| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2022-03-07 02:49:04 UTC |
|---|
| HMDB ID | HMDB0000631 |
|---|
| Secondary Accession Numbers | - HMDB0004012
- HMDB00631
- HMDB04012
|
|---|
| Metabolite Identification |
|---|
| Common Name | Deoxycholic acid glycine conjugate |
|---|
| Description | Deoxycholic acid glycine conjugate, or or Deoxyglycocholic acid or Deoxygcholylglycine is a bile salt formed in the liver by conjugation of deoxycholate with glycine. It usually exists as the sodium salt. Deoxygcholylglycine is a secondary bile acid produced by the action of enzymes existing in the microbial flora of the colonic environment. In hepatocytes, both primary and secondary bile acids undergo amino acid conjugation at the C-24 carboxylic acid on the side chain, and almost all bile acids in the bile duct therefore exist in a glycine conjugated form (PMID: 16949895 ). As a bile acid Deoxyglycocholic acid acts as a detergent to solubilize fats for absorption and is itself absorbed. Bile acids are steroid acids found predominantly in the bile of mammals. The distinction between different bile acids is minute, and depends only on the presence or absence of hydroxyl groups on positions 3, 7, and 12. Bile acids are physiological detergents that facilitate excretion, absorption, and transport of fats and sterols in the intestine and liver. Bile acids are also steroidal amphipathic molecules derived from the catabolism of cholesterol. They modulate bile flow and lipid secretion, are essential for the absorption of dietary fats and vitamins, and have been implicated in the regulation of all the key enzymes involved in cholesterol homeostasis. Bile acids recirculate through the liver, bile ducts, small intestine, and portal vein to form an enterohepatic circuit. They exist as anions at physiological pH, and consequently require a carrier for transport across the membranes of the enterohepatic tissues. The unique detergent properties of bile acids are essential for the digestion and intestinal absorption of hydrophobic nutrients. Bile acids have potent toxic properties (e.g. membrane disruption) and there are a plethora of mechanisms to limit their accumulation in blood and tissues (PMID: 11316487 , 16037564 , 12576301 , 11907135 ). Deoxyglycocholic acid is used as a cholagogue and choleretic. |
|---|
| Structure | [H][C@]12CCC3C4CC[C@H]([C@H](C)CCC(=O)NCC(O)=O)[C@@]4(C)[C@@H](O)CC3[C@@]1(C)CC[C@@H](O)C2 InChI=1S/C26H43NO5/c1-15(4-9-23(30)27-14-24(31)32)19-7-8-20-18-6-5-16-12-17(28)10-11-25(16,2)21(18)13-22(29)26(19,20)3/h15-22,28-29H,4-14H2,1-3H3,(H,27,30)(H,31,32)/t15-,16-,17-,18?,19-,20?,21?,22+,25+,26-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| Deoxycholate glycine conjugate | Generator | | Deoxycholic acid glycine conjugic acid | Generator | | Acid, glycodeoxycholic | MeSH | | Deoxycholate, glycine | MeSH | | Glycine deoxycholate | MeSH | | Glycodeoxycholic acid | MeSH | | Glycodeoxycholate | MeSH | | Deoxycholylglycine | MeSH | | Deoxyglycocholate | HMDB | | Deoxyglycocholic acid | HMDB | | Glycodesoxycholate | HMDB | | Glycodesoxycholic acid | HMDB | | Glycyldeoxycholate | HMDB | | Glycyldeoxycholic acid | HMDB | | N-(3a,12a-Dihydroxy-5b-cholan-24-oyl)-glycine | HMDB | | N-(Carboxymethyl)-3a,12a-dihydroxy-5b-cholan-24-amide | HMDB |
|
|---|
| Chemical Formula | C26H43NO5 |
|---|
| Average Molecular Weight | 449.6233 |
|---|
| Monoisotopic Molecular Weight | 449.314123491 |
|---|
| IUPAC Name | 2-[(4R)-4-[(2S,5R,7R,14R,15R,16S)-5,16-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-yl]pentanamido]acetic acid |
|---|
| Traditional Name | [(4R)-4-[(2S,5R,7R,14R,15R,16S)-5,16-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-yl]pentanamido]acetic acid |
|---|
| CAS Registry Number | 360-65-6 |
|---|
| SMILES | [H][C@]12CCC3C4CC[C@H]([C@H](C)CCC(=O)NCC(O)=O)[C@@]4(C)[C@@H](O)CC3[C@@]1(C)CC[C@@H](O)C2 |
|---|
| InChI Identifier | InChI=1S/C26H43NO5/c1-15(4-9-23(30)27-14-24(31)32)19-7-8-20-18-6-5-16-12-17(28)10-11-25(16,2)21(18)13-22(29)26(19,20)3/h15-22,28-29H,4-14H2,1-3H3,(H,27,30)(H,31,32)/t15-,16-,17-,18?,19-,20?,21?,22+,25+,26-/m1/s1 |
|---|
| InChI Key | WVULKSPCQVQLCU-ZMBDPXIHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as glycinated bile acids and derivatives. Glycinated bile acids and derivatives are compounds with a structure characterized by the presence of a glycine linked to a bile acid skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Bile acids, alcohols and derivatives |
|---|
| Direct Parent | Glycinated bile acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Glycinated bile acid
- Dihydroxy bile acid, alcohol, or derivatives
- Hydroxy bile acid, alcohol, or derivatives
- 3-hydroxysteroid
- 12-hydroxysteroid
- Hydroxysteroid
- 3-alpha-hydroxysteroid
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- Alpha-amino acid or derivatives
- Fatty amide
- Fatty acyl
- N-acyl-amine
- Cyclic alcohol
- Carboxamide group
- Secondary alcohol
- Secondary carboxylic acid amide
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Hydrocarbon derivative
- Organic oxide
- Alcohol
- Organic nitrogen compound
- Organopnictogen compound
- Carbonyl group
- Organic oxygen compound
- Organooxygen compound
- Organonitrogen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 0.0027 mg/mL | Not Available | | LogP | 2.25 | RODA,A ET AL. (1990) |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | | Show more...
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| Deoxycholic acid glycine conjugate,1TMS,isomer #1 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 3905.7 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,1TMS,isomer #2 | C[C@H](CCC(=O)NCC(=O)O)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3915.1 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,1TMS,isomer #3 | C[C@H](CCC(=O)NCC(=O)O)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 3963.6 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,1TMS,isomer #4 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 3935.6 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TMS,isomer #1 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 3863.5 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TMS,isomer #2 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3850.3 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TMS,isomer #3 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 3876.0 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TMS,isomer #4 | C[C@H](CCC(=O)NCC(=O)O)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3844.1 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TMS,isomer #5 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3847.5 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TMS,isomer #6 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 3919.5 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TMS,isomer #1 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3771.0 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TMS,isomer #2 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 3828.2 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TMS,isomer #3 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3794.6 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TMS,isomer #4 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3780.3 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,4TMS,isomer #1 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3744.9 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,4TMS,isomer #1 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 3713.5 | Standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,4TMS,isomer #1 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C)[C@@]21C | 4078.9 | Standard polar | 33892256 | | Deoxycholic acid glycine conjugate,1TBDMS,isomer #1 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 4148.6 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,1TBDMS,isomer #2 | C[C@H](CCC(=O)NCC(=O)O)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C | 4129.1 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,1TBDMS,isomer #3 | C[C@H](CCC(=O)NCC(=O)O)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 4184.0 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,1TBDMS,isomer #4 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 4155.9 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TBDMS,isomer #1 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 4285.4 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TBDMS,isomer #2 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C | 4262.4 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TBDMS,isomer #3 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 4324.1 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TBDMS,isomer #4 | C[C@H](CCC(=O)NCC(=O)O)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C | 4268.2 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TBDMS,isomer #5 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C | 4295.5 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,2TBDMS,isomer #6 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 4363.0 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TBDMS,isomer #1 | C[C@H](CCC(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C | 4400.1 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TBDMS,isomer #2 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)C3C[C@H](O)[C@@]21C | 4497.8 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TBDMS,isomer #3 | C[C@H](CCC(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C | 4467.0 | Semi standard non polar | 33892256 | | Deoxycholic acid glycine conjugate,3TBDMS,isomer #4 | C[C@H](CCC(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[C@H]1CCC2C3CC[C@@H]4C[C@H](O[Si](C)(C)C(C)(C)C)CC[C@]4(C)C3C[C@H](O[Si](C)(C)C(C)(C)C)[C@@]21C | 4473.1 | Semi standard non polar | 33892256 |
| Show more...
|---|
| Spectra |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (Non-derivatized) - 70eV, Positive | splash10-001i-0124900000-9935025b691abf76f837 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (3 TMS) - 70eV, Positive | splash10-0udj-2101139000-a160ba76c005cfc50ac7 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_1_1) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_1_3) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_1_4) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_2_1) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_2_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_2_3) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_2_4) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_2_5) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_2_6) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_3_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_3_3) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TMS_3_4) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_1_3) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_1_4) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_2_1) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_2_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_2_3) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_2_4) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Deoxycholic acid glycine conjugate GC-MS (TBDMS_2_5) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate Quattro_QQQ 10V, Positive-QTOF (Annotated) | splash10-0ik9-0000900000-4a3e850d7237d0106c35 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate Quattro_QQQ 25V, Positive-QTOF (Annotated) | splash10-01ti-3894300000-fa0e7861362d7bf348bb | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate Quattro_QQQ 40V, Positive-QTOF (Annotated) | splash10-0a7i-5910000000-17134bf01487bc71d373 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 10V, Positive-QTOF | splash10-0gz9-2002900000-02545f99732b7619c59d | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 20V, Positive-QTOF | splash10-056r-9005400000-282d54d2414239340bc6 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 40V, Positive-QTOF | splash10-004i-9104100000-4eb16c3c40c7f98e3788 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 10V, Negative-QTOF | splash10-0002-0000900000-9e4d382267acd7d0da8c | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 20V, Negative-QTOF | splash10-00ea-4103900000-d0ad37cdb8c4ebcc8b3f | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 40V, Negative-QTOF | splash10-05fr-9001000000-23d56965528e66b241e7 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 10V, Negative-QTOF | splash10-0002-0000900000-1659120ddaaa97329ed4 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 20V, Negative-QTOF | splash10-0002-1001900000-74add9101b276384e02f | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 40V, Negative-QTOF | splash10-00dj-9003300000-7b2d9962126a043e2444 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 10V, Positive-QTOF | splash10-0ue9-0001900000-e79d7064b1c912d32774 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 20V, Positive-QTOF | splash10-0kuu-4855900000-7091abc8f240404bf159 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Deoxycholic acid glycine conjugate 40V, Positive-QTOF | splash10-0avi-9730300000-e569a38323705f9cdfc6 | 2021-09-22 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Experimental 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | 2012-12-04 | Wishart Lab | View Spectrum | | Experimental 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, CD3OD, experimental) | 2012-12-05 | Wishart Lab | View Spectrum |
| Show more...
|---|
| Biological Properties |
|---|
| Cellular Locations | |
|---|
| Biospecimen Locations | |
|---|
| Tissue Locations | |
|---|
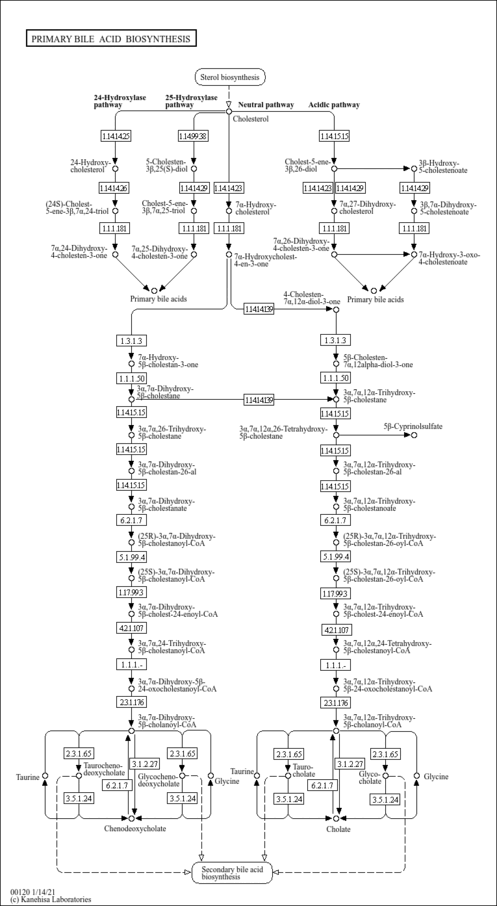
| Pathways | |
|---|
| Normal Concentrations |
|---|
| |
| Bile | Detected and Quantified | >0.01 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Feces | Detected and Quantified | 19.19 +/- 13.69 nmol/g dry feces | Not Specified | Not Specified | Normal | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details |
| Show more...
|---|
| Abnormal Concentrations |
|---|
| |
| Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Pancreatic ductal adenocarcinoma | | details | | Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Hepatocellular carcinoma | | details | | Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Liver Cirrhosis | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Colorectal Cancer | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Colorectal cancer | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Female | Epithelial ovarian cancer | | details |
|
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | | Hepatocellular carcinoma |
|---|
- Ressom HW, Xiao JF, Tuli L, Varghese RS, Zhou B, Tsai TH, Ranjbar MR, Zhao Y, Wang J, Di Poto C, Cheema AK, Tadesse MG, Goldman R, Shetty K: Utilization of metabolomics to identify serum biomarkers for hepatocellular carcinoma in patients with liver cirrhosis. Anal Chim Acta. 2012 Sep 19;743:90-100. doi: 10.1016/j.aca.2012.07.013. Epub 2012 Jul 20. [PubMed:22882828 ]
| | Cirrhosis |
|---|
- Ressom HW, Xiao JF, Tuli L, Varghese RS, Zhou B, Tsai TH, Ranjbar MR, Zhao Y, Wang J, Di Poto C, Cheema AK, Tadesse MG, Goldman R, Shetty K: Utilization of metabolomics to identify serum biomarkers for hepatocellular carcinoma in patients with liver cirrhosis. Anal Chim Acta. 2012 Sep 19;743:90-100. doi: 10.1016/j.aca.2012.07.013. Epub 2012 Jul 20. [PubMed:22882828 ]
| | Colorectal cancer |
|---|
- Brown DG, Rao S, Weir TL, O'Malia J, Bazan M, Brown RJ, Ryan EP: Metabolomics and metabolic pathway networks from human colorectal cancers, adjacent mucosa, and stool. Cancer Metab. 2016 Jun 6;4:11. doi: 10.1186/s40170-016-0151-y. eCollection 2016. [PubMed:27275383 ]
- Goedert JJ, Sampson JN, Moore SC, Xiao Q, Xiong X, Hayes RB, Ahn J, Shi J, Sinha R: Fecal metabolomics: assay performance and association with colorectal cancer. Carcinogenesis. 2014 Sep;35(9):2089-96. doi: 10.1093/carcin/bgu131. Epub 2014 Jul 18. [PubMed:25037050 ]
|
|
|---|
| Associated OMIM IDs | - 114550 (Hepatocellular carcinoma)
- 114500 (Colorectal cancer)
|
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | FDB022152 |
|---|
| KNApSAcK ID | C00030411 |
|---|
| Chemspider ID | 17215983 |
|---|
| KEGG Compound ID | C05464 |
|---|
| BioCyc ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| METLIN ID | 5604 |
|---|
| PubChem Compound | 22833539 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 27471 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | M01989 |
|---|
| MarkerDB ID | Not Available |
|---|
| Good Scents ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Parmentier G; Eyssen H Synthesis and characteristics of the specific monosulfates of chenodeoxycholate, deoxycholate and their taurine or glycine conjugates. Steroids (1977), 30(5), 583-90. |
|---|
| Material Safety Data Sheet (MSDS) | Download (PDF) |
|---|
| General References | - Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [PubMed:19212411 ]
- Hepner GW, Demers LM: Dynamics of the enterohepatic circulation of the glycine conjugates of cholic, chenodeoxycholic, deoxycholic, and sulfolithocholic acid in man. Gastroenterology. 1977 Mar;72(3):499-501. [PubMed:832799 ]
- Bloch CA, Watkins JB: Determination of conjugated bile acids in human bile and duodenal fluid by reverse-phase high-performance liquid chromatography. J Lipid Res. 1978 May;19(4):510-3. [PubMed:659989 ]
- Demers LM, Hepner GW: Levels of immunoreactive glycine-conjugated bile acids in health and hepatobiliary disease. Am J Clin Pathol. 1976 Nov;66(5):831-9. [PubMed:983991 ]
- Demers LM, Hepner G: Radioimmunoassay of bile acids in serum. Clin Chem. 1976 May;22(5):602-6. [PubMed:1261008 ]
- Goto T, Myint KT, Sato K, Wada O, Kakiyama G, Iida T, Hishinuma T, Mano N, Goto J: LC/ESI-tandem mass spectrometric determination of bile acid 3-sulfates in human urine 3beta-Sulfooxy-12alpha-hydroxy-5beta-cholanoic acid is an abundant nonamidated sulfate. J Chromatogr B Analyt Technol Biomed Life Sci. 2007 Feb 1;846(1-2):69-77. Epub 2006 Sep 1. [PubMed:16949895 ]
- St-Pierre MV, Kullak-Ublick GA, Hagenbuch B, Meier PJ: Transport of bile acids in hepatic and non-hepatic tissues. J Exp Biol. 2001 May;204(Pt 10):1673-86. [PubMed:11316487 ]
- Claudel T, Staels B, Kuipers F: The Farnesoid X receptor: a molecular link between bile acid and lipid and glucose metabolism. Arterioscler Thromb Vasc Biol. 2005 Oct;25(10):2020-30. Epub 2005 Jul 21. [PubMed:16037564 ]
- Chiang JY: Bile acid regulation of hepatic physiology: III. Bile acids and nuclear receptors. Am J Physiol Gastrointest Liver Physiol. 2003 Mar;284(3):G349-56. [PubMed:12576301 ]
- Davis RA, Miyake JH, Hui TY, Spann NJ: Regulation of cholesterol-7alpha-hydroxylase: BAREly missing a SHP. J Lipid Res. 2002 Apr;43(4):533-43. [PubMed:11907135 ]
|
|---|