| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2006-05-22 14:17:42 UTC |
|---|
| Update Date | 2021-09-14 15:20:03 UTC |
|---|
| HMDB ID | HMDB0002217 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | (S)-Methylmalonic acid semialdehyde |
|---|
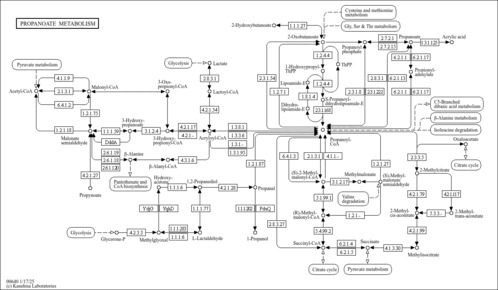
| Description | (S)-Methylmalonic acid semialdehyde belongs to the class of organic compounds known as 1,3-dicarbonyl compounds. These are carbonyl compounds with the generic formula O=C(R)C(H)C(R')=O, where R and R' can be any group (S)-Methylmalonic acid semialdehyde exists in all living organisms, ranging from bacteria to humans. In humans, (S)-methylmalonic acid semialdehyde is involved in the metabolic disorder called the 2-methyl-3-hydroxybutyryl-coa dehydrogenase deficiency pathway (S)-Methylmalonic acid semialdehyde has been detected, but not quantified in, several different foods, such as black-eyed peas (Vigna unguiculata ssp. unguiculata), vaccinium (blueberry, cranberry, huckleberry), leeks (Allium porrum), fenugreeks (Trigonella foenum-graecum), and sweet oranges (Citrus sinensis). This could make (S)-methylmalonic acid semialdehyde a potential biomarker for the consumption of these foods (S)-Methylmalonic acid semialdehyde is a primary metabolite. Primary metabolites are metabolically or physiologically essential metabolites. They are directly involved in an organism’s growth, development or reproduction. Based on a literature review a small amount of articles have been published on (S)-Methylmalonic acid semialdehyde. |
|---|
| Structure | InChI=1S/C4H6O3/c1-3(2-5)4(6)7/h2-3H,1H3,(H,6,7)/t3-/m0/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| (S)-Methylmalonate semialdehyde | ChEBI | | (2S)-2-Methyl-3-oxopropanoate | HMDB | | (2S)-2-Methyl-3-oxopropanoic acid | HMDB |
|
|---|
| Chemical Formula | C4H6O3 |
|---|
| Average Molecular Weight | 102.0886 |
|---|
| Monoisotopic Molecular Weight | 102.031694058 |
|---|
| IUPAC Name | (2S)-2-methyl-3-oxopropanoic acid |
|---|
| Traditional Name | (S)-methylmalonaldehydic acid |
|---|
| CAS Registry Number | 99043-16-0 |
|---|
| SMILES | C[C@@H](C=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C4H6O3/c1-3(2-5)4(6)7/h2-3H,1H3,(H,6,7)/t3-/m0/s1 |
|---|
| InChI Key | VOKUMXABRRXHAR-VKHMYHEASA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as 1,3-dicarbonyl compounds. These are carbonyl compounds with the generic formula O=C(R)C(H)C(R')=O, where R and R' can be any group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbonyl compounds |
|---|
| Direct Parent | 1,3-dicarbonyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1,3-dicarbonyl compound
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxide
- Hydrocarbon derivative
- Short-chain aldehyde
- Aldehyde
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| (S)-Methylmalonic acid semialdehyde,1TMS,isomer #1 | C[C@@H](C=O)C(=O)O[Si](C)(C)C | 1010.0 | Semi standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,1TMS,isomer #2 | CC(=CO[Si](C)(C)C)C(=O)O | 1121.8 | Semi standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,2TMS,isomer #1 | CC(=CO[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1222.1 | Semi standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,2TMS,isomer #1 | CC(=CO[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1140.6 | Standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,2TMS,isomer #1 | CC(=CO[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1241.6 | Standard polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,1TBDMS,isomer #1 | C[C@@H](C=O)C(=O)O[Si](C)(C)C(C)(C)C | 1224.3 | Semi standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,1TBDMS,isomer #2 | CC(=CO[Si](C)(C)C(C)(C)C)C(=O)O | 1366.4 | Semi standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,2TBDMS,isomer #1 | CC(=CO[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1627.5 | Semi standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,2TBDMS,isomer #1 | CC(=CO[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1567.2 | Standard non polar | 33892256 | | (S)-Methylmalonic acid semialdehyde,2TBDMS,isomer #1 | CC(=CO[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1564.5 | Standard polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - (S)-Methylmalonic acid semialdehyde GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-9000000000-8b181508e3ce65539b46 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - (S)-Methylmalonic acid semialdehyde GC-MS (1 TMS) - 70eV, Positive | splash10-00di-9600000000-15e42a2e09123d5f22ad | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - (S)-Methylmalonic acid semialdehyde GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 10V, Positive-QTOF | splash10-0zg0-9400000000-e46caa43ec4709ce7686 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 20V, Positive-QTOF | splash10-0a4i-9000000000-6c1632ad1d7983990262 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 40V, Positive-QTOF | splash10-0a4i-9000000000-6762a2cf4db52c84495c | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 10V, Negative-QTOF | splash10-0udi-2900000000-9066dc83487b73946c99 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 20V, Negative-QTOF | splash10-0a4i-9200000000-8fde49823ba2083f055f | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 40V, Negative-QTOF | splash10-0a4i-9000000000-a320f14ddfc09e798938 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 10V, Negative-QTOF | splash10-0uk9-8900000000-0a76d84228c38b1cfe1d | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 20V, Negative-QTOF | splash10-0pb9-9300000000-dd49ef94a8379e8517cd | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 40V, Negative-QTOF | splash10-0a4i-9000000000-4f42a2a7dc0b37e742da | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 10V, Positive-QTOF | splash10-0a4i-9000000000-b5c480248a004c069416 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 20V, Positive-QTOF | splash10-0a4i-9000000000-6de8f6b91e5efd633c28 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - (S)-Methylmalonic acid semialdehyde 40V, Positive-QTOF | splash10-0a4r-9000000000-a0e9ace5e33e8c50915e | 2021-09-22 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-22 | Wishart Lab | View Spectrum |
|
|---|