z)) (PathBank: SMP0015409)
- Phosphatidylethanolamine Biosynthesis PE(20:0/18:1(9Z)) (PathBank: SMP0015410)
- Phosphatidylethanolamine Biosynthesis PE(20:0/18:2(9Z,12Z)) (PathBank: SMP0015411)
- Phosphatidylethanolamine Biosynthesis PE(20:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015412)
- Phosphatidylethanolamine Biosynthesis PE(20:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015413)
- Phosphatidylethanolamine Biosynthesis PE(20:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015414)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:0) (PathBank: SMP0015415)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:1(11Z)) (PathBank: SMP0015416)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:2(11Z,14Z)) (PathBank: SMP0015417)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015418)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015419)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015420)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015421)
- Phosphatidylethanolamine Biosynthesis PE(20:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015422)
- Phosphatidylethanolamine Biosynthesis PE(20:0/22:0) (PathBank: SMP0015423)
- Phosphatidylethanolamine Biosynthesis PE(20:0/22:1(13Z)) (PathBank: SMP0015424)
- Phosphatidylethanolamine Biosynthesis PE(20:0/22:2(13Z,16Z)) (PathBank: SMP0015425)
- Phosphatidylethanolamine Biosynthesis PE(20:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015426)
- Phosphatidylethanolamine Biosynthesis PE(20:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015427)
- Phosphatidylethanolamine Biosynthesis PE(20:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015428)
- Phosphatidylethanolamine Biosynthesis PE(20:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015429)
- Phosphatidylethanolamine Biosynthesis PE(20:0/24:0) (PathBank: SMP0015430)
- Phosphatidylethanolamine Biosynthesis PE(20:0/24:1(15Z)) (PathBank: SMP0015431)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/14:0) (PathBank: SMP0015432)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/14:1(9Z)) (PathBank: SMP0015433)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/15:0) (PathBank: SMP0015434)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/16:0) (PathBank: SMP0015435)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/16:1(9Z)) (PathBank: SMP0015436)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/18:0) (PathBank: SMP0015437)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/18:1(11Z)) (PathBank: SMP0015438)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/18:1(9Z)) (PathBank: SMP0015439)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/18:2(9Z,12Z)) (PathBank: SMP0015440)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015441)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015442)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015443)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:0) (PathBank: SMP0015444)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:1(11Z)) (PathBank: SMP0015445)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:2(11Z,14Z)) (PathBank: SMP0015446)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015447)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015448)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015449)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015450)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015451)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/22:0) (PathBank: SMP0015452)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/22:1(13Z)) (PathBank: SMP0015453)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/22:2(13Z,16Z)) (PathBank: SMP0015454)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015455)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015456)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015457)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015458)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/24:0) (PathBank: SMP0015459)
- Phosphatidylethanolamine Biosynthesis PE(20:1(11Z)/24:1(15Z)) (PathBank: SMP0015460)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/14:0) (PathBank: SMP0015461)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/14:1(9Z)) (PathBank: SMP0015462)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/15:0) (PathBank: SMP0015463)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/16:0) (PathBank: SMP0015464)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/16:1(9Z)) (PathBank: SMP0015465)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/18:0) (PathBank: SMP0015466)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/18:1(11Z)) (PathBank: SMP0015467)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/18:1(9Z)) (PathBank: SMP0015468)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/18:2(9Z,12Z)) (PathBank: SMP0015469)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015470)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015471)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015472)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:0) (PathBank: SMP0015473)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:1(11Z)) (PathBank: SMP0015474)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:2(11Z,14Z)) (PathBank: SMP0015475)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015476)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015477)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015478)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015479)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015480)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/22:0) (PathBank: SMP0015481)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/22:1(13Z)) (PathBank: SMP0015482)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/22:2(13Z,16Z)) (PathBank: SMP0015483)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015484)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015485)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015486)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015487)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/24:0) (PathBank: SMP0015488)
- Phosphatidylethanolamine Biosynthesis PE(20:2(11Z,14Z)/24:1(15Z)) (PathBank: SMP0015489)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/14:0) (PathBank: SMP0015490)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/14:1(9Z)) (PathBank: SMP0015491)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/15:0) (PathBank: SMP0015492)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/16:0) (PathBank: SMP0015493)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/16:1(9Z)) (PathBank: SMP0015494)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/18:0) (PathBank: SMP0015495)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/18:1(11Z)) (PathBank: SMP0015496)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/18:1(9Z)) (PathBank: SMP0015497)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/18:2(9Z,12Z)) (PathBank: SMP0015498)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015499)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015500)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015501)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:0) (PathBank: SMP0015502)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:1(11Z)) (PathBank: SMP0015503)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:2(11Z,14Z)) (PathBank: SMP0015504)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015505)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015506)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015507)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015508)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015509)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/22:0) (PathBank: SMP0015510)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/22:1(13Z)) (PathBank: SMP0015511)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/22:2(13Z,16Z)) (PathBank: SMP0015512)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015513)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015514)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015515)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015516)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/24:0) (PathBank: SMP0015517)
- Phosphatidylethanolamine Biosynthesis PE(20:3(5Z,8Z,11Z)/24:1(15Z)) (PathBank: SMP0015518)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/14:0) (PathBank: SMP0015519)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/14:1(9Z)) (PathBank: SMP0015520)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/15:0) (PathBank: SMP0015521)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/16:0) (PathBank: SMP0015522)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/16:1(9Z)) (PathBank: SMP0015523)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/18:0) (PathBank: SMP0015524)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/18:1(11Z)) (PathBank: SMP0015525)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/18:1(9Z)) (PathBank: SMP0015526)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/18:2(9Z,12Z)) (PathBank: SMP0015527)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015528)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015529)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015530)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/20:0) (PathBank: SMP0015531)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/20:2(11Z,14Z)) (PathBank: SMP0015533)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015534)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015535)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015536)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015537)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015538)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/22:0) (PathBank: SMP0015539)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/22:1(13Z)) (PathBank: SMP0015540)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/22:2(13Z,16Z)) (PathBank: SMP0015541)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015542)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015543)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015544)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015545)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/24:0) (PathBank: SMP0015546)
- Phosphatidylethanolamine Biosynthesis PE(20:3(8Z,11Z,14Z)/24:1(15Z)) (PathBank: SMP0015547)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/14:0) (PathBank: SMP0015548)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/14:1(9Z)) (PathBank: SMP0015549)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/15:0) (PathBank: SMP0015550)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/16:0) (PathBank: SMP0015551)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/16:1(9Z)) (PathBank: SMP0015552)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/18:0) (PathBank: SMP0015553)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/18:1(11Z)) (PathBank: SMP0015554)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/18:1(9Z)) (PathBank: SMP0015555)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/18:2(9Z,12Z)) (PathBank: SMP0015556)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015557)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015558)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015559)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:0) (PathBank: SMP0015560)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:1(11Z)) (PathBank: SMP0015561)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:2(11Z,14Z)) (PathBank: SMP0015562)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015563)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015564)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015565)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015566)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015567)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/22:0) (PathBank: SMP0015568)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/22:1(13Z)) (PathBank: SMP0015569)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/22:2(13Z,16Z)) (PathBank: SMP0015570)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015571)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015572)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015573)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015574)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/24:0) (PathBank: SMP0015575)
- Phosphatidylethanolamine Biosynthesis PE(20:4(5Z,8Z,11Z,14Z)/24:1(15Z)) (PathBank: SMP0015576)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/14:0) (PathBank: SMP0015577)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/14:1(9Z)) (PathBank: SMP0015578)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/15:0) (PathBank: SMP0015579)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/16:0) (PathBank: SMP0015580)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/16:1(9Z)) (PathBank: SMP0015581)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/18:0) (PathBank: SMP0015582)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/18:1(11Z)) (PathBank: SMP0015583)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/18:1(9Z)) (PathBank: SMP0015584)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/18:2(9Z,12Z)) (PathBank: SMP0015585)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015586)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015587)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015588)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:0) (PathBank: SMP0015589)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:1(11Z)) (PathBank: SMP0015590)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:2(11Z,14Z)) (PathBank: SMP0015591)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015592)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015593)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015594)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015595)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015596)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/22:0) (PathBank: SMP0015597)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/22:1(13Z)) (PathBank: SMP0015598)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/22:2(13Z,16Z)) (PathBank: SMP0015599)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015600)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015601)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015602)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015603)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/24:0) (PathBank: SMP0015604)
- Phosphatidylethanolamine Biosynthesis PE(20:4(8Z,11Z,14Z,17Z)/24:1(15Z)) (PathBank: SMP0015605)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/14:0) (PathBank: SMP0015606)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/14:1(9Z)) (PathBank: SMP0015607)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/15:0) (PathBank: SMP0015608)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/16:0) (PathBank: SMP0015609)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/16:1(9Z)) (PathBank: SMP0015610)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/18:0) (PathBank: SMP0015611)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/18:1(11Z)) (PathBank: SMP0015612)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/18:1(9Z)) (PathBank: SMP0015613)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/18:2(9Z,12Z)) (PathBank: SMP0015614)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015615)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015616)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015617)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:0) (PathBank: SMP0015618)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:1(11Z)) (PathBank: SMP0015619)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:2(11Z,14Z)) (PathBank: SMP0015620)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015621)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015622)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015623)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015624)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015625)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/22:0) (PathBank: SMP0015626)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/22:1(13Z)) (PathBank: SMP0015627)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/22:2(13Z,16Z)) (PathBank: SMP0015628)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015629)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015630)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015631)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015632)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/24:0) (PathBank: SMP0015633)
- Phosphatidylethanolamine Biosynthesis PE(20:5(5Z,8Z,11Z,14Z,17Z)/24:1(15Z)) (PathBank: SMP0015634)
- Phosphatidylethanolamine Biosynthesis PE(22:0/14:0) (PathBank: SMP0015635)
- Phosphatidylethanolamine Biosynthesis PE(22:0/14:1(9Z)) (PathBank: SMP0015636)
- Phosphatidylethanolamine Biosynthesis PE(22:0/15:0) (PathBank: SMP0015637)
- Phosphatidylethanolamine Biosynthesis PE(22:0/16:0) (PathBank: SMP0015638)
- Phosphatidylethanolamine Biosynthesis PE(22:0/16:1(9Z)) (PathBank: SMP0015639)
- Phosphatidylethanolamine Biosynthesis PE(22:0/18:0) (PathBank: SMP0015640)
- Phosphatidylethanolamine Biosynthesis PE(22:0/18:1(11Z)) (PathBank: SMP0015641)
- Phosphatidylethanolamine Biosynthesis PE(22:0/18:1(9Z)) (PathBank: SMP0015642)
- Phosphatidylethanolamine Biosynthesis PE(22:0/18:2(9Z,12Z)) (PathBank: SMP0015643)
- Phosphatidylethanolamine Biosynthesis PE(22:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015644)
- Phosphatidylethanolamine Biosynthesis PE(22:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015645)
- Phosphatidylethanolamine Biosynthesis PE(22:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015646)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:0) (PathBank: SMP0015647)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:1(11Z)) (PathBank: SMP0015648)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:2(11Z,14Z)) (PathBank: SMP0015649)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015650)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015651)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015652)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015653)
- Phosphatidylethanolamine Biosynthesis PE(22:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015654)
- Phosphatidylethanolamine Biosynthesis PE(22:0/22:0) (PathBank: SMP0015655)
- Phosphatidylethanolamine Biosynthesis PE(22:0/22:1(13Z)) (PathBank: SMP0015656)
- Phosphatidylethanolamine Biosynthesis PE(22:0/22:2(13Z,16Z)) (PathBank: SMP0015657)
- Phosphatidylethanolamine Biosynthesis PE(22:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015658)
- Phosphatidylethanolamine Biosynthesis PE(22:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015659)
- Phosphatidylethanolamine Biosynthesis PE(22:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015660)
- Phosphatidylethanolamine Biosynthesis PE(22:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015661)
- Phosphatidylethanolamine Biosynthesis PE(22:0/24:0) (PathBank: SMP0015662)
- Phosphatidylethanolamine Biosynthesis PE(22:0/24:1(15Z)) (PathBank: SMP0015663)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/14:0) (PathBank: SMP0015664)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/14:1(9Z)) (PathBank: SMP0015665)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/15:0) (PathBank: SMP0015666)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/16:0) (PathBank: SMP0015667)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/16:1(9Z)) (PathBank: SMP0015668)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/18:0) (PathBank: SMP0015669)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/18:1(9Z)) (PathBank: SMP0015671)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/18:2(9Z,12Z)) (PathBank: SMP0015672)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015673)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015674)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015675)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:0) (PathBank: SMP0015676)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:1(11Z)) (PathBank: SMP0015677)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:2(11Z,14Z)) (PathBank: SMP0015678)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015679)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015680)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015681)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015682)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015683)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/22:0) (PathBank: SMP0015684)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/22:1(13Z)) (PathBank: SMP0015685)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/22:2(13Z,16Z)) (PathBank: SMP0015686)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015687)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015688)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015689)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015690)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/24:0) (PathBank: SMP0015691)
- Phosphatidylethanolamine Biosynthesis PE(22:1(13Z)/24:1(15Z)) (PathBank: SMP0015692)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/14:0) (PathBank: SMP0015693)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/14:1(9Z)) (PathBank: SMP0015694)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/15:0) (PathBank: SMP0015695)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/16:0) (PathBank: SMP0015696)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/16:1(9Z)) (PathBank: SMP0015697)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/18:0) (PathBank: SMP0015698)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/18:1(11Z)) (PathBank: SMP0015699)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/18:1(9Z)) (PathBank: SMP0015700)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/18:2(9Z,12Z)) (PathBank: SMP0015701)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015702)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015703)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015704)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:0) (PathBank: SMP0015705)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:1(11Z)) (PathBank: SMP0015706)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:2(11Z,14Z)) (PathBank: SMP0015707)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015708)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015709)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015710)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015711)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015712)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/22:0) (PathBank: SMP0015713)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/22:1(13Z)) (PathBank: SMP0015714)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/22:2(13Z,16Z)) (PathBank: SMP0015715)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015716)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015717)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015718)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015719)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/24:0) (PathBank: SMP0015720)
- Phosphatidylethanolamine Biosynthesis PE(22:2(13Z,16Z)/24:1(15Z)) (PathBank: SMP0015721)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/14:0) (PathBank: SMP0015722)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/14:1(9Z)) (PathBank: SMP0015723)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/15:0) (PathBank: SMP0015724)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/16:0) (PathBank: SMP0015725)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/16:1(9Z)) (PathBank: SMP0015726)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/18:0) (PathBank: SMP0015727)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/18:1(11Z)) (PathBank: SMP0015728)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/18:1(9Z)) (PathBank: SMP0015729)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/18:2(9Z,12Z)) (PathBank: SMP0015730)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015731)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015732)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015733)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:0) (PathBank: SMP0015734)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:1(11Z)) (PathBank: SMP0015735)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:2(11Z,14Z)) (PathBank: SMP0015736)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015737)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015738)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015739)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015740)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015741)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/22:0) (PathBank: SMP0015742)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/22:1(13Z)) (PathBank: SMP0015743)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/22:2(13Z,16Z)) (PathBank: SMP0015744)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015745)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015746)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015747)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015748)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/24:0) (PathBank: SMP0015749)
- Phosphatidylethanolamine Biosynthesis PE(22:4(7Z,10Z,13Z,16Z)/24:1(15Z)) (PathBank: SMP0015750)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/14:0) (PathBank: SMP0015751)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/14:1(9Z)) (PathBank: SMP0015752)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/15:0) (PathBank: SMP0015753)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/16:0) (PathBank: SMP0015754)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/16:1(9Z)) (PathBank: SMP0015755)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/18:0) (PathBank: SMP0015756)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/18:1(11Z)) (PathBank: SMP0015757)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/18:1(9Z)) (PathBank: SMP0015758)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/18:2(9Z,12Z)) (PathBank: SMP0015759)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015760)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015761)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015762)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:0) (PathBank: SMP0015763)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:1(11Z)) (PathBank: SMP0015764)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:2(11Z,14Z)) (PathBank: SMP0015765)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015766)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015767)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015768)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015769)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015770)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/22:0) (PathBank: SMP0015771)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/22:1(13Z)) (PathBank: SMP0015772)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/22:2(13Z,16Z)) (PathBank: SMP0015773)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015774)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015775)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015776)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015777)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/24:0) (PathBank: SMP0015778)
- Phosphatidylethanolamine Biosynthesis PE(22:5(4Z,7Z,10Z,13Z,16Z)/24:1(15Z)) (PathBank: SMP0015779)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/14:0) (PathBank: SMP0015780)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/14:1(9Z)) (PathBank: SMP0015781)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/15:0) (PathBank: SMP0015782)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/16:0) (PathBank: SMP0015783)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/16:1(9Z)) (PathBank: SMP0015784)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/18:0) (PathBank: SMP0015785)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/18:1(11Z)) (PathBank: SMP0015786)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/18:1(9Z)) (PathBank: SMP0015787)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/18:2(9Z,12Z)) (PathBank: SMP0015788)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015789)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015790)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015791)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:0) (PathBank: SMP0015792)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:1(11Z)) (PathBank: SMP0015793)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:2(11Z,14Z)) (PathBank: SMP0015794)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015795)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015796)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015797)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015798)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015799)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/22:0) (PathBank: SMP0015800)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/22:1(13Z)) (PathBank: SMP0015801)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/22:2(13Z,16Z)) (PathBank: SMP0015802)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015803)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015804)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015805)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015806)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/24:0) (PathBank: SMP0015807)
- Phosphatidylethanolamine Biosynthesis PE(22:5(7Z,10Z,13Z,16Z,19Z)/24:1(15Z)) (PathBank: SMP0015808)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/14:0) (PathBank: SMP0015809)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/14:1(9Z)) (PathBank: SMP0015810)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/15:0) (PathBank: SMP0015811)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/16:0) (PathBank: SMP0015812)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/16:1(9Z)) (PathBank: SMP0015813)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:0) (PathBank: SMP0015814)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:1(11Z)) (PathBank: SMP0015815)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:1(9Z)) (PathBank: SMP0015816)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:2(9Z,12Z)) (PathBank: SMP0015817)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015818)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015819)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015820)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:0) (PathBank: SMP0015821)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:1(11Z)) (PathBank: SMP0015822)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:2(11Z,14Z)) (PathBank: SMP0015823)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015824)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015825)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015826)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015827)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015828)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:0) (PathBank: SMP0015829)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:1(13Z)) (PathBank: SMP0015830)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:2(13Z,16Z)) (PathBank: SMP0015831)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015832)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015833)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015834)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015835)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/24:0) (PathBank: SMP0015836)
- Phosphatidylethanolamine Biosynthesis PE(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/24:1(15Z)) (PathBank: SMP0015837)
- Phosphatidylethanolamine Biosynthesis PE(24:0/14:0) (PathBank: SMP0015838)
- Phosphatidylethanolamine Biosynthesis PE(24:0/14:1(9Z)) (PathBank: SMP0015839)
- Phosphatidylethanolamine Biosynthesis PE(24:0/15:0) (PathBank: SMP0015840)
- Phosphatidylethanolamine Biosynthesis PE(24:0/16:0) (PathBank: SMP0015841)
- Phosphatidylethanolamine Biosynthesis PE(24:0/16:1(9Z)) (PathBank: SMP0015842)
- Phosphatidylethanolamine Biosynthesis PE(24:0/18:0) (PathBank: SMP0015843)
- Phosphatidylethanolamine Biosynthesis PE(24:0/18:1(11Z)) (PathBank: SMP0015844)
- Phosphatidylethanolamine Biosynthesis PE(24:0/18:1(9Z)) (PathBank: SMP0015845)
- Phosphatidylethanolamine Biosynthesis PE(24:0/18:2(9Z,12Z)) (PathBank: SMP0015846)
- Phosphatidylethanolamine Biosynthesis PE(24:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0015847)
- Phosphatidylethanolamine Biosynthesis PE(24:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0015848)
- Phosphatidylethanolamine Biosynthesis PE(24:0/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015849)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:0) (PathBank: SMP0015850)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:1(11Z)) (PathBank: SMP0015851)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:2(11Z,14Z)) (PathBank: SMP0015852)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0015853)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0015854)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015855)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015856)
- Phosphatidylethanolamine Biosynthesis PE(24:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015857)
- Phosphatidylethanolamine Biosynthesis PE(24:0/22:0) (PathBank: SMP0015858)
- Phosphatidylethanolamine Biosynthesis PE(24:0/22:1(13Z)) (PathBank: SMP0015859)
- Phosphatidylethanolamine Biosynthesis PE(24:0/22:2(13Z,16Z)) (PathBank: SMP0015860)
- Phosphatidylethanolamine Biosynthesis PE(24:0/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015861)
- Phosphatidylethanolamine Biosynthesis PE(24:0/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015862)
- Phosphatidylethanolamine Biosynthesis PE(24:0/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015863)
- Phosphatidylethanolamine Biosynthesis PE(24:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015864)
- Phosphatidylethanolamine Biosynthesis PE(24:0/24:0) (PathBank: SMP0015865)
- Phosphatidylethanolamine Biosynthesis PE(24:0/24:1(15Z)) (PathBank: SMP0015866)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/14:0) (PathBank: SMP0015867)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/14:1(9Z)) (PathBank: SMP0015868)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/15:0) (PathBank: SMP0015869)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/16:0) (PathBank: SMP0015870)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/16:1(9Z)) (PathBank: SMP0015871)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/18:0) (PathBank: SMP0015872)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/18:1(11Z)) (PathBank: SMP0015873)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/18:1(9Z)) (PathBank: SMP0015874)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/18:2(9Z,12Z)) (PathBank: SMP0015875)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0015876)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0015877)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/18:4(6Z,9Z,12Z,15Z)) (PathBank: SMP0015878)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:0) (PathBank: SMP0015879)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:1(11Z)) (PathBank: SMP0015880)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:2(11Z,14Z)) (PathBank: SMP0015881)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0015882)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0015883)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0015884)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0015885)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0015886)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/22:0) (PathBank: SMP0015887)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/22:1(13Z)) (PathBank: SMP0015888)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/22:2(13Z,16Z)) (PathBank: SMP0015889)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/22:4(7Z,10Z,13Z,16Z)) (PathBank: SMP0015890)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/22:5(4Z,7Z,10Z,13Z,16Z)) (PathBank: SMP0015891)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/22:5(7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015892)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) (PathBank: SMP0015893)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/24:0) (PathBank: SMP0015894)
- Phosphatidylethanolamine Biosynthesis PE(24:1(15Z)/24:1(15Z)) (PathBank: SMP0015895)
- Phosphatidylcholine Biosynthesis PC(18:1(9Z)/24:1(15Z)) (PathBank: SMP0030234)
- Phosphatidylethanolamine Biosynthesis PE(16:1(9Z)/18:0) (PathBank: SMP0030235)
- Phosphatidylcholine Biosynthesis PC(11D3/11D3) (PathBank: SMP0030241)
- Phosphatidylcholine Biosynthesis PC(11D3/11D5) (PathBank: SMP0030242)
- Phosphatidylcholine Biosynthesis PC(11D3/13D5) (PathBank: SMP0030243)
- Phosphatidylcholine Biosynthesis PC(11D3/9D3) (PathBank: SMP0030244)
- Phosphatidylcholine Biosynthesis PC(11D3/9D5) (PathBank: SMP0030245)
- Phosphatidylcholine Biosynthesis PC(11D3/11M3) (PathBank: SMP0030246)
- Phosphatidylcholine Biosynthesis PC(11D3/11M5) (PathBank: SMP0030247)
- Phosphatidylcholine Biosynthesis PC(11D3/13M5) (PathBank: SMP0030248)
- Phosphatidylcholine Biosynthesis PC(11D3/9M5) (PathBank: SMP0030249)
- Phosphatidylcholine Biosynthesis PC(11D5/11D3) (PathBank: SMP0030250)
- Phosphatidylcholine Biosynthesis PC(11D5/11D5) (PathBank: SMP0030251)
- Phosphatidylcholine Biosynthesis PC(11D5/13D5) (PathBank: SMP0030252)
- Phosphatidylcholine Biosynthesis PC(11D5/9D3) (PathBank: SMP0030253)
- Phosphatidylcholine Biosynthesis PC(11D5/9D5) (PathBank: SMP0030254)
- Phosphatidylcholine Biosynthesis PC(11D5/11M3) (PathBank: SMP0030255)
- Phosphatidylcholine Biosynthesis PC(11D5/11M5) (PathBank: SMP0030256)
- Phosphatidylcholine Biosynthesis PC(11D5/13M5) (PathBank: SMP0030257)
- Phosphatidylcholine Biosynthesis PC(11D5/9M5) (PathBank: SMP0030258)
- Phosphatidylcholine Biosynthesis PC(13D5/11D3) (PathBank: SMP0030259)
- Phosphatidylcholine Biosynthesis PC(13D5/11D5) (PathBank: SMP0030260)
- Phosphatidylcholine Biosynthesis PC(13D5/13D5) (PathBank: SMP0030261)
- Phosphatidylcholine Biosynthesis PC(13D5/9D3) (PathBank: SMP0030262)
- Phosphatidylcholine Biosynthesis PC(13D5/9D5) (PathBank: SMP0030263)
- Phosphatidylcholine Biosynthesis PC(13D5/11M3) (PathBank: SMP0030264)
- Phosphatidylcholine Biosynthesis PC(13D5/11M5) (PathBank: SMP0030265)
- Phosphatidylcholine Biosynthesis PC(13D5/13M5) (PathBank: SMP0030266)
- Phosphatidylcholine Biosynthesis PC(13D5/9M5) (PathBank: SMP0030267)
- Phosphatidylcholine Biosynthesis PC(9D3/11D3) (PathBank: SMP0030268)
- Phosphatidylcholine Biosynthesis PC(9D3/11D5) (PathBank: SMP0030269)
- Phosphatidylcholine Biosynthesis PC(9D3/13D5) (PathBank: SMP0030270)
- Phosphatidylcholine Biosynthesis PC(9D3/9D3) (PathBank: SMP0030271)
- Phosphatidylcholine Biosynthesis PC(9D3/9D5) (PathBank: SMP0030272)
- Phosphatidylcholine Biosynthesis PC(9D3/11M3) (PathBank: SMP0030273)
- Phosphatidylcholine Biosynthesis PC(9D3/11M5) (PathBank: SMP0030274)
- Phosphatidylcholine Biosynthesis PC(9D3/13M5) (PathBank: SMP0030275)
- Phosphatidylcholine Biosynthesis PC(9D3/9M5) (PathBank: SMP0030276)
- Phosphatidylcholine Biosynthesis PC(9D5/11D3) (PathBank: SMP0030277)
- Phosphatidylcholine Biosynthesis PC(9D5/11D5) (PathBank: SMP0030278)
- Phosphatidylcholine Biosynthesis PC(9D5/13D5) (PathBank: SMP0030279)
- Phosphatidylcholine Biosynthesis PC(9D5/9D3) (PathBank: SMP0030280)
- Phosphatidylcholine Biosynthesis PC(9D5/9D5) (PathBank: SMP0030281)
- Phosphatidylcholine Biosynthesis PC(9D5/11M3) (PathBank: SMP0030282)
- Phosphatidylcholine Biosynthesis PC(9D5/11M5) (PathBank: SMP0030283)
- Phosphatidylcholine Biosynthesis PC(9D5/13M5) (PathBank: SMP0030284)
- Phosphatidylcholine Biosynthesis PC(9D5/9M5) (PathBank: SMP0030285)
- Phosphatidylcholine Biosynthesis PC(11M3/11D3) (PathBank: SMP0030286)
- Phosphatidylcholine Biosynthesis PC(11M3/11D5) (PathBank: SMP0030287)
- Phosphatidylcholine Biosynthesis PC(11M3/13D5) (PathBank: SMP0030288)
- Phosphatidylcholine Biosynthesis PC(11M3/9D3) (PathBank: SMP0030289)
- Phosphatidylcholine Biosynthesis PC(11M3/9D5) (PathBank: SMP0030290)
- Phosphatidylcholine Biosynthesis PC(11M3/11M3) (PathBank: SMP0030291)
- Phosphatidylcholine Biosynthesis PC(11M3/11M5) (PathBank: SMP0030292)
- Phosphatidylcholine Biosynthesis PC(11M3/13M5) (PathBank: SMP0030293)
- Phosphatidylcholine Biosynthesis PC(11M3/9M5) (PathBank: SMP0030294)
- Phosphatidylcholine Biosynthesis PC(11M5/11D3) (PathBank: SMP0030295)
- Phosphatidylcholine Biosynthesis PC(11M5/11D5) (PathBank: SMP0030296)
- Phosphatidylcholine Biosynthesis PC(11M5/13D5) (PathBank: SMP0030297)
- Phosphatidylcholine Biosynthesis PC(11M5/9D3) (PathBank: SMP0030298)
- Phosphatidylcholine Biosynthesis PC(11M5/9D5) (PathBank: SMP0030299)
- Phosphatidylcholine Biosynthesis PC(11M5/11M3) (PathBank: SMP0030300)
- Phosphatidylcholine Biosynthesis PC(11M5/11M5) (PathBank: SMP0030301)
- Phosphatidylcholine Biosynthesis PC(11M5/13M5) (PathBank: SMP0030302)
- Phosphatidylcholine Biosynthesis PC(11M5/9M5) (PathBank: SMP0030303)
- Phosphatidylcholine Biosynthesis PC(13M5/11D3) (PathBank: SMP0030304)
- Phosphatidylcholine Biosynthesis PC(13M5/11D5) (PathBank: SMP0030305)
- Phosphatidylcholine Biosynthesis PC(13M5/13D5) (PathBank: SMP0030306)
- Phosphatidylcholine Biosynthesis PC(13M5/9D3) (PathBank: SMP0030307)
- Phosphatidylcholine Biosynthesis PC(13M5/9D5) (PathBank: SMP0030308)
- Phosphatidylcholine Biosynthesis PC(13M5/11M3) (PathBank: SMP0030309)
- Phosphatidylcholine Biosynthesis PC(13M5/11M5) (PathBank: SMP0030310)
- Phosphatidylcholine Biosynthesis PC(13M5/13M5) (PathBank: SMP0030311)
- Phosphatidylcholine Biosynthesis PC(13M5/9M5) (PathBank: SMP0030312)
- Phosphatidylcholine Biosynthesis PC(9M5/11D3) (PathBank: SMP0030313)
- Phosphatidylcholine Biosynthesis PC(9M5/11D5) (PathBank: SMP0030314)
- Phosphatidylcholine Biosynthesis PC(9M5/13D5) (PathBank: SMP0030315)
- Phosphatidylcholine Biosynthesis PC(9M5/9D3) (PathBank: SMP0030316)
- Phosphatidylcholine Biosynthesis PC(9M5/9D5) (PathBank: SMP0030317)
- Phosphatidylcholine Biosynthesis PC(9M5/11M3) (PathBank: SMP0030318)
- Phosphatidylcholine Biosynthesis PC(9M5/11M5) (PathBank: SMP0030319)
- Phosphatidylcholine Biosynthesis PC(9M5/13M5) (PathBank: SMP0030320)
- Phosphatidylcholine Biosynthesis PC(9M5/9M5) (PathBank: SMP0030321)
- Phosphatidylethanolamine Biosynthesis PE(11D3/11D3) (PathBank: SMP0030322)
- Phosphatidylethanolamine Biosynthesis PE(11D3/11D5) (PathBank: SMP0030323)
- Phosphatidylethanolamine Biosynthesis PE(11D3/13D5) (PathBank: SMP0030324)
- Phosphatidylethanolamine Biosynthesis PE(11D3/9D3) (PathBank: SMP0030325)
- Phosphatidylethanolamine Biosynthesis PE(11D3/9D5) (PathBank: SMP0030326)
- Phosphatidylethanolamine Biosynthesis PE(11D3/11M3) (PathBank: SMP0030327)
- Phosphatidylethanolamine Biosynthesis PE(11D3/11M5) (PathBank: SMP0030328)
- Phosphatidylethanolamine Biosynthesis PE(11D3/13M5) (PathBank: SMP0030329)
- Phosphatidylethanolamine Biosynthesis PE(11D3/9M5) (PathBank: SMP0030330)
- Phosphatidylethanolamine Biosynthesis PE(11D5/11D3) (PathBank: SMP0030331)
- Phosphatidylethanolamine Biosynthesis PE(11D5/11D5) (PathBank: SMP0030332)
- Phosphatidylethanolamine Biosynthesis PE(11D5/13D5) (PathBank: SMP0030333)
- Phosphatidylethanolamine Biosynthesis PE(11D5/9D3) (PathBank: SMP0030334)
- Phosphatidylethanolamine Biosynthesis PE(11D5/9D5) (PathBank: SMP0030335)
- Phosphatidylethanolamine Biosynthesis PE(11D5/11M3) (PathBank: SMP0030336)
- Phosphatidylethanolamine Biosynthesis PE(11D5/11M5) (PathBank: SMP0030337)
- Phosphatidylethanolamine Biosynthesis PE(11D5/13M5) (PathBank: SMP0030338)
- Phosphatidylethanolamine Biosynthesis PE(11D5/9M5) (PathBank: SMP0030339)
- Phosphatidylethanolamine Biosynthesis PE(13D5/11D3) (PathBank: SMP0030340)
- Phosphatidylethanolamine Biosynthesis PE(13D5/11D5) (PathBank: SMP0030341)
- Phosphatidylethanolamine Biosynthesis PE(13D5/13D5) (PathBank: SMP0030342)
- Phosphatidylethanolamine Biosynthesis PE(13D5/9D3) (PathBank: SMP0030343)
- Phosphatidylethanolamine Biosynthesis PE(13D5/9D5) (PathBank: SMP0030344)
- Phosphatidylethanolamine Biosynthesis PE(13D5/11M3) (PathBank: SMP0030345)
- Phosphatidylethanolamine Biosynthesis PE(13D5/11M5) (PathBank: SMP0030346)
- Phosphatidylethanolamine Biosynthesis PE(13D5/13M5) (PathBank: SMP0030347)
- Phosphatidylethanolamine Biosynthesis PE(13D5/9M5) (PathBank: SMP0030348)
- Phosphatidylethanolamine Biosynthesis PE(9D3/11D3) (PathBank: SMP0030349)
- Phosphatidylethanolamine Biosynthesis PE(9D3/11D5) (PathBank: SMP0030350)
- Phosphatidylethanolamine Biosynthesis PE(9D3/13D5) (PathBank: SMP0030351)
- Phosphatidylethanolamine Biosynthesis PE(9D3/9D3) (PathBank: SMP0030352)
- Phosphatidylethanolamine Biosynthesis PE(9D3/9D5) (PathBank: SMP0030353)
- Phosphatidylethanolamine Biosynthesis PE(9D3/11M3) (PathBank: SMP0030354)
- Phosphatidylethanolamine Biosynthesis PE(9D3/11M5) (PathBank: SMP0030355)
- Phosphatidylethanolamine Biosynthesis PE(9D3/13M5) (PathBank: SMP0030356)
- Phosphatidylethanolamine Biosynthesis PE(9D3/9M5) (PathBank: SMP0030357)
- Phosphatidylethanolamine Biosynthesis PE(9D5/11D3) (PathBank: SMP0030358)
- Phosphatidylethanolamine Biosynthesis PE(9D5/11D5) (PathBank: SMP0030359)
- Phosphatidylethanolamine Biosynthesis PE(9D5/13D5) (PathBank: SMP0030360)
- Phosphatidylethanolamine Biosynthesis PE(9D5/9D3) (PathBank: SMP0030361)
- Phosphatidylethanolamine Biosynthesis PE(9D5/9D5) (PathBank: SMP0030362)
- Phosphatidylethanolamine Biosynthesis PE(9D5/11M3) (PathBank: SMP0030363)
- Phosphatidylethanolamine Biosynthesis PE(9D5/11M5) (PathBank: SMP0030364)
- Phosphatidylethanolamine Biosynthesis PE(9D5/13M5) (PathBank: SMP0030365)
- Phosphatidylethanolamine Biosynthesis PE(9D5/9M5) (PathBank: SMP0030366)
- Phosphatidylethanolamine Biosynthesis PE(11M3/11D3) (PathBank: SMP0030367)
- Phosphatidylethanolamine Biosynthesis PE(11M3/11D5) (PathBank: SMP0030368)
- Phosphatidylethanolamine Biosynthesis PE(11M3/13D5) (PathBank: SMP0030369)
- Phosphatidylethanolamine Biosynthesis PE(11M3/9D3) (PathBank: SMP0030370)
- Phosphatidylethanolamine Biosynthesis PE(11M3/9D5) (PathBank: SMP0030371)
- Phosphatidylethanolamine Biosynthesis PE(11M3/11M3) (PathBank: SMP0030372)
- Phosphatidylethanolamine Biosynthesis PE(11M3/11M5) (PathBank: SMP0030373)
- Phosphatidylethanolamine Biosynthesis PE(11M3/13M5) (PathBank: SMP0030374)
- Phosphatidylethanolamine Biosynthesis PE(11M3/9M5) (PathBank: SMP0030375)
- Phosphatidylethanolamine Biosynthesis PE(11M5/11D3) (PathBank: SMP0030376)
- Phosphatidylethanolamine Biosynthesis PE(11M5/11D5) (PathBank: SMP0030377)
- Phosphatidylethanolamine Biosynthesis PE(11M5/13D5) (PathBank: SMP0030378)
- Phosphatidylethanolamine Biosynthesis PE(11M5/9D3) (PathBank: SMP0030379)
- Phosphatidylethanolamine Biosynthesis PE(11M5/9D5) (PathBank: SMP0030380)
- Phosphatidylethanolamine Biosynthesis PE(11M5/11M3) (PathBank: SMP0030381)
- Phosphatidylethanolamine Biosynthesis PE(11M5/11M5) (PathBank: SMP0030382)
- Phosphatidylethanolamine Biosynthesis PE(11M5/13M5) (PathBank: SMP0030383)
- Phosphatidylethanolamine Biosynthesis PE(11M5/9M5) (PathBank: SMP0030384)
- Phosphatidylethanolamine Biosynthesis PE(13M5/11D3) (PathBank: SMP0030385)
- Phosphatidylethanolamine Biosynthesis PE(13M5/11D5) (PathBank: SMP0030386)
- Phosphatidylethanolamine Biosynthesis PE(13M5/13D5) (PathBank: SMP0030387)
- Phosphatidylethanolamine Biosynthesis PE(13M5/9D3) (PathBank: SMP0030388)
- Phosphatidylethanolamine Biosynthesis PE(13M5/9D5) (PathBank: SMP0030389)
- Phosphatidylethanolamine Biosynthesis PE(13M5/11M3) (PathBank: SMP0030390)
- Phosphatidylethanolamine Biosynthesis PE(13M5/11M5) (PathBank: SMP0030391)
- Phosphatidylethanolamine Biosynthesis PE(13M5/13M5) (PathBank: SMP0030392)
- Phosphatidylethanolamine Biosynthesis PE(13M5/9M5) (PathBank: SMP0030393)
- Phosphatidylethanolamine Biosynthesis PE(9M5/11D3) (PathBank: SMP0030394)
- Phosphatidylethanolamine Biosynthesis PE(9M5/11D5) (PathBank: SMP0030395)
- Phosphatidylethanolamine Biosynthesis PE(9M5/13D5) (PathBank: SMP0030396)
- Phosphatidylethanolamine Biosynthesis PE(9M5/9D3) (PathBank: SMP0030397)
- Phosphatidylethanolamine Biosynthesis PE(9M5/9D5) (PathBank: SMP0030398)
- Phosphatidylethanolamine Biosynthesis PE(9M5/11M3) (PathBank: SMP0030399)
- Phosphatidylethanolamine Biosynthesis PE(9M5/11M5) (PathBank: SMP0030400)
- Phosphatidylethanolamine Biosynthesis PE(9M5/13M5) (PathBank: SMP0030401)
- Phosphatidylethanolamine Biosynthesis PE(9M5/9M5) (PathBank: SMP0030402)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/14:0) | PE(14:0/14:0) (PathBank: SMP0100121)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/16:0) | PE(14:0/16:0) (PathBank: SMP0100124)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/16:1(9Z)) | PE(14:0/16:1(9Z)) (PathBank: SMP0100125)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/18:0) | PE(14:0/18:0) (PathBank: SMP0100127)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/18:1(9Z)) | PE(14:0/18:1(9Z)) (PathBank: SMP0100128)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/18:1(11Z)) | PE(14:0/18:1(11Z)) (PathBank: SMP0100129)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/18:2(9Z,12Z)) | PE(14:0/18:2(9Z,12Z)) (PathBank: SMP0100130)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/18:3(6Z,9Z,12Z)) | PE(14:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0100131)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/18:3(9Z,12Z,15Z)) | PE(14:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0100132)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/20:0) | PE(14:0/20:0) (PathBank: SMP0100133)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/20:3(5Z,8Z,11Z)) | PE(14:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0100134)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/20:3(8Z,11Z,14Z)) | PE(14:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0100135)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/20:4(5Z,8Z,11Z,14Z)) | PE(14:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100136)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/20:4(8Z,11Z,14Z,17Z)) | PE(14:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100137)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(14:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100138)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/16:0) | PE(16:0/16:0) (PathBank: SMP0100155)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/16:1(9Z)) | PE(16:0/16:1(9Z)) (PathBank: SMP0100156)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/18:0) | PE(16:0/18:0) (PathBank: SMP0100158)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/18:1(9Z)) | PE(16:0/18:1(9Z)) (PathBank: SMP0100159)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/18:1(11Z)) | PE(16:0/18:1(11Z)) (PathBank: SMP0100160)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/18:2(9Z,12Z)) | PE(16:0/18:2(9Z,12Z)) (PathBank: SMP0100161)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/18:3(6Z,9Z,12Z)) | PE(16:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0100162)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/18:3(9Z,12Z,15Z)) | PE(16:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0100163)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/20:0) | PE(16:0/20:0) (PathBank: SMP0100164)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/20:3(5Z,8Z,11Z)) | PE(16:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0100165)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/20:3(8Z,11Z,14Z)) | PE(16:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0100166)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/20:4(5Z,8Z,11Z,14Z)) | PE(16:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100167)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/20:4(8Z,11Z,14Z,17Z)) | PE(16:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100168)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(16:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100169)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/16:1(9Z)) | PE(16:1(9Z)/16:1(9Z)) (PathBank: SMP0100170)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/18:0) | PE(16:1(9Z)/18:0) (PathBank: SMP0100172)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/18:1(9Z)) | PE(16:1(9Z)/18:1(9Z)) (PathBank: SMP0100173)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/18:1(11Z)) | PE(16:1(9Z)/18:1(11Z)) (PathBank: SMP0100174)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/18:2(9Z,12Z)) | PE(16:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0100175)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/18:3(6Z,9Z,12Z)) | PE(16:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0100176)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/18:3(9Z,12Z,15Z)) | PE(16:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0100177)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/20:0) | PE(16:1(9Z)/20:0) (PathBank: SMP0100178)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/20:3(5Z,8Z,11Z)) | PE(16:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0100179)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/20:3(8Z,11Z,14Z)) | PE(16:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100180)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/20:4(5Z,8Z,11Z,14Z)) | PE(16:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100181)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/20:4(8Z,11Z,14Z,17Z)) | PE(16:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100182)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(16:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100183)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/18:0) | PE(18:0/18:0) (PathBank: SMP0100197)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/18:1(9Z)) | PE(18:0/18:1(9Z)) (PathBank: SMP0100198)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/18:1(11Z)) | PE(18:0/18:1(11Z)) (PathBank: SMP0100199)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/18:2(9Z,12Z)) | PE(18:0/18:2(9Z,12Z)) (PathBank: SMP0100200)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/18:3(6Z,9Z,12Z)) | PE(18:0/18:3(6Z,9Z,12Z)) (PathBank: SMP0100201)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/18:3(9Z,12Z,15Z)) | PE(18:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0100202)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/20:0) | PE(18:0/20:0) (PathBank: SMP0100203)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/20:3(5Z,8Z,11Z)) | PE(18:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0100204)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/20:3(8Z,11Z,14Z)) | PE(18:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0100205)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/20:4(5Z,8Z,11Z,14Z)) | PE(18:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100206)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/20:4(8Z,11Z,14Z,17Z)) | PE(18:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100207)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(18:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100208)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/18:1(9Z)) | PE(18:1(9Z)/18:1(9Z)) (PathBank: SMP0100209)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/18:1(11Z)) | PE(18:1(9Z)/18:1(11Z)) (PathBank: SMP0100210)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/18:2(9Z,12Z)) | PE(18:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0100211)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/18:3(6Z,9Z,12Z)) | PE(18:1(9Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0100212)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/18:3(9Z,12Z,15Z)) | PE(18:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0100213)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/20:0) | PE(18:1(9Z)/20:0) (PathBank: SMP0100214)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/20:3(5Z,8Z,11Z)) | PE(18:1(9Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0100215)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/20:3(8Z,11Z,14Z)) | PE(18:1(9Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100216)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/20:4(5Z,8Z,11Z,14Z)) | PE(18:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100217)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/20:4(8Z,11Z,14Z,17Z)) | PE(18:1(9Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100218)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(18:1(9Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100219)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/18:1(11Z)) | PE(18:1(11Z)/18:1(11Z)) (PathBank: SMP0100220)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/18:2(9Z,12Z)) | PE(18:1(11Z)/18:2(9Z,12Z)) (PathBank: SMP0100221)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/18:3(6Z,9Z,12Z)) | PE(18:1(11Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0100222)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/18:3(9Z,12Z,15Z)) | PE(18:1(11Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0100223)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/20:0) | PE(18:1(11Z)/20:0) (PathBank: SMP0100224)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/20:3(5Z,8Z,11Z)) | PE(18:1(11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0100225)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/20:3(8Z,11Z,14Z)) | PE(18:1(11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100226)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/20:4(5Z,8Z,11Z,14Z)) | PE(18:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100227)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/20:4(8Z,11Z,14Z,17Z)) | PE(18:1(11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100228)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(18:1(11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100229)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/18:2(9Z,12Z)) | PE(18:2(9Z,12Z)/18:2(9Z,12Z)) (PathBank: SMP0100230)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/18:3(6Z,9Z,12Z)) | PE(18:2(9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0100231)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/18:3(9Z,12Z,15Z)) | PE(18:2(9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0100232)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/20:0) | PE(18:2(9Z,12Z)/20:0) (PathBank: SMP0100233)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/20:3(5Z,8Z,11Z)) | PE(18:2(9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0100234)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/20:3(8Z,11Z,14Z)) | PE(18:2(9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100235)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) | PE(18:2(9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100236)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) | PE(18:2(9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100237)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(18:2(9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100238)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:3(6Z,9Z,12Z)) | PE(18:3(6Z,9Z,12Z)/18:3(6Z,9Z,12Z)) (PathBank: SMP0100239)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/18:3(9Z,12Z,15Z)) | PE(18:3(6Z,9Z,12Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0100240)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:0) | PE(18:3(6Z,9Z,12Z)/20:0) (PathBank: SMP0100241)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:3(5Z,8Z,11Z)) | PE(18:3(6Z,9Z,12Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0100242)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:3(8Z,11Z,14Z)) | PE(18:3(6Z,9Z,12Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100243)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) | PE(18:3(6Z,9Z,12Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100244)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) | PE(18:3(6Z,9Z,12Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100245)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(6Z,9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(18:3(6Z,9Z,12Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100246)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/18:3(9Z,12Z,15Z)) | PE(18:3(9Z,12Z,15Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0100247)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:0) | PE(18:3(9Z,12Z,15Z)/20:0) (PathBank: SMP0100248)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:3(5Z,8Z,11Z)) | PE(18:3(9Z,12Z,15Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0100249)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:3(8Z,11Z,14Z)) | PE(18:3(9Z,12Z,15Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100250)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:4(5Z,8Z,11Z,14Z)) | PE(18:3(9Z,12Z,15Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100251)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:4(8Z,11Z,14Z,17Z)) | PE(18:3(9Z,12Z,15Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100252)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(18:3(9Z,12Z,15Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100253)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:0/20:0) | PE(20:0/20:0) (PathBank: SMP0100254)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:0/20:3(5Z,8Z,11Z)) | PE(20:0/20:3(5Z,8Z,11Z)) (PathBank: SMP0100255)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:0/20:3(8Z,11Z,14Z)) | PE(20:0/20:3(8Z,11Z,14Z)) (PathBank: SMP0100256)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:0/20:4(5Z,8Z,11Z,14Z)) | PE(20:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100257)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:0/20:4(8Z,11Z,14Z,17Z)) | PE(20:0/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100258)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:0/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(20:0/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100259)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:3(5Z,8Z,11Z)) | PE(20:3(5Z,8Z,11Z)/20:3(5Z,8Z,11Z)) (PathBank: SMP0100260)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:3(8Z,11Z,14Z)) | PE(20:3(5Z,8Z,11Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100261)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:4(5Z,8Z,11Z,14Z)) | PE(20:3(5Z,8Z,11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100262)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:4(8Z,11Z,14Z,17Z)) | PE(20:3(5Z,8Z,11Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100263)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(5Z,8Z,11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(20:3(5Z,8Z,11Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100264)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:3(8Z,11Z,14Z)) | PE(20:3(8Z,11Z,14Z)/20:3(8Z,11Z,14Z)) (PathBank: SMP0100265)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) | PE(20:3(8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100266)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) | PE(20:3(8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100267)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:3(8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(20:3(8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100268)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) | PE(20:4(5Z,8Z,11Z,14Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100269)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) | PE(20:4(5Z,8Z,11Z,14Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100270)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(20:4(5Z,8Z,11Z,14Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100271)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:4(8Z,11Z,14Z,17Z)) | PE(20:4(8Z,11Z,14Z,17Z)/20:4(8Z,11Z,14Z,17Z)) (PathBank: SMP0100272)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:4(8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(20:4(8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100273)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:5(5Z,8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) | PE(20:5(5Z,8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)) (PathBank: SMP0100274)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/14:1(9Z)) | PE(14:0/14:1(9Z)) (PathBank: SMP0100279)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/15:0) | PE(14:0/15:0) (PathBank: SMP0100280)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/20:1(11Z)) | PE(14:0/20:1(11Z)) (PathBank: SMP0100287)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:0/22:0) | PE(14:0/22:0) (PathBank: SMP0100289)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/14:1(9Z)) | PE(14:1(9Z)/14:1(9Z)) (PathBank: SMP0100290)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/15:0) | PE(14:1(9Z)/15:0) (PathBank: SMP0100291)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/16:0) | PE(14:1(9Z)/16:0) (PathBank: SMP0100292)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/18:0) | PE(14:1(9Z)/18:0) (PathBank: SMP0100293)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/18:1(9Z)) | PE(14:1(9Z)/18:1(9Z)) (PathBank: SMP0100294)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/18:1(11Z)) | PE(14:1(9Z)/18:1(11Z)) (PathBank: SMP0100295)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/18:2(9Z,12Z)) | PE(14:1(9Z)/18:2(9Z,12Z)) (PathBank: SMP0100296)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/18:3(9Z,12Z,15Z)) | PE(14:1(9Z)/18:3(9Z,12Z,15Z)) (PathBank: SMP0100297)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/20:1(11Z)) | PE(14:1(9Z)/20:1(11Z)) (PathBank: SMP0100298)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/20:4(5Z,8Z,11Z,14Z)) | PE(14:1(9Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100299)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(14:1(9Z)/22:0) | PE(14:1(9Z)/22:0) (PathBank: SMP0100300)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/15:0) | PE(15:0/15:0) (PathBank: SMP0100301)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/16:0) | PE(15:0/16:0) (PathBank: SMP0100302)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/18:0) | PE(15:0/18:0) (PathBank: SMP0100303)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/18:1(9Z)) | PE(15:0/18:1(9Z)) (PathBank: SMP0100304)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/18:1(11Z)) | PE(15:0/18:1(11Z)) (PathBank: SMP0100305)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/18:2(9Z,12Z)) | PE(15:0/18:2(9Z,12Z)) (PathBank: SMP0100306)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/18:3(9Z,12Z,15Z)) | PE(15:0/18:3(9Z,12Z,15Z)) (PathBank: SMP0100307)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/20:1(11Z)) | PE(15:0/20:1(11Z)) (PathBank: SMP0100308)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/20:4(5Z,8Z,11Z,14Z)) | PE(15:0/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100309)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(15:0/22:0) | PE(15:0/22:0) (PathBank: SMP0100310)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/20:1(11Z)) | PE(16:0/20:1(11Z)) (PathBank: SMP0100317)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(16:0/22:0) | PE(16:0/22:0) (PathBank: SMP0100319)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/20:1(11Z)) | PE(18:0/20:1(11Z)) (PathBank: SMP0100325)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:0/22:0) | PE(18:0/22:0) (PathBank: SMP0100327)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/20:1(11Z)) | PE(18:1(9Z)/20:1(11Z)) (PathBank: SMP0100332)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(9Z)/22:0) | PE(18:1(9Z)/22:0) (PathBank: SMP0100334)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/20:1(11Z)) | PE(18:1(11Z)/20:1(11Z)) (PathBank: SMP0100338)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:1(11Z)/22:0) | PE(18:1(11Z)/22:0) (PathBank: SMP0100340)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/20:1(11Z)) | PE(18:2(9Z,12Z)/20:1(11Z)) (PathBank: SMP0100343)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:2(9Z,12Z)/22:0) | PE(18:2(9Z,12Z)/22:0) (PathBank: SMP0100345)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/20:1(11Z)) | PE(18:3(9Z,12Z,15Z)/20:1(11Z)) (PathBank: SMP0100347)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(18:3(9Z,12Z,15Z)/22:0) | PE(18:3(9Z,12Z,15Z)/22:0) (PathBank: SMP0100349)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:1(11Z)/20:1(11Z)) | PE(20:1(11Z)/20:1(11Z)) (PathBank: SMP0100350)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:1(11Z)/20:4(5Z,8Z,11Z,14Z)) | PE(20:1(11Z)/20:4(5Z,8Z,11Z,14Z)) (PathBank: SMP0100351)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:1(11Z)/22:0) | PE(20:1(11Z)/22:0) (PathBank: SMP0100352)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(20:4(5Z,8Z,11Z,14Z)/22:0) | PE(20:4(5Z,8Z,11Z,14Z)/22:0) (PathBank: SMP0100354)
- Phosphatidylcholine/Phosphatidylethanolamine Biosynthesis PC(22:0/22:0) | PE(22:0/22:0) (PathBank: SMP0100355)
Signaling pathwayDisease pathwayDrug action pathway Biochemical processCellular process |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatized |
|---|