| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2021-11-15 21:04:28 UTC |
|---|
| HMDB ID | HMDB0000169 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | D-Mannose |
|---|
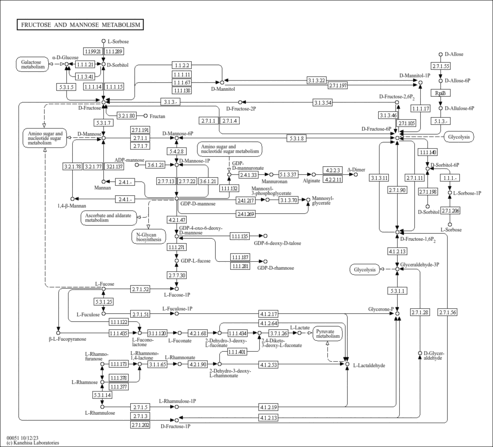
| Description | D-Mannose (also called Mannose or D-mannopyranose) is a hexose or a six-carbon sugar. It is also classified as an aldohexose. It is fermentable monosaccharide and an isomer of glucose. Mannose commonly exists as two different-sized rings, the pyranose (six-membered) form and the furanose (five-membered) form. Formally, D-Mannose is the 2-epimer of glucose and exists primarily as sweet-tasting alpha- (67%) or as a bitter-tasting beta- (33%) anomer of the pyranose form (PMID: 24931670 ). Mannose is not an essential nutrient, meaning that it can be produced in the human body from glucose or converted into glucose. Mannose is ~5x as active as glucose in non-enzyamtic glycation, which may explain why evolution did not favor it as a biological energy source (PMID: 24931670 ). Mannose occurs in microbes, plants and animals. Free mannose is found in small amounts in many fruits such as oranges, apples and peaches and in mammalian plasma at 50-100 uM (PMID: 24931670 ). More often, mannose occurs in homo-or hetero-polymers such as yeast mannans (alpha-mannose) where it can account for nearly 16% of dry weight or in galactomannans. Coffee beans, fenugreek and guar gums are rich sources of galactomannans. However, these plant polysaccharides are not degraded in the mammalian GI tract and, therefore, provide very little bio-available mannose for glycan synthesis. The digestion of many polysaccharides and glycoproteins also yields mannose. Once mannose is released, it is phosphorylated by hexokinase to generate mannose-6-phosphate. Mannose-6-phosphate is then converted to fructose-6-phosphate, by the enzyme phosphomannose isomerase, whereupon it enters the glycolytic pathway or is converted to glucose-6-phosphate by the gluconeogenic pathway. Mannose is a dominant monosaccharide in N-linked glycosylation, which is a post-translational modification of proteins. N-linked glycosylation is initiated by the transfer of Glc3Man9GlcNAc2 to nascent glycoproteins in the endoplasmic reticulum in a co-translational manner as the protein enters the transport system. Typically, mature human glycoproteins only contain three mannose residues buried under sequential modification by GlcNAc, galactose, and sialic acid. High-mannose-type oligosaccharides have been shown to play important roles in protein quality control. Several intracellular proteins such as lectins, chaperones, and glycan-processing enzymes, are involved in this process. These include calnexin/calreticulin, UDP-glucose:glycoprotein glucosyltransferase (UGGT), cargo receptors (such as VIP36 and ERGIC-53), mannosidase-like proteins (e.g. EDEM and Htm1p) and ubiquitin ligase (Fbs). They are thought to recognize high-mannose-type glycans with subtly different structures. Mannose-binding lectin (MBL) is an important constituent of the innate immune system. This protein binds through multiple lectin domains to the repeating sugar arrays that decorate many microbial surfaces and is then able to activate the complement system through a specific protease called MBL-associated protease-2. Mannose (D-mannose) is used as a nutritional supplement, packaged as "D-mannose", to prevent recurrent urinary tract infections (PMID: 21105658 ). D-mannose prevents FimH-mediated bacterial adhesion in the urinary tract through a competitive inhibition mechanism. This mechanism is based on the structural similarity between D-mannose and urothelial mannosylated receptors exposed by the epithelium of the urinary tract (PMID: 21105658 ). When D-mannose is administered in sufficient amounts, it is rapidly absorbed and then excreted by the urinary tract where it saturates bacterial FimH, thereby preventing bacterial binding to urothelial cells. |
|---|
| Structure | OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O)[C@@H]1O InChI=1S/C6H12O6/c7-1-2-3(8)4(9)5(10)6(11)12-2/h2-11H,1H2/t2-,3-,4+,5+,6+/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| alpha-D-Man | ChEBI | | a-D-Man | Generator | | α-D-Man | Generator | | a-D-Mannose | Generator | | α-D-Mannose | Generator | | alpha-D-Mannopyranose | ChEBI | | a-D-Mannopyranose | Generator | | α-D-Mannopyranose | Generator | | (+)-Mannose | HMDB | | Carubinose | HMDB | | D(+)-Mannose | HMDB | | Mannose | HMDB | | Seminose | HMDB | | alpha-D-Mannose | HMDB | | alpha-Mannose | HMDB | | α-Mannose | HMDB |
|
|---|
| Chemical Formula | C6H12O6 |
|---|
| Average Molecular Weight | 180.1559 |
|---|
| Monoisotopic Molecular Weight | 180.063388116 |
|---|
| IUPAC Name | (2S,3S,4S,5S,6R)-6-(hydroxymethyl)oxane-2,3,4,5-tetrol |
|---|
| Traditional Name | α-D-mannose |
|---|
| CAS Registry Number | 3458-28-4 |
|---|
| SMILES | OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O)[C@@H]1O |
|---|
| InChI Identifier | InChI=1S/C6H12O6/c7-1-2-3(8)4(9)5(10)6(11)12-2/h2-11H,1H2/t2-,3-,4+,5+,6+/m1/s1 |
|---|
| InChI Key | WQZGKKKJIJFFOK-PQMKYFCFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as hexoses. These are monosaccharides in which the sugar unit is a is a six-carbon containing moeity. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Hexoses |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hexose monosaccharide
- Oxane
- Secondary alcohol
- Hemiacetal
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Hydrocarbon derivative
- Primary alcohol
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | 132 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 713 mg/mL at 17 °C | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections| Adduct Type | Data Source | CCS Value (Å2) | Reference |
|---|
| [M-H]- | Astarita_neg | 130.3 | 30932474 |
|
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 0.47 minutes | 32390414 | | Predicted by Siyang on May 30, 2022 | 9.6866 minutes | 33406817 | | Predicted by Siyang using ReTip algorithm on June 8, 2022 | 6.01 minutes | 32390414 | | AjsUoB = Accucore 150 Amide HILIC with 10mM Ammonium Formate, 0.1% Formic Acid | 302.0 seconds | 40023050 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 634.4 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 309.4 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 33.2 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 183.4 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 76.7 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 293.2 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 226.3 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 703.6 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 577.8 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 42.0 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 785.4 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 196.6 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 260.1 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 650.8 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 366.5 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 323.2 seconds | 40023050 |
Predicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| D-Mannose,1TMS,isomer #1 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O)[C@@H]1O | 1738.4 | Semi standard non polar | 33892256 | | D-Mannose,1TMS,isomer #2 | C[Si](C)(C)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1O | 1673.8 | Semi standard non polar | 33892256 | | D-Mannose,1TMS,isomer #3 | C[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O | 1683.6 | Semi standard non polar | 33892256 | | D-Mannose,1TMS,isomer #4 | C[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](O)O[C@H](CO)[C@H]1O | 1688.3 | Semi standard non polar | 33892256 | | D-Mannose,1TMS,isomer #5 | C[Si](C)(C)O[C@@H]1[C@@H](CO)O[C@H](O)[C@@H](O)[C@H]1O | 1678.0 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #1 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O)[C@@H](O)[C@@H]1O | 1722.4 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #10 | C[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](O)O[C@H](CO)[C@H]1O[Si](C)(C)C | 1681.6 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #2 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C)[C@@H](O)[C@@H]1O | 1737.0 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #3 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O[Si](C)(C)C)[C@@H]1O | 1742.6 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #4 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O)[C@@H]1O[Si](C)(C)C | 1712.2 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #5 | C[Si](C)(C)O[C@@H]1[C@@H](CO)O[C@H](O[Si](C)(C)C)[C@@H](O)[C@H]1O | 1701.4 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #6 | C[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](O[Si](C)(C)C)O[C@H](CO)[C@H]1O | 1703.5 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #7 | C[Si](C)(C)O[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[Si](C)(C)C | 1679.6 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #8 | C[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O[Si](C)(C)C)[C@@H]1O | 1697.1 | Semi standard non polar | 33892256 | | D-Mannose,2TMS,isomer #9 | C[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[Si](C)(C)C | 1692.2 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #1 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H](O)[C@@H]1O | 1703.6 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #10 | C[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O[Si](C)(C)C)[C@@H]1O[Si](C)(C)C | 1736.7 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #2 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O)[C@@H](O[Si](C)(C)C)[C@@H]1O | 1753.4 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #3 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O)[C@@H](O)[C@@H]1O[Si](C)(C)C | 1717.5 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #4 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H]1O | 1748.2 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #5 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C)[C@@H](O)[C@@H]1O[Si](C)(C)C | 1766.0 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #6 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O[Si](C)(C)C)[C@@H]1O[Si](C)(C)C | 1701.0 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #7 | C[Si](C)(C)O[C@H]1[C@H](O)[C@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)O[C@@H]1CO | 1733.4 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #8 | C[Si](C)(C)O[C@H]1O[C@H](CO)[C@@H](O[Si](C)(C)C)[C@H](O[Si](C)(C)C)[C@@H]1O | 1739.0 | Semi standard non polar | 33892256 | | D-Mannose,3TMS,isomer #9 | C[Si](C)(C)O[C@H]1[C@@H](O[Si](C)(C)C)[C@H](O)[C@@H](CO)O[C@@H]1O[Si](C)(C)C | 1725.4 | Semi standard non polar | 33892256 | | D-Mannose,4TMS,isomer #1 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H]1O | 1815.5 | Semi standard non polar | 33892256 | | D-Mannose,4TMS,isomer #2 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H](O)[C@@H]1O[Si](C)(C)C | 1815.2 | Semi standard non polar | 33892256 | | D-Mannose,4TMS,isomer #3 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O)[C@@H](O[Si](C)(C)C)[C@@H]1O[Si](C)(C)C | 1820.5 | Semi standard non polar | 33892256 | | D-Mannose,4TMS,isomer #4 | C[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H]1O[Si](C)(C)C | 1819.4 | Semi standard non polar | 33892256 | | D-Mannose,4TMS,isomer #5 | C[Si](C)(C)O[C@H]1[C@@H](O[Si](C)(C)C)[C@H](O[Si](C)(C)C)[C@@H](CO)O[C@@H]1O[Si](C)(C)C | 1800.9 | Semi standard non polar | 33892256 | | D-Mannose,5TMS,isomer #1 | C[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H](O[Si](C)(C)C)[C@@H]1O[Si](C)(C)C | 1877.4 | Semi standard non polar | 33892256 | | D-Mannose,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O)[C@@H]1O | 2002.6 | Semi standard non polar | 33892256 | | D-Mannose,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1O | 1940.1 | Semi standard non polar | 33892256 | | D-Mannose,1TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O | 1940.5 | Semi standard non polar | 33892256 | | D-Mannose,1TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](O)O[C@H](CO)[C@H]1O | 1947.1 | Semi standard non polar | 33892256 | | D-Mannose,1TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](CO)O[C@H](O)[C@@H](O)[C@H]1O | 1943.8 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H](O)[C@@H]1O | 2208.7 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](O)O[C@H](CO)[C@H]1O[Si](C)(C)C(C)(C)C | 2181.6 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H]1O | 2210.9 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O | 2219.8 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O)[C@@H]1O[Si](C)(C)C(C)(C)C | 2197.5 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](CO)O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@H]1O | 2193.9 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)O[C@H](CO)[C@H]1O | 2195.0 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)O[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[Si](C)(C)C(C)(C)C | 2180.7 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O | 2190.8 | Semi standard non polar | 33892256 | | D-Mannose,2TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[Si](C)(C)C(C)(C)C | 2192.8 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H]1O | 2441.4 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #10 | CC(C)(C)[Si](C)(C)O[C@@H]1[C@@H](O)O[C@H](CO)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O[Si](C)(C)C(C)(C)C | 2455.2 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O | 2449.6 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H](O)[C@@H]1O[Si](C)(C)C(C)(C)C | 2445.1 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O | 2466.7 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H]1O[Si](C)(C)C(C)(C)C | 2468.7 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O[Si](C)(C)C(C)(C)C | 2439.7 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #7 | CC(C)(C)[Si](C)(C)O[C@H]1[C@H](O)[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)O[C@@H]1CO | 2451.3 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #8 | CC(C)(C)[Si](C)(C)O[C@H]1O[C@H](CO)[C@@H](O[Si](C)(C)C(C)(C)C)[C@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O | 2451.2 | Semi standard non polar | 33892256 | | D-Mannose,3TBDMS,isomer #9 | CC(C)(C)[Si](C)(C)O[C@H]1[C@@H](O[Si](C)(C)C(C)(C)C)[C@H](O)[C@@H](CO)O[C@@H]1O[Si](C)(C)C(C)(C)C | 2454.9 | Semi standard non polar | 33892256 | | D-Mannose,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O | 2671.4 | Semi standard non polar | 33892256 | | D-Mannose,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H]1O[Si](C)(C)C(C)(C)C | 2668.4 | Semi standard non polar | 33892256 | | D-Mannose,4TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O[Si](C)(C)C(C)(C)C | 2664.4 | Semi standard non polar | 33892256 | | D-Mannose,4TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O[Si](C)(C)C(C)(C)C | 2681.0 | Semi standard non polar | 33892256 | | D-Mannose,4TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)O[C@H]1[C@@H](O[Si](C)(C)C(C)(C)C)[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](CO)O[C@@H]1O[Si](C)(C)C(C)(C)C | 2669.4 | Semi standard non polar | 33892256 | | D-Mannose,5TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC[C@H]1O[C@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H](O[Si](C)(C)C(C)(C)C)[C@@H]1O[Si](C)(C)C(C)(C)C | 2890.3 | Semi standard non polar | 33892256 |
|
|---|
| General References | - Wu X, Rush JS, Karaoglu D, Krasnewich D, Lubinsky MS, Waechter CJ, Gilmore R, Freeze HH: Deficiency of UDP-GlcNAc:Dolichol Phosphate N-Acetylglucosamine-1 Phosphate Transferase (DPAGT1) causes a novel congenital disorder of Glycosylation Type Ij. Hum Mutat. 2003 Aug;22(2):144-50. [PubMed:12872255 ]
- Thio CL, Mosbruger T, Astemborski J, Greer S, Kirk GD, O'Brien SJ, Thomas DL: Mannose binding lectin genotypes influence recovery from hepatitis B virus infection. J Virol. 2005 Jul;79(14):9192-6. [PubMed:15994813 ]
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [PubMed:19212411 ]
- Lee SH, Nam SY, Chung BC: Altered profile of endogenous steroids in the urine of patients with prolactinoma. Clin Biochem. 1998 Oct;31(7):529-35. [PubMed:9812172 ]
- Shoemaker JD, Elliott WH: Automated screening of urine samples for carbohydrates, organic and amino acids after treatment with urease. J Chromatogr. 1991 Jan 2;562(1-2):125-38. [PubMed:2026685 ]
- Alton G, Hasilik M, Niehues R, Panneerselvam K, Etchison JR, Fana F, Freeze HH: Direct utilization of mannose for mammalian glycoprotein biosynthesis. Glycobiology. 1998 Mar;8(3):285-95. [PubMed:9451038 ]
- Nakagawa F, Schulte BA, Spicer SS: Lectin cytochemical evaluation of somatosensory neurons and their peripheral and central processes in rat and man. Cell Tissue Res. 1986;245(3):579-89. [PubMed:3757018 ]
- Otter M, Zockova P, Kuiper J, van Berkel TJ, Barrett-Bergshoeff MM, Rijken DC: Isolation and characterization of the mannose receptor from human liver potentially involved in the plasma clearance of tissue-type plasminogen activator. Hepatology. 1992 Jul;16(1):54-9. [PubMed:1618483 ]
- Hu P, Parenti G, Keulemans J, Hoogeveen AT: Lysosomal tartrate sensitive acid phosphatase deficiency in cells which contain lysosomal "high uptake forms". Biochem Biophys Res Commun. 1990 Mar 16;167(2):520-7. [PubMed:2322240 ]
- Hermentin P, Witzel R, Kanzy EJ, Diderrich G, Hoffmann D, Metzner H, Vorlop J, Haupt H: The hypothetical N-glycan charge: a number that characterizes protein glycosylation. Glycobiology. 1996 Mar;6(2):217-30. [PubMed:8727793 ]
- DeRossi C, Bode L, Eklund EA, Zhang F, Davis JA, Westphal V, Wang L, Borowsky AD, Freeze HH: Ablation of mouse phosphomannose isomerase (Mpi) causes mannose 6-phosphate accumulation, toxicity, and embryonic lethality. J Biol Chem. 2006 Mar 3;281(9):5916-27. Epub 2005 Dec 8. [PubMed:16339137 ]
- Miras MT, Aunis D, Mandel P: Studies on the interaction of dopamine beta-hydroxylase from various sources with phytohaemagglutinins. Clin Chim Acta. 1975 Nov 3;64(3):293-302. [PubMed:1183043 ]
- Van der Ploeg AT, Kroos MA, Willemsen R, Brons NH, Reuser AJ: Intravenous administration of phosphorylated acid alpha-glucosidase leads to uptake of enzyme in heart and skeletal muscle of mice. J Clin Invest. 1991 Feb;87(2):513-8. [PubMed:1991835 ]
- Cavallone D, Malagolini N, Monti A, Wu XR, Serafini-Cessi F: Variation of high mannose chains of Tamm-Horsfall glycoprotein confers differential binding to type 1-fimbriated Escherichia coli. J Biol Chem. 2004 Jan 2;279(1):216-22. Epub 2003 Oct 21. [PubMed:14570881 ]
- Adlerberth I, Ahrne S, Johansson ML, Molin G, Hanson LA, Wold AE: A mannose-specific adherence mechanism in Lactobacillus plantarum conferring binding to the human colonic cell line HT-29. Appl Environ Microbiol. 1996 Jul;62(7):2244-51. [PubMed:8779562 ]
- Hung CS, Bouckaert J, Hung D, Pinkner J, Widberg C, DeFusco A, Auguste CG, Strouse R, Langermann S, Waksman G, Hultgren SJ: Structural basis of tropism of Escherichia coli to the bladder during urinary tract infection. Mol Microbiol. 2002 May;44(4):903-15. [PubMed:12010488 ]
- Condaminet B, Peguet-Navarro J, Stahl PD, Dalbiez-Gauthier C, Schmitt D, Berthier-Vergnes O: Human epidermal Langerhans cells express the mannose-fucose binding receptor. Eur J Immunol. 1998 Nov;28(11):3541-51. [PubMed:9842897 ]
- Go S, Sato C, Furuhata K, Kitajima K: Oral ingestion of mannose alters the expression level of deaminoneuraminic acid (KDN) in mouse organs. Glycoconj J. 2006 Jul;23(5-6):411-21. [PubMed:16897182 ]
- Burk MR, Troeger C, Brinkhaus R, Holzgreve W, Hahn S: Severely reduced presence of tissue macrophages in the basal plate of pre-eclamptic placentae. Placenta. 2001 Apr;22(4):309-16. [PubMed:11286566 ]
- Sarkar K, Das PK: Protective effect of neoglycoprotein-conjugated muramyl dipeptide against Leishmania donovani infection: the role of cytokines. J Immunol. 1997 Jun 1;158(11):5357-65. [PubMed:9164956 ]
- Kusmierz J, DeGeorge JJ, Sweeney D, May C, Rapoport SI: Quantitative analysis of polyols in human plasma and cerebrospinal fluid. J Chromatogr. 1989 Dec 29;497:39-48. [PubMed:2625478 ]
- Biessen EA, Noorman F, van Teijlingen ME, Kuiper J, Barrett-Bergshoeff M, Bijsterbosch MK, Rijken DC, van Berkel TJ: Lysine-based cluster mannosides that inhibit ligand binding to the human mannose receptor at nanomolar concentration. J Biol Chem. 1996 Nov 8;271(45):28024-30. [PubMed:8910412 ]
- Smith ME: Phagocytic properties of microglia in vitro: implications for a role in multiple sclerosis and EAE. Microsc Res Tech. 2001 Jul 15;54(2):81-94. [PubMed:11455615 ]
- Della Porta M, Danova M, Rigolin GM, Brugnatelli S, Rovati B, Tronconi C, Fraulini C, Russo Rossi A, Riccardi A, Castoldi G: Dendritic cells and vascular endothelial growth factor in colorectal cancer: correlations with clinicobiological findings. Oncology. 2005;68(2-3):276-84. Epub 2005 Jul 7. [PubMed:16015045 ]
- Patnaik SK, Stanley P: Mouse large can modify complex N- and mucin O-glycans on alpha-dystroglycan to induce laminin binding. J Biol Chem. 2005 May 27;280(21):20851-9. Epub 2005 Mar 23. [PubMed:15788414 ]
- Brysk MM, Miller J: Concanavalin A binding glycoprotein in human stratum corneum. J Invest Dermatol. 1984 Mar;82(3):280-2. [PubMed:6421939 ]
- Takahashi I, Takahashi T, Mikami T, Komatsu M, Ohura T, Schuchman EH, Takada G: Acid sphingomyelinase: relation of 93lysine residue on the ratio of intracellular to secreted enzyme activity. Tohoku J Exp Med. 2005 Aug;206(4):333-40. [PubMed:15997205 ]
- Akazawa S, Metzger BE, Freinkel N: Relationships between glucose and mannose during late gestation in normal pregnancy and pregnancy complicated by diabetes mellitus: concurrent concentrations in maternal plasma and amniotic fluid. J Clin Endocrinol Metab. 1986 May;62(5):984-9. [PubMed:3958133 ]
- Blach-Olszewska Z: Innate immunity: cells, receptors, and signaling pathways. Arch Immunol Ther Exp (Warsz). 2005 May-Jun;53(3):245-53. [PubMed:15995585 ]
- Pietila EM, Tuusa JT, Apaja PM, Aatsinki JT, Hakalahti AE, Rajaniemi HJ, Petaja-Repo UE: Inefficient maturation of the rat luteinizing hormone receptor. A putative way to regulate receptor numbers at the cell surface. J Biol Chem. 2005 Jul 15;280(28):26622-9. Epub 2005 May 18. [PubMed:15901736 ]
- Carchon HA, Jaeken J: Determination of D-mannose in serum by capillary electrophoresis. Clin Chem. 2001;47(7):1319-21. [PubMed:11427470 ]
- Park SH, Pastuszak I, Drake R, Elbein AD: Purification to apparent homogeneity and properties of pig kidney L-fucose kinase. J Biol Chem. 1998 Mar 6;273(10):5685-91. [PubMed:9488699 ]
- Ito Y, Hagihara S, Matsuo I, Totani K: Structural approaches to the study of oligosaccharides in glycoprotein quality control. Curr Opin Struct Biol. 2005 Oct;15(5):481-9. [PubMed:16154739 ]
- Jack DL, Klein NJ, Turner MW: Mannose-binding lectin: targeting the microbial world for complement attack and opsonophagocytosis. Immunol Rev. 2001 Apr;180:86-99. [PubMed:11414367 ]
- Elshenawy S, Pinney SE, Stuart T, Doulias PT, Zura G, Parry S, Elovitz MA, Bennett MJ, Bansal A, Strauss JF 3rd, Ischiropoulos H, Simmons RA: The Metabolomic Signature of the Placenta in Spontaneous Preterm Birth. Int J Mol Sci. 2020 Feb 4;21(3). pii: ijms21031043. doi: 10.3390/ijms21031043. [PubMed:32033212 ]
- Sharma V, Ichikawa M, Freeze HH: Mannose metabolism: more than meets the eye. Biochem Biophys Res Commun. 2014 Oct 17;453(2):220-8. doi: 10.1016/j.bbrc.2014.06.021. Epub 2014 Jun 12. [PubMed:24931670 ]
- Klein T, Abgottspon D, Wittwer M, Rabbani S, Herold J, Jiang X, Kleeb S, Luthi C, Scharenberg M, Bezencon J, Gubler E, Pang L, Smiesko M, Cutting B, Schwardt O, Ernst B: FimH antagonists for the oral treatment of urinary tract infections: from design and synthesis to in vitro and in vivo evaluation. J Med Chem. 2010 Dec 23;53(24):8627-41. doi: 10.1021/jm101011y. Epub 2010 Nov 24. [PubMed:21105658 ]
|
|---|