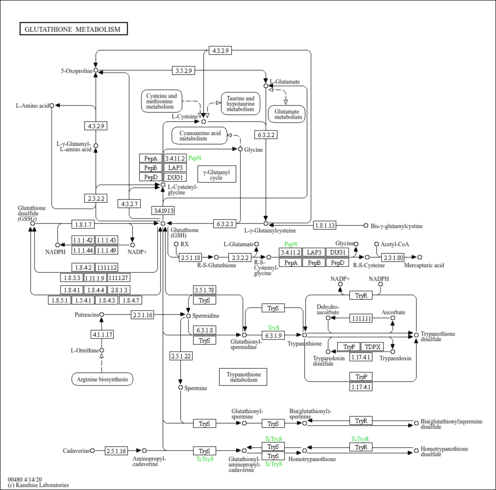
| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| Glutathione,1TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O | 2805.9 | Semi standard non polar | 33892256 |
| Glutathione,1TMS,isomer #2 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O | 2768.8 | Semi standard non polar | 33892256 |
| Glutathione,1TMS,isomer #3 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@H](N)C(=O)O)C(=O)NCC(=O)O | 2899.5 | Semi standard non polar | 33892256 |
| Glutathione,1TMS,isomer #4 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)C(=O)O | 2849.4 | Semi standard non polar | 33892256 |
| Glutathione,1TMS,isomer #5 | C[Si](C)(C)N(C(=O)CC[C@H](N)C(=O)O)[C@@H](CS)C(=O)NCC(=O)O | 2808.2 | Semi standard non polar | 33892256 |
| Glutathione,1TMS,isomer #6 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O | 2826.0 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C | 2769.5 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)C(=O)O | 2936.6 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #11 | C[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2845.4 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #12 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@H](N)C(=O)O)C(=O)N(CC(=O)O)[Si](C)(C)C | 2883.0 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #13 | C[Si](C)(C)N([C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)C(=O)O)[Si](C)(C)C | 2930.5 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #14 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O | 2844.0 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #15 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O | 2864.3 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #16 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2746.7 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #2 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O | 2864.5 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #3 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O | 2849.1 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #4 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2781.0 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #5 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2760.9 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #6 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O | 2843.9 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #7 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C | 2821.6 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #8 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C | 2743.3 | Semi standard non polar | 33892256 |
| Glutathione,2TMS,isomer #9 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C | 2755.9 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C | 2846.8 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C | 2788.4 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C | 4692.5 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2812.7 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2700.1 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3904.1 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2744.4 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2720.9 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C)[Si](C)(C)C | 4235.7 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #12 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C | 2907.9 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #12 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C | 2797.5 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #12 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C | 4331.3 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #13 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C | 2810.4 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #13 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C | 2816.6 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #13 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C | 4633.3 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #14 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C | 2854.7 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #14 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C | 2799.3 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #14 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C | 4558.1 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2920.5 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2707.7 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 3942.1 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #16 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2781.4 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #16 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2715.3 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #16 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3845.2 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #17 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2806.3 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #17 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2678.7 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #17 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3846.0 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #18 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2733.9 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #18 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2701.7 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #18 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C | 4106.7 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #19 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)NCC(=O)O | 3025.7 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #19 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)NCC(=O)O | 2877.4 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #19 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)NCC(=O)O | 4422.0 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #2 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2813.0 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #2 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2699.7 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #2 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3831.7 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #20 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O | 2894.2 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #20 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O | 2863.4 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #20 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O | 4399.4 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #21 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O | 2938.7 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #21 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O | 2856.3 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #21 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O | 4304.2 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #22 | C[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2841.3 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #22 | C[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2872.5 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #22 | C[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 4746.9 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #23 | C[Si](C)(C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[C@@H](CS)C(=O)NCC(=O)O | 2911.9 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #23 | C[Si](C)(C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[C@@H](CS)C(=O)NCC(=O)O | 2775.1 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #23 | C[Si](C)(C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[C@@H](CS)C(=O)NCC(=O)O | 3984.1 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #24 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2919.0 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #24 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2739.8 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #24 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 3977.8 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #25 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2790.9 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #25 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2734.2 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #25 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3905.3 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #3 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2718.9 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #3 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2671.5 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #3 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 4125.4 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #4 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2727.5 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #4 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2713.0 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #4 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 4085.7 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #5 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O | 2932.5 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #5 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O | 2845.5 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #5 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O | 4421.5 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #6 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2843.4 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #6 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2840.2 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #6 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 4804.3 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #7 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2813.7 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #7 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 2868.7 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #7 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C | 4836.1 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #8 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2944.5 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #8 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2754.8 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #8 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 3962.8 | Standard polar | 33892256 |
| Glutathione,3TMS,isomer #9 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2791.7 | Semi standard non polar | 33892256 |
| Glutathione,3TMS,isomer #9 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2749.3 | Standard non polar | 33892256 |
| Glutathione,3TMS,isomer #9 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3880.9 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #1 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2905.4 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #1 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2856.7 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #1 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3890.5 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2891.8 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2884.2 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3922.7 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2827.3 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2903.4 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C)[Si](C)(C)C | 4450.2 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #12 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2909.3 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #12 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2825.5 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #12 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3754.7 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #13 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2901.1 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #13 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2877.1 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #13 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3731.0 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #14 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2752.0 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #14 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2828.1 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #14 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3681.5 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 3007.5 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2886.0 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 3960.1 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #16 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2853.1 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #16 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2878.3 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #16 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3863.3 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #17 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2900.3 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #17 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2872.1 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #17 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3811.0 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #18 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2839.8 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #18 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C | 2864.8 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #18 | C[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C | 4238.0 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #19 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2925.3 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #19 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2842.6 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #19 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3715.8 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #2 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2792.7 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #2 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2833.8 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #2 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 4325.9 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #20 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2925.5 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #20 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2798.4 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #20 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3723.5 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #21 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2736.1 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #21 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2814.0 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #21 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3645.0 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #22 | C[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2983.9 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #22 | C[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2968.3 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #22 | C[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 4002.1 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #23 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C | 3034.9 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #23 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C | 2958.1 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #23 | C[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C | 3933.2 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #24 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2887.8 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #24 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2927.1 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #24 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3904.4 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #25 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2922.4 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #25 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2874.6 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #25 | C[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3751.6 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #3 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2781.8 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #3 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 2855.8 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #3 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C | 4338.6 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #4 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2929.1 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #4 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2813.3 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #4 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3708.5 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #5 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2733.6 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #5 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2820.0 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #5 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3619.1 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #6 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2751.2 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #6 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2770.0 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #6 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3646.3 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #7 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2704.3 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #7 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2792.9 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #7 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3885.8 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #8 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 3030.7 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #8 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 2935.0 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #8 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C | 3991.1 | Standard polar | 33892256 |
| Glutathione,4TMS,isomer #9 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2868.5 | Semi standard non polar | 33892256 |
| Glutathione,4TMS,isomer #9 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2908.5 | Standard non polar | 33892256 |
| Glutathione,4TMS,isomer #9 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3927.1 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2993.9 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2936.0 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #1 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3606.7 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2867.6 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 2960.1 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #10 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O | 3562.0 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2913.0 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2961.6 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #11 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3577.5 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #12 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2996.6 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #12 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2971.6 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #12 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3596.4 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #13 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3009.7 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #13 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2957.9 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #13 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3558.9 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #14 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2857.8 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #14 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2949.9 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #14 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3469.9 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2930.5 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 2938.8 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #15 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3558.3 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #16 | C[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3023.5 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #16 | C[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3041.4 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #16 | C[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3639.3 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #2 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2817.2 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #2 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2930.7 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #2 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3457.9 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #3 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2835.9 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #3 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2913.1 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #3 | C[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3485.7 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #4 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2780.2 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #4 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2911.8 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #4 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 4055.4 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #5 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2864.7 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #5 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2885.3 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #5 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3556.7 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #6 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2899.1 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #6 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2939.7 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #6 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3530.4 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #7 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2694.0 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #7 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2903.0 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #7 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3472.1 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #8 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2999.7 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #8 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2986.6 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #8 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3612.6 | Standard polar | 33892256 |
| Glutathione,5TMS,isomer #9 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2992.8 | Semi standard non polar | 33892256 |
| Glutathione,5TMS,isomer #9 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3013.2 | Standard non polar | 33892256 |
| Glutathione,5TMS,isomer #9 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3609.7 | Standard polar | 33892256 |
| Glutathione,6TMS,isomer #1 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2967.4 | Semi standard non polar | 33892256 |
| Glutathione,6TMS,isomer #1 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2995.1 | Standard non polar | 33892256 |
| Glutathione,6TMS,isomer #1 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3293.0 | Standard polar | 33892256 |
| Glutathione,6TMS,isomer #2 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2987.1 | Semi standard non polar | 33892256 |
| Glutathione,6TMS,isomer #2 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3015.8 | Standard non polar | 33892256 |
| Glutathione,6TMS,isomer #2 | C[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3279.3 | Standard polar | 33892256 |
| Glutathione,6TMS,isomer #3 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2812.5 | Semi standard non polar | 33892256 |
| Glutathione,6TMS,isomer #3 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 2989.9 | Standard non polar | 33892256 |
| Glutathione,6TMS,isomer #3 | C[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)C(=O)O[Si](C)(C)C | 3187.5 | Standard polar | 33892256 |
| Glutathione,6TMS,isomer #4 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 2916.4 | Semi standard non polar | 33892256 |
| Glutathione,6TMS,isomer #4 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3025.6 | Standard non polar | 33892256 |
| Glutathione,6TMS,isomer #4 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3433.6 | Standard polar | 33892256 |
| Glutathione,6TMS,isomer #5 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3004.7 | Semi standard non polar | 33892256 |
| Glutathione,6TMS,isomer #5 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3068.2 | Standard non polar | 33892256 |
| Glutathione,6TMS,isomer #5 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3327.9 | Standard polar | 33892256 |
| Glutathione,6TMS,isomer #6 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3019.7 | Semi standard non polar | 33892256 |
| Glutathione,6TMS,isomer #6 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3046.6 | Standard non polar | 33892256 |
| Glutathione,6TMS,isomer #6 | C[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C)[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C | 3290.2 | Standard polar | 33892256 |
| Glutathione,7TMS,isomer #1 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3016.4 | Semi standard non polar | 33892256 |
| Glutathione,7TMS,isomer #1 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3081.6 | Standard non polar | 33892256 |
| Glutathione,7TMS,isomer #1 | C[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C)N([Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C)[Si](C)(C)C | 3108.7 | Standard polar | 33892256 |
| Glutathione,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O | 3091.4 | Semi standard non polar | 33892256 |
| Glutathione,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O | 3057.5 | Semi standard non polar | 33892256 |
| Glutathione,1TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@H](N)C(=O)O)C(=O)NCC(=O)O | 3168.9 | Semi standard non polar | 33892256 |
| Glutathione,1TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)C(=O)O | 3119.1 | Semi standard non polar | 33892256 |
| Glutathione,1TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N(C(=O)CC[C@H](N)C(=O)O)[C@@H](CS)C(=O)NCC(=O)O | 3090.2 | Semi standard non polar | 33892256 |
| Glutathione,1TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O | 3096.6 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C | 3277.2 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)C(=O)O | 3410.5 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3351.3 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@H](N)C(=O)O)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 3354.2 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)N([C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)C(=O)O)[Si](C)(C)C(C)(C)C | 3427.1 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 3340.9 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 3332.4 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3247.5 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O | 3374.4 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O | 3345.8 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3285.8 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3292.7 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O | 3319.2 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C(C)(C)C | 3304.8 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C | 3266.0 | Semi standard non polar | 33892256 |
| Glutathione,2TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 3256.9 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C | 3588.2 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C | 3362.7 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C | 4480.3 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3530.8 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3241.5 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 4013.9 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3459.6 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3274.9 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4243.8 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C(C)(C)C | 3606.3 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C(C)(C)C | 3350.4 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)C(=O)O[Si](C)(C)C(C)(C)C | 4155.1 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C | 3555.9 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C | 3388.3 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C | 4432.9 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 3552.2 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 3361.2 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 4409.3 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3642.6 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3235.9 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4014.1 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3514.0 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3246.6 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3962.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #17 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3501.0 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #17 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3215.6 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #17 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3980.4 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #18 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3431.6 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #18 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3252.6 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #18 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4174.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #19 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O | 3729.4 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #19 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O | 3405.5 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #19 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O | 4217.2 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3530.9 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3231.1 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3958.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #20 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 3613.5 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #20 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 3393.4 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #20 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 4200.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #21 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 3606.3 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #21 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 3389.8 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #21 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O | 4163.0 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #22 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3535.9 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #22 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3401.6 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #22 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 4530.9 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #23 | CC(C)(C)[Si](C)(C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[C@@H](CS)C(=O)NCC(=O)O | 3633.4 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #23 | CC(C)(C)[Si](C)(C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[C@@H](CS)C(=O)NCC(=O)O | 3284.9 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #23 | CC(C)(C)[Si](C)(C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[C@@H](CS)C(=O)NCC(=O)O | 4029.6 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #24 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3609.9 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #24 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3254.2 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #24 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4044.8 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #25 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3499.2 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #25 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3268.1 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #25 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 4014.2 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3484.8 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3236.1 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4192.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3484.7 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3259.5 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4155.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O | 3649.8 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O | 3384.5 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O | 4208.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3574.8 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3394.4 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 4567.7 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3581.0 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 3409.1 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C | 4564.6 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3648.6 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3258.2 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4024.4 | Standard polar | 33892256 |
| Glutathione,3TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3538.5 | Semi standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3272.8 | Standard non polar | 33892256 |
| Glutathione,3TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3985.6 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3815.7 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3519.7 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3933.4 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3815.5 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3552.4 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3970.7 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3753.7 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3581.7 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4354.0 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3839.1 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3490.8 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3937.5 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3859.7 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3528.8 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3908.3 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3690.7 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3507.0 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3902.1 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3939.4 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3563.8 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3977.6 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3793.3 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3541.5 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3931.3 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #17 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3784.8 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #17 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3527.3 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #17 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3920.2 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #18 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3731.9 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #18 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3545.7 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #18 | CC(C)(C)[Si](C)(C)OC(=O)[C@@H](N)CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4229.3 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #19 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3855.0 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #19 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3513.2 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #19 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3899.5 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3755.4 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3529.1 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4251.5 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #20 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3840.6 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #20 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3472.3 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #20 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3925.6 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #21 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3658.1 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #21 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3482.2 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #21 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3881.2 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #22 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3932.8 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #22 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3609.3 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #22 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)NCC(=O)O)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4005.7 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #23 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 3918.7 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #23 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 3593.7 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #23 | CC(C)(C)[Si](C)(C)SC[C@H](NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C | 3983.7 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #24 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3780.5 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #24 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3569.7 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #24 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3982.4 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #25 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3806.1 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #25 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3530.8 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #25 | CC(C)(C)[Si](C)(C)N(CC(=O)O)C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3943.6 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3764.8 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3540.5 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4245.0 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3845.2 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3467.2 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3894.7 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3691.5 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3487.3 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3844.7 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3693.2 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3450.1 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3876.7 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3637.0 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3480.5 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4077.5 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3956.0 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3579.8 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3988.4 | Standard polar | 33892256 |
| Glutathione,4TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3823.0 | Semi standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3560.9 | Standard non polar | 33892256 |
| Glutathione,4TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3961.9 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4153.4 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3704.0 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3793.9 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3980.1 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3721.3 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O | 3808.5 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4029.6 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3743.4 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #11 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3899.6 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4146.7 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3739.8 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #12 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3792.8 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4136.1 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3726.9 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #13 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3779.5 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3948.1 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3703.1 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #14 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3764.5 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4019.5 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3724.3 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #15 | CC(C)(C)[Si](C)(C)OC(=O)[C@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3891.8 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4113.1 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3774.0 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #16 | CC(C)(C)[Si](C)(C)SC[C@@H](C(=O)N(CC(=O)O)[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3828.7 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3984.5 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3684.0 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)NCC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3745.7 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3963.0 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3680.6 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N[C@@H](CS[Si](C)(C)C(C)(C)C)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3759.9 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3936.8 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3704.3 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@H](N)C(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4104.5 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4049.2 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3688.5 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS)NC(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3877.9 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4053.9 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3723.9 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS)N(C(=O)CC[C@@H](C(=O)O[Si](C)(C)C(C)(C)C)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3846.0 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3842.9 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3694.9 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)N[C@@H](CCC(=O)N([C@@H](CS)C(=O)N(CC(=O)O[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 3837.8 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4146.8 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3744.8 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)OC(=O)CN(C(=O)[C@H](CS[Si](C)(C)C(C)(C)C)NC(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3798.4 | Standard polar | 33892256 |
| Glutathione,5TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 4157.3 | Semi standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3749.7 | Standard non polar | 33892256 |
| Glutathione,5TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)OC(=O)CNC(=O)[C@H](CS[Si](C)(C)C(C)(C)C)N(C(=O)CC[C@@H](C(=O)O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 3792.6 | Standard polar | 33892256 |