| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2006-05-22 14:17:49 UTC |
|---|
| Update Date | 2022-09-22 17:43:49 UTC |
|---|
| HMDB ID | HMDB0002343 |
|---|
| Secondary Accession Numbers | - HMDB0004675
- HMDB02343
- HMDB04675
|
|---|
| Metabolite Identification |
|---|
| Common Name | 5,6-DHET |
|---|
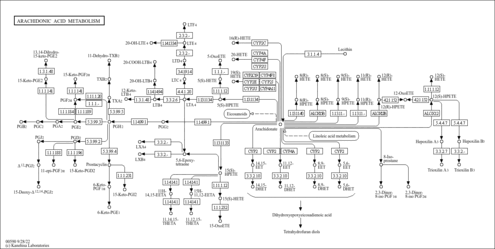
| Description | 5,6-DHET is an epoxide intermediate in the oxygenation of arachidonic acid by hepatic monooxygenases pathway. 5,6-DHET is the hydrolysis metabolite of cis-5(6)Epoxy-cis-8,11,14-eicosatrienoic acid by epoxide hydrolases. Many drugs, chemicals, and endogenous compounds are oxygenated in mammalian tissues and in some instances reactive and potentially toxic or carcinogenic epoxides are formed. Naturally occurring olefins may also be oxygenated by mammalian enzymes. The most well known are lipoxygenases and microsomal cytochrome P-450-linked monooxygenases. The epoxides may be chemically labile or may be enzymatically hydrolyzed. When arene or olefinic epoxides are formed by microsomal P-450-linked monooxygenases, they are often rapidly converted to less reactive trans-diols through the action of microsomal epoxide hydrolases. (PMID: 6801052 , 6548162 ). |
|---|
| Structure | CCCCC\C=C/C\C=C/C\C=C/CC(O)C(O)CCCC(O)=O InChI=1S/C20H34O4/c1-2-3-4-5-6-7-8-9-10-11-12-13-15-18(21)19(22)16-14-17-20(23)24/h6-7,9-10,12-13,18-19,21-22H,2-5,8,11,14-17H2,1H3,(H,23,24)/b7-6-,10-9-,13-12- |
|---|
| Synonyms | | Value | Source |
|---|
| (+/-)5,6-dihetre | ChEBI | | (8Z,11Z,14Z)-5,6-Dihydroxyeicosa-8,11,14-trienoic acid | ChEBI | | (8Z,11Z,14Z)-5,6-Dihydroxyicosa-8,11,14-trienoic acid | ChEBI | | 5,6-DiHETrE | ChEBI | | 5,6-Dihydroxy-8Z,11Z,14Z-eicosatrienoic acid | ChEBI | | 5,6-Dihydroxy-8Z,11Z,14Z-icosatrienoic acid | ChEBI | | (8Z,11Z,14Z)-5,6-Dihydroxyeicosa-8,11,14-trienoate | Generator | | (8Z,11Z,14Z)-5,6-Dihydroxyicosa-8,11,14-trienoate | Generator | | 5,6-Dihydroxy-8Z,11Z,14Z-eicosatrienoate | Generator | | 5,6-Dihydroxy-8Z,11Z,14Z-icosatrienoate | Generator |
|
|---|
| Chemical Formula | C20H34O4 |
|---|
| Average Molecular Weight | 338.4816 |
|---|
| Monoisotopic Molecular Weight | 338.245709576 |
|---|
| IUPAC Name | (8Z,11Z,14Z)-5,6-dihydroxyicosa-8,11,14-trienoic acid |
|---|
| Traditional Name | 5,6-DiHETrE |
|---|
| CAS Registry Number | 213382-49-1 |
|---|
| SMILES | CCCCC\C=C/C\C=C/C\C=C/CC(O)C(O)CCCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H34O4/c1-2-3-4-5-6-7-8-9-10-11-12-13-15-18(21)19(22)16-14-17-20(23)24/h6-7,9-10,12-13,18-19,21-22H,2-5,8,11,14-17H2,1H3,(H,23,24)/b7-6-,10-9-,13-12- |
|---|
| InChI Key | GFNYAPAJUNPMGH-QNEBEIHSSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as hydroxyeicosatrienoic acids. These are eicosanoic acids with an attached hydroxyl group and three CC double bonds. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Eicosanoids |
|---|
| Direct Parent | Hydroxyeicosatrienoic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hydroxyeicosatrienoic acid
- Long-chain fatty acid
- Hydroxy fatty acid
- Unsaturated fatty acid
- Fatty acid
- Secondary alcohol
- 1,2-diol
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 5,6-DHET,1TMS,isomer #1 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C)C(O)CCCC(=O)O | 2824.5 | Semi standard non polar | 33892256 | | 5,6-DHET,1TMS,isomer #2 | CCCCC/C=C\C/C=C\C/C=C\CC(O)C(CCCC(=O)O)O[Si](C)(C)C | 2815.5 | Semi standard non polar | 33892256 | | 5,6-DHET,1TMS,isomer #3 | CCCCC/C=C\C/C=C\C/C=C\CC(O)C(O)CCCC(=O)O[Si](C)(C)C | 2736.1 | Semi standard non polar | 33892256 | | 5,6-DHET,2TMS,isomer #1 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C)C(CCCC(=O)O)O[Si](C)(C)C | 2803.9 | Semi standard non polar | 33892256 | | 5,6-DHET,2TMS,isomer #2 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C)C(O)CCCC(=O)O[Si](C)(C)C | 2762.6 | Semi standard non polar | 33892256 | | 5,6-DHET,2TMS,isomer #3 | CCCCC/C=C\C/C=C\C/C=C\CC(O)C(CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 2750.9 | Semi standard non polar | 33892256 | | 5,6-DHET,3TMS,isomer #1 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C)C(CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 2739.1 | Semi standard non polar | 33892256 | | 5,6-DHET,1TBDMS,isomer #1 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C(C)(C)C)C(O)CCCC(=O)O | 3076.0 | Semi standard non polar | 33892256 | | 5,6-DHET,1TBDMS,isomer #2 | CCCCC/C=C\C/C=C\C/C=C\CC(O)C(CCCC(=O)O)O[Si](C)(C)C(C)(C)C | 3071.3 | Semi standard non polar | 33892256 | | 5,6-DHET,1TBDMS,isomer #3 | CCCCC/C=C\C/C=C\C/C=C\CC(O)C(O)CCCC(=O)O[Si](C)(C)C(C)(C)C | 2980.0 | Semi standard non polar | 33892256 | | 5,6-DHET,2TBDMS,isomer #1 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C(C)(C)C)C(CCCC(=O)O)O[Si](C)(C)C(C)(C)C | 3255.4 | Semi standard non polar | 33892256 | | 5,6-DHET,2TBDMS,isomer #2 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C(C)(C)C)C(O)CCCC(=O)O[Si](C)(C)C(C)(C)C | 3234.5 | Semi standard non polar | 33892256 | | 5,6-DHET,2TBDMS,isomer #3 | CCCCC/C=C\C/C=C\C/C=C\CC(O)C(CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3223.5 | Semi standard non polar | 33892256 | | 5,6-DHET,3TBDMS,isomer #1 | CCCCC/C=C\C/C=C\C/C=C\CC(O[Si](C)(C)C(C)(C)C)C(CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3431.4 | Semi standard non polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - 5,6-DHET GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-5893000000-644b265c8d0033c153a9 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 5,6-DHET GC-MS (3 TMS) - 70eV, Positive | splash10-009l-9222440000-5ade17a2dea5f318127b | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 5,6-DHET GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 10V, Positive-QTOF | splash10-00di-0119000000-f4202d6e168a885cfc7a | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 20V, Positive-QTOF | splash10-0006-9633000000-4bbc37276adca2cc7764 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 40V, Positive-QTOF | splash10-0006-9630000000-158b1997b1c24dfab32b | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 10V, Negative-QTOF | splash10-000i-0019000000-94be53b1436416a5170f | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 20V, Negative-QTOF | splash10-014u-5689000000-25170110627624a1f600 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 40V, Negative-QTOF | splash10-0a4i-9340000000-1bd1c79e4bc1b6ce57aa | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 10V, Negative-QTOF | splash10-000i-0009000000-945cd3bf83aabf5d0673 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 20V, Negative-QTOF | splash10-014s-7649000000-6b70b3117d96d37e7df3 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 40V, Negative-QTOF | splash10-05mn-9200000000-e6d9c0d22c30c01eb8f5 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 10V, Positive-QTOF | splash10-0fk9-2209000000-a0f3d1a2de3540d03cfc | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 20V, Positive-QTOF | splash10-00e9-9703000000-9a45a450e1c54e75f564 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5,6-DHET 40V, Positive-QTOF | splash10-05nf-9200000000-357fecf17a9ed28b52d6 | 2021-09-24 | Wishart Lab | View Spectrum |
|
|---|