| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2009-07-13 12:46:54 UTC |
|---|
| Update Date | 2022-03-07 02:51:26 UTC |
|---|
| HMDB ID | HMDB0012454 |
|---|
| Secondary Accession Numbers | - HMDB0062211
- HMDB12454
- HMDB62211
|
|---|
| Metabolite Identification |
|---|
| Common Name | 3beta,7alpha-Dihydroxy-5-cholestenoate |
|---|
| Description | 3beta,7alpha-Dihydroxy-5-cholestenoate, also known as 3b,7a-dihydroxy-5-cholesten-26-Oic acid, belongs to the class of organic compounds known as dihydroxy bile acids, alcohols and derivatives. Dihydroxy bile acids, alcohols and derivatives are compounds containing or derived from a bile acid or alcohol, and which bears exactly two carboxylic acid groups. 3beta,7alpha-Dihydroxy-5-cholestenoate is an extremely weak basic (essentially neutral) compound (based on its pKa). 3beta,7alpha-Dihydroxy-5-cholestenoate is a potentially toxic compound. |
|---|
| Structure | [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCCC(C)C(O)=O InChI=1S/C27H44O4/c1-16(6-5-7-17(2)25(30)31)20-8-9-21-24-22(11-13-27(20,21)4)26(3)12-10-19(28)14-18(26)15-23(24)29/h15-17,19-24,28-29H,5-14H2,1-4H3,(H,30,31)/t16-,17?,19+,20-,21+,22+,23-,24+,26+,27-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 3beta,7alpha-Dihydroxy-5-cholesten-26-Oic acid | Kegg | | 3b,7a-Dihydroxy-5-cholesten-26-Oate | Generator | | 3b,7a-Dihydroxy-5-cholesten-26-Oic acid | Generator | | 3beta,7alpha-Dihydroxy-5-cholesten-26-Oate | Generator | | 3Β,7α-dihydroxy-5-cholesten-26-Oate | Generator | | 3Β,7α-dihydroxy-5-cholesten-26-Oic acid | Generator | | 3b,7a-Dihydroxy-5-cholestenoate | Generator | | 3b,7a-Dihydroxy-5-cholestenoic acid | Generator | | 3beta,7alpha-Dihydroxy-5-cholestenoic acid | Generator | | 3Β,7α-dihydroxy-5-cholestenoate | Generator | | 3Β,7α-dihydroxy-5-cholestenoic acid | Generator | | 3,7-Dihydroxy-5-cholestenoic acid | HMDB | | 3 beta,7 alpha-Dihydroxy-5-cholesten-26-Oic acid | HMDB |
|
|---|
| Chemical Formula | C27H44O4 |
|---|
| Average Molecular Weight | 432.645 |
|---|
| Monoisotopic Molecular Weight | 432.323959897 |
|---|
| IUPAC Name | (6R)-6-[(1S,2R,5S,9S,10S,11S,14R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-7-en-14-yl]-2-methylheptanoic acid |
|---|
| Traditional Name | (6R)-6-[(1S,2R,5S,9S,10S,11S,14R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-7-en-14-yl]-2-methylheptanoic acid |
|---|
| CAS Registry Number | 115538-84-6 |
|---|
| SMILES | [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCCC(C)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C27H44O4/c1-16(6-5-7-17(2)25(30)31)20-8-9-21-24-22(11-13-27(20,21)4)26(3)12-10-19(28)14-18(26)15-23(24)29/h15-17,19-24,28-29H,5-14H2,1-4H3,(H,30,31)/t16-,17?,19+,20-,21+,22+,23-,24+,26+,27-/m1/s1 |
|---|
| InChI Key | GYJSAWZGYQXRBS-GRJZKGIBSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as dihydroxy bile acids, alcohols and derivatives. Dihydroxy bile acids, alcohols and derivatives are compounds containing or derived from a bile acid or alcohol, and which bears exactly two carboxylic acid groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Bile acids, alcohols and derivatives |
|---|
| Direct Parent | Dihydroxy bile acids, alcohols and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Dihydroxy bile acid, alcohol, or derivatives
- Steroid acid
- 3-hydroxy-delta-5-steroid
- 3-hydroxysteroid
- 7-hydroxysteroid
- 3-beta-hydroxysteroid
- 3-beta-hydroxy-delta-5-steroid
- Hydroxysteroid
- Delta-5-steroid
- Medium-chain fatty acid
- Hydroxy fatty acid
- Fatty acyl
- Cyclic alcohol
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | | Show more...
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 3beta,7alpha-Dihydroxy-5-cholestenoate,1TMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O | 3651.5 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,1TMS,isomer #2 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O | 3664.9 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,1TMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C | 3574.1 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,2TMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O | 3557.4 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,2TMS,isomer #2 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C | 3556.9 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,2TMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C | 3584.3 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,3TMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C)C=C4C[C@@H](O[Si](C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C | 3488.5 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,1TBDMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O | 3875.2 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,1TBDMS,isomer #2 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O | 3870.9 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,1TBDMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C(C)(C)C | 3845.9 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,2TBDMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O | 3986.8 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,2TBDMS,isomer #2 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C(C)(C)C | 4043.4 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,2TBDMS,isomer #3 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C(C)(C)C | 4053.2 | Semi standard non polar | 33892256 | | 3beta,7alpha-Dihydroxy-5-cholestenoate,3TBDMS,isomer #1 | CC(CCC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O[Si](C)(C)C(C)(C)C)C=C4C[C@@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)[C@H]3CC[C@@]21C)C(=O)O[Si](C)(C)C(C)(C)C | 4181.9 | Semi standard non polar | 33892256 |
| Show more...
|---|
| Spectra |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate GC-MS (Non-derivatized) - 70eV, Positive | splash10-0v4r-0247900000-6844e920e0f45971085c | 2017-09-20 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate GC-MS (3 TMS) - 70eV, Positive | splash10-001i-1110149000-9481ac260199d7f5b542 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 10V, Positive-QTOF | splash10-014j-0007900000-ba51b1e65739e23c2f83 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 20V, Positive-QTOF | splash10-014j-0009200000-ad2af6637f0b3ce7d674 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 40V, Positive-QTOF | splash10-014i-2119000000-87d5606a65d11c0d0bc2 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 10V, Negative-QTOF | splash10-001i-0001900000-11d955c76d3b48e69882 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 20V, Negative-QTOF | splash10-02ar-0007900000-1344438f254eaafa390d | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 40V, Negative-QTOF | splash10-0600-6009200000-7340abd91104a8c2fb9b | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 10V, Positive-QTOF | splash10-00ls-1106900000-2985a04589bff796de8f | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 20V, Positive-QTOF | splash10-01ba-2159100000-99496085551528fe3dce | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 40V, Positive-QTOF | splash10-052s-7690000000-313265b61c2dcc7ed040 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 10V, Negative-QTOF | splash10-001i-0000900000-3cca5453e09966b932ed | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 20V, Negative-QTOF | splash10-001i-0002900000-acd26e8db7d05ca4d858 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3beta,7alpha-Dihydroxy-5-cholestenoate 40V, Negative-QTOF | splash10-004i-1002900000-97654ceb214ce880e583 | 2021-09-22 | Wishart Lab | View Spectrum |
| Show more...
|---|
| Biological Properties |
|---|
| Cellular Locations | |
|---|
| Biospecimen Locations | |
|---|
| Tissue Locations | Not Available |
|---|
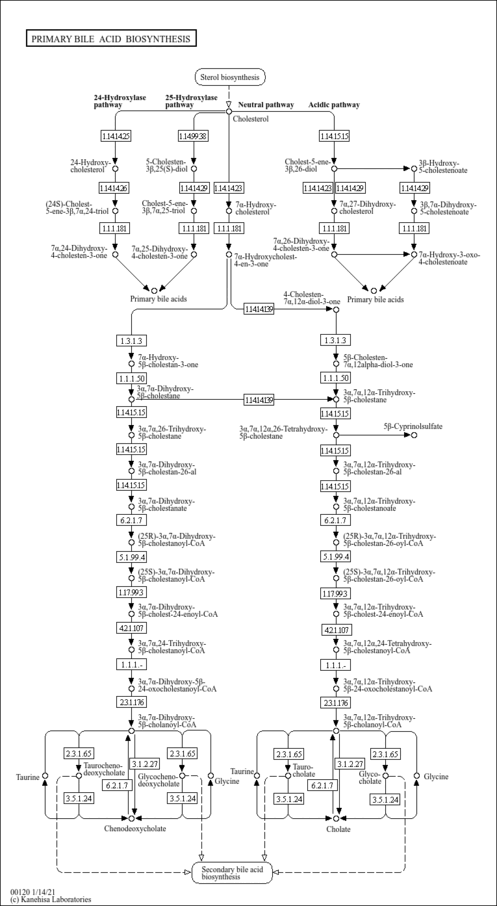
| Pathways | |
|---|
| Normal Concentrations |
|---|
| |
| Blood | Detected and Quantified | <0.1 uM | Infant (0-1 year old) | Not Specified | Normal | | details |
|
|---|
| Abnormal Concentrations |
|---|
| |
| Blood | Detected and Quantified | 25.4 uM | Infant (0-1 year old) | Not Specified | D-Bifunctional Protein Deficiency | | details | | Blood | Detected and Quantified | 0.005 uM | Newborn (0-30 days old) | Female | Rhizomelic chondrodysplasia punctata | | details | | Blood | Detected and Quantified | 0.319 uM | Infant (0-1 year old) | Male | Peroxisomal biogenesis disorder | | details | | Blood | Detected and Quantified | 5.71 uM | Newborn (0-30 days old) | Not Specified | Zellweger syndrome | | details | | Blood | Detected and Quantified | 0-4.500 uM | Children (1-13 years old) | Female | Peroxisomal biogenesis disorder | | details | | Blood | Detected and Quantified | 0.0360-7.0300 uM | Newborn (0-30 days old) | Female | Peroxisomal biogenesis disorder | | details |
|
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | | Rhizomelic chondrodysplasia punctata |
|---|
- Baumgartner MR, Poll-The BT, Verhoeven NM, Jakobs C, Espeel M, Roels F, Rabier D, Levade T, Rolland MO, Martinez M, Wanders RJ, Saudubray JM: Clinical approach to inherited peroxisomal disorders: a series of 27 patients. Ann Neurol. 1998 Nov;44(5):720-30. [PubMed:9818927 ]
| | Peroxisomal biogenesis defect |
|---|
- Baumgartner MR, Poll-The BT, Verhoeven NM, Jakobs C, Espeel M, Roels F, Rabier D, Levade T, Rolland MO, Martinez M, Wanders RJ, Saudubray JM: Clinical approach to inherited peroxisomal disorders: a series of 27 patients. Ann Neurol. 1998 Nov;44(5):720-30. [PubMed:9818927 ]
| | D-Bifunctional protein deficiency |
|---|
- Rizzo C, Boenzi S, Wanders RJ, Duran M, Caruso U, Dionisi-Vici C: Characteristic acylcarnitine profiles in inherited defects of peroxisome biogenesis: a novel tool for screening diagnosis using tandem mass spectrometry. Pediatr Res. 2003 Jun;53(6):1013-8. doi: 10.1203/01.PDR.0000064902.59052.0F. Epub 2003 Mar 19. [PubMed:12646728 ]
|
|
|---|
| Associated OMIM IDs | - 215100 (Rhizomelic chondrodysplasia punctata)
- 214100 (Peroxisomal biogenesis defect)
- 261515 (D-Bifunctional protein deficiency)
|
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| KEGG Compound ID | C17335 |
|---|
| BioCyc ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PubChem Compound | 3081084 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 81015 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | Not Available |
|---|
| MarkerDB ID | Not Available |
|---|
| Good Scents ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| Material Safety Data Sheet (MSDS) | Not Available |
|---|
| General References | - Simons K, Toomre D: Lipid rafts and signal transduction. Nat Rev Mol Cell Biol. 2000 Oct;1(1):31-9. [PubMed:11413487 ]
- Watson AD: Thematic review series: systems biology approaches to metabolic and cardiovascular disorders. Lipidomics: a global approach to lipid analysis in biological systems. J Lipid Res. 2006 Oct;47(10):2101-11. Epub 2006 Aug 10. [PubMed:16902246 ]
- Sethi JK, Vidal-Puig AJ: Thematic review series: adipocyte biology. Adipose tissue function and plasticity orchestrate nutritional adaptation. J Lipid Res. 2007 Jun;48(6):1253-62. Epub 2007 Mar 20. [PubMed:17374880 ]
- Lingwood D, Simons K: Lipid rafts as a membrane-organizing principle. Science. 2010 Jan 1;327(5961):46-50. doi: 10.1126/science.1174621. [PubMed:20044567 ]
- Gunstone, Frank D., John L. Harwood, and Albert J. Dijkstra (2007). The lipid handbook with CD-ROM. CRC Press.
|
|---|