| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2009-07-13 13:21:08 UTC |
|---|
| Update Date | 2022-03-07 02:51:26 UTC |
|---|
| HMDB ID | HMDB0012456 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA |
|---|
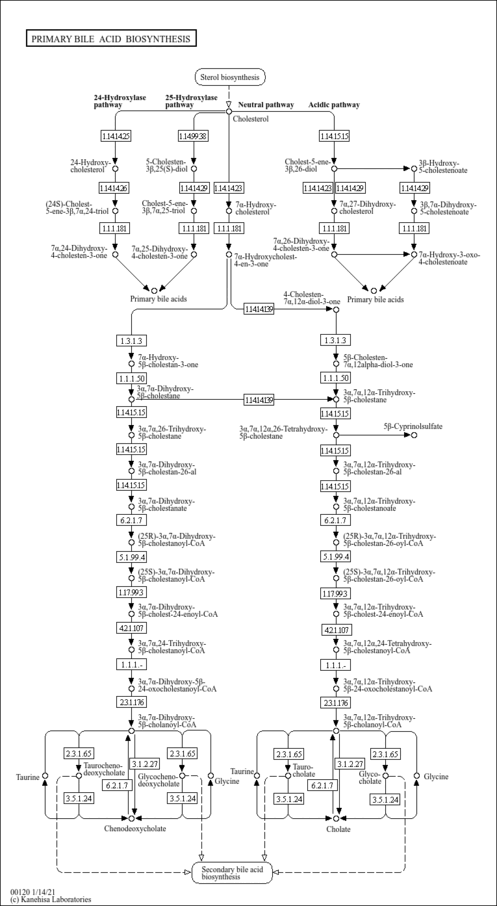
| Description | 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA is found in the primary bile acid biosynthesis pathway. 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA is created from 3 alpha,7 alpha-Dihydroxy-5beta-cholest-24-enoyl-CoA through the action of HSD17B4 (EC4.2.1.107). 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA is then converted to 3 alpha,7 alpha-Dihydroxy-5beta-24-oxocholestanoyl-CoA by HSD17B4 (EC1.1.1.35). |
|---|
| Structure | [H][C@@]12C[C@H](O)CC[C@]1(C)C1CC[C@]3(C)C(CCC3C1[C@H](O)C2)C(C)CCC(O)C(C)C(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C1N=CN=C2N InChI=1S/C48H80N7O20P3S/c1-25(29-8-9-30-36-31(12-15-48(29,30)6)47(5)14-11-28(56)19-27(47)20-33(36)58)7-10-32(57)26(2)45(63)79-18-17-50-35(59)13-16-51-43(62)40(61)46(3,4)22-72-78(69,70)75-77(67,68)71-21-34-39(74-76(64,65)66)38(60)44(73-34)55-24-54-37-41(49)52-23-53-42(37)55/h23-34,36,38-40,44,56-58,60-61H,7-22H2,1-6H3,(H,50,59)(H,51,62)(H,67,68)(H,69,70)(H2,49,52,53)(H2,64,65,66)/t25?,26?,27-,28+,29?,30?,31?,32?,33+,34+,36?,38+,39+,40?,44+,47-,48+/m0/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 3 a,7 a,24-Trihydroxy-5b-cholestanoyl-CoA | Generator | | 3 Α,7 α,24-trihydroxy-5β-cholestanoyl-CoA | Generator | | 3alpha,7alpha,24-Trihydroxy-5beta-cholestan-26-oyl-coenzyme A | ChEBI | | 3alpha,7alpha,24-Trihydroxy-5beta-cholestanoyl-CoA | ChEBI | | (24R,25R)-3alpha,7alpha,24-Trihydroxy-5beta-cholestan-26-oyl-CoA | KEGG | | 3a,7a,24-Trihydroxy-5b-cholestan-26-oyl-coenzyme A | Generator | | 3Α,7α,24-trihydroxy-5β-cholestan-26-oyl-coenzyme A | Generator | | 3a,7a,24-Trihydroxy-5b-cholestanoyl-CoA | Generator | | 3Α,7α,24-trihydroxy-5β-cholestanoyl-CoA | Generator | | (24R,25R)-3a,7a,24-Trihydroxy-5b-cholestan-26-oyl-CoA | Generator | | (24R,25R)-3Α,7α,24-trihydroxy-5β-cholestan-26-oyl-CoA | Generator |
|
|---|
| Chemical Formula | C48H80N7O20P3S |
|---|
| Average Molecular Weight | 1200.17 |
|---|
| Monoisotopic Molecular Weight | 1199.439168261 |
|---|
| IUPAC Name | {[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-2-[({[({3-[(2-{[2-({6-[(2S,5R,7S,9R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-3-hydroxy-2-methylheptanoyl}sulfanyl)ethyl]carbamoyl}ethyl)carbamoyl]-3-hydroxy-2,2-dimethylpropoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)methyl]-4-hydroxyoxolan-3-yl]oxy}phosphonic acid |
|---|
| Traditional Name | [(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-2-{[({3-[(2-{[2-({6-[(2S,5R,7S,9R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-3-hydroxy-2-methylheptanoyl}sulfanyl)ethyl]carbamoyl}ethyl)carbamoyl]-3-hydroxy-2,2-dimethylpropoxy(hydroxy)phosphoryl}oxy(hydroxy)phosphoryl)oxy]methyl}-4-hydroxyoxolan-3-yl]oxyphosphonic acid |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H][C@@]12C[C@H](O)CC[C@]1(C)C1CC[C@]3(C)C(CCC3C1[C@H](O)C2)C(C)CCC(O)C(C)C(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C1N=CN=C2N |
|---|
| InChI Identifier | InChI=1S/C48H80N7O20P3S/c1-25(29-8-9-30-36-31(12-15-48(29,30)6)47(5)14-11-28(56)19-27(47)20-33(36)58)7-10-32(57)26(2)45(63)79-18-17-50-35(59)13-16-51-43(62)40(61)46(3,4)22-72-78(69,70)75-77(67,68)71-21-34-39(74-76(64,65)66)38(60)44(73-34)55-24-54-37-41(49)52-23-53-42(37)55/h23-34,36,38-40,44,56-58,60-61H,7-22H2,1-6H3,(H,50,59)(H,51,62)(H,67,68)(H,69,70)(H2,49,52,53)(H2,64,65,66)/t25?,26?,27-,28+,29?,30?,31?,32?,33+,34+,36?,38+,39+,40?,44+,47-,48+/m0/s1 |
|---|
| InChI Key | SZBMUAIJWNJARR-ATZLTZGYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as 3-hydroxyacyl coas. These are organic compounds containing a 3-hydroxyl acylated coenzyme A derivative. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acyl thioesters |
|---|
| Direct Parent | 3-hydroxyacyl CoAs |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coenzyme a or derivatives
- Purine ribonucleoside 3',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Steroidal glycoside
- Trihydroxy bile acid, alcohol, or derivatives
- 24-hydroxysteroid
- Hydroxy bile acid, alcohol, or derivatives
- Cholane-skeleton
- Bile acid, alcohol, or derivatives
- 3-hydroxysteroid
- Hydroxysteroid
- 3-alpha-hydroxysteroid
- 7-hydroxysteroid
- Steroid
- Ribonucleoside 3'-phosphate
- Pentose phosphate
- Pentose-5-phosphate
- Beta amino acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- Organic pyrophosphate
- 6-aminopurine
- Monosaccharide phosphate
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Aminopyrimidine
- Pyrimidine
- Fatty amide
- Monosaccharide
- N-acyl-amine
- Phosphoric acid ester
- N-substituted imidazole
- Imidolactam
- Organic phosphoric acid derivative
- Alkyl phosphate
- Azole
- Tetrahydrofuran
- Cyclic alcohol
- Imidazole
- Heteroaromatic compound
- Amino acid or derivatives
- Carbothioic s-ester
- Carboxamide group
- Secondary alcohol
- Secondary carboxylic acid amide
- Thiocarboxylic acid ester
- Oxacycle
- Azacycle
- Thiocarboxylic acid or derivatives
- Sulfenyl compound
- Organoheterocyclic compound
- Carboxylic acid derivative
- Organonitrogen compound
- Organopnictogen compound
- Organic nitrogen compound
- Carbonyl group
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Amine
- Primary amine
- Alcohol
- Organosulfur compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesNot Available |
|---|
| MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 10V, Positive-QTOF | splash10-01p9-1900012000-622aba437f7430e017fb | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 20V, Positive-QTOF | splash10-000i-0900225000-2ec5547c65b2f24e0c94 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 40V, Positive-QTOF | splash10-000i-1900011000-ed3cd21797c8357fb61c | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 10V, Negative-QTOF | splash10-001i-2900432410-2a5b364cc28a01f6501b | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 20V, Negative-QTOF | splash10-001i-3900121010-4fe3346cb382c95a93b6 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 40V, Negative-QTOF | splash10-057i-5900100000-fd2f0a5af0fa8cb2631c | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 10V, Negative-QTOF | splash10-0002-0900000000-65cf19f1888cb6ceb838 | 2021-09-21 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 20V, Negative-QTOF | splash10-000t-1900301100-c1a7499a6c8ed6e7c77c | 2021-09-21 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 40V, Negative-QTOF | splash10-05ra-5603903700-dc786a780fcf9d33e01b | 2021-09-21 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 10V, Positive-QTOF | splash10-0ue9-0890000000-de8bcb28860c798ed88e | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 20V, Positive-QTOF | splash10-03el-0910000030-ef9afdd365f76dc605db | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3 alpha,7 alpha,24-Trihydroxy-5beta-cholestanoyl-CoA 40V, Positive-QTOF | splash10-0006-0000019000-74c41a4f5ab647d8c277 | 2021-09-25 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum |
|
|---|