| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2006-07-26 10:28:39 UTC |
|---|
| Update Date | 2021-09-14 15:42:00 UTC |
|---|
| HMDB ID | HMDB0003325 |
|---|
| Secondary Accession Numbers | - HMDB0006518
- HMDB03325
- HMDB06518
|
|---|
| Metabolite Identification |
|---|
| Common Name | Bilirubin diglucuronide |
|---|
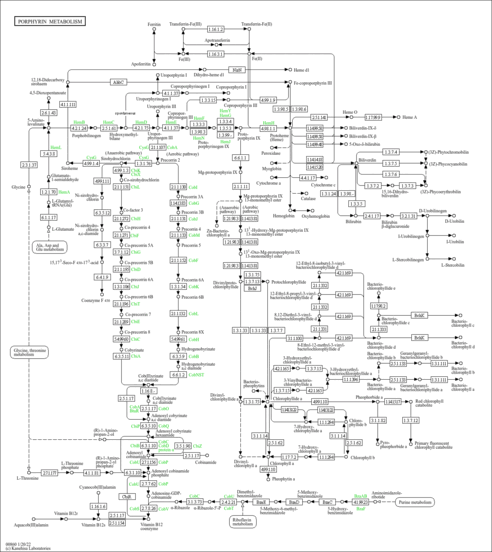
| Description | Bilirubin diglucuronide is a glucuronidated version of bilirubin, a tetrapyrrole compound produced via heme degradation. Heme is the red pigment in haemoglobin and red blood cells (RBCs). RBCs have a life span of about 120 days. When the RBCs have reached the end of their useful lifespan, the cells are engulfed by macrophages and their constituents recycled or disposed of. Heme is broken down when the heme ring is opened by the enzyme known as heme oxygenase, which is found in the endoplasmic reticulum of the macrophages. The oxidation process produces the linear tetrapyrrole known as biliverdin along with ferric iron (Fe3+), and carbon monoxide (CO). In the next reaction, a second methylene group (located between rings III and IV of the porphyrin ring) is reduced by the enzyme known as biliverdin reductase, producing bilirubin. Bilirubin is significantly less extensively conjugated than biliverdin. This reduction causes a change in the color of the biliverdin molecule from blue-green (vert or verd for green) to yellow-red, which is the color of bilirubin (ruby or rubi for red). In plasma virtually all the bilirubin is tightly bound to plasma proteins, largely albumin, because it is only sparingly soluble in aqueous solutions at physiological pH. In the sinusoids unconjugated bilirubin dissociates from albumin, enters the liver cells across the cell membrane through non-ionic diffusion to the smooth endoplasmatic reticulum. In hepatocytes, bilirubin-UDP-glucuronyltransferase (bilirubin-UGT) adds 2 additional glucuronic acid molecules to bilirubin to produce the more water-soluble version of the molecule known as bilirubin diglucuronide. The bilirubin diglucuronide is transferred rapidly across the canalicular membrane into the bile canaliculi where it is then excreted as bile into the large intestine. |
|---|
| Structure | CC1=C(C=C)\C(NC1=O)=C\C1=C(C)C(CCC(=O)O[C@@H]2O[C@@H]([C@@H](O)[C@H](O)[C@H]2O)C(O)=O)=C(CC2=C(CCC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)C(C)=C(N2)\C=C2/NC(=O)C(C=C)=C2C)N1 InChI=1S/C45H52N4O18/c1-7-20-19(6)40(58)49-27(20)14-25-18(5)23(10-12-31(51)65-45-37(57)33(53)35(55)39(67-45)43(62)63)29(47-25)15-28-22(17(4)24(46-28)13-26-16(3)21(8-2)41(59)48-26)9-11-30(50)64-44-36(56)32(52)34(54)38(66-44)42(60)61/h7-8,13-14,32-39,44-47,52-57H,1-2,9-12,15H2,3-6H3,(H,48,59)(H,49,58)(H,60,61)(H,62,63)/b26-13-,27-14-/t32-,33-,34-,35-,36+,37+,38-,39-,44+,45+/m0/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| Bilirubin-bisglucuronoside | Kegg | | Bilirubin ixalpha diglucuronide | HMDB | | Bilirubin ixα diglucuronide | HMDB | | Bilirubin glucuronide | HMDB | | Bilirubin diglucuronide | MeSH |
|
|---|
| Chemical Formula | C45H52N4O18 |
|---|
| Average Molecular Weight | 936.921 |
|---|
| Monoisotopic Molecular Weight | 936.32766085 |
|---|
| IUPAC Name | (2S,3S,4S,5R,6S)-6-{[3-(2-{[3-(3-{[(2S,3R,4S,5S,6S)-6-carboxy-3,4,5-trihydroxyoxan-2-yl]oxy}-3-oxopropyl)-5-{[(2E)-3-ethenyl-5-hydroxy-4-methyl-2H-pyrrol-2-ylidene]methyl}-4-methyl-1H-pyrrol-2-yl]methyl}-5-{[(2E)-4-ethenyl-5-hydroxy-3-methyl-2H-pyrrol-2-ylidene]methyl}-4-methyl-1H-pyrrol-3-yl)propanoyl]oxy}-3,4,5-trihydroxyoxane-2-carboxylic acid |
|---|
| Traditional Name | (2S,3S,4S,5R,6S)-6-{[3-(2-{[3-(3-{[(2S,3R,4S,5S,6S)-6-carboxy-3,4,5-trihydroxyoxan-2-yl]oxy}-3-oxopropyl)-5-{[(2E)-3-ethenyl-5-hydroxy-4-methylpyrrol-2-ylidene]methyl}-4-methyl-1H-pyrrol-2-yl]methyl}-5-{[(2E)-4-ethenyl-5-hydroxy-3-methylpyrrol-2-ylidene]methyl}-4-methyl-1H-pyrrol-3-yl)propanoyl]oxy}-3,4,5-trihydroxyoxane-2-carboxylic acid |
|---|
| CAS Registry Number | 17459-92-6 |
|---|
| SMILES | CC1=C(C=C)\C(NC1=O)=C\C1=C(C)C(CCC(=O)O[C@@H]2O[C@@H]([C@@H](O)[C@H](O)[C@H]2O)C(O)=O)=C(CC2=C(CCC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)C(C)=C(N2)\C=C2/NC(=O)C(C=C)=C2C)N1 |
|---|
| InChI Identifier | InChI=1S/C45H52N4O18/c1-7-20-19(6)40(58)49-27(20)14-25-18(5)23(10-12-31(51)65-45-37(57)33(53)35(55)39(67-45)43(62)63)29(47-25)15-28-22(17(4)24(46-28)13-26-16(3)21(8-2)41(59)48-26)9-11-30(50)64-44-36(56)32(52)34(54)38(66-44)42(60)61/h7-8,13-14,32-39,44-47,52-57H,1-2,9-12,15H2,3-6H3,(H,48,59)(H,49,58)(H,60,61)(H,62,63)/b26-13-,27-14-/t32-,33-,34-,35-,36+,37+,38-,39-,44+,45+/m0/s1 |
|---|
| InChI Key | SCJLWMXOOYZBTH-BTVQFETGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as bilirubins. These are organic compounds containing a dicarboxylic acyclic tetrapyrrole derivative. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Tetrapyrroles and derivatives |
|---|
| Sub Class | Bilirubins |
|---|
| Direct Parent | Bilirubins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Bilirubin skeleton
- O-glucuronide
- 1-o-glucuronide
- Tetracarboxylic acid or derivatives
- Glucuronic acid or derivatives
- Beta-hydroxy acid
- Fatty acid ester
- Monosaccharide
- Oxane
- Pyran
- Substituted pyrrole
- Fatty acyl
- Hydroxy acid
- Pyrroline
- Pyrrole
- Heteroaromatic compound
- Secondary alcohol
- Secondary carboxylic acid amide
- Carboxylic acid ester
- Lactam
- Carboxamide group
- Acetal
- Carboxylic acid derivative
- Carboxylic acid
- Oxacycle
- Azacycle
- Polyol
- Organic oxide
- Alcohol
- Organopnictogen compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Organic oxygen compound
- Carbonyl group
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 4.48 minutes | 32390414 | | Predicted by Siyang on May 30, 2022 | 16.3318 minutes | 33406817 | | Predicted by Siyang using ReTip algorithm on June 8, 2022 | 5.22 minutes | 32390414 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 3364.7 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 161.0 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 167.8 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 169.1 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 135.3 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 466.4 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 714.9 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 198.0 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 1213.9 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 741.7 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 2227.1 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 442.6 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 461.2 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 316.2 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 186.9 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 37.4 seconds | 40023050 |
Predicted Kovats Retention IndicesNot Available |
|---|
| MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 10V, Positive-QTOF | splash10-00kf-0200030903-3f731d3255f730a58952 | 2019-02-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 20V, Positive-QTOF | splash10-016u-0300070902-a0595a946705461ddb24 | 2019-02-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 40V, Positive-QTOF | splash10-0059-2200094431-2cedb95f9845511ac143 | 2019-02-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 10V, Negative-QTOF | splash10-052f-0200025912-c488140c352914da3532 | 2019-02-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 20V, Negative-QTOF | splash10-00kf-2800020923-c9e3902c3b856732b595 | 2019-02-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 40V, Negative-QTOF | splash10-01r7-6900003740-e2aa55faca0003c1270f | 2019-02-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 10V, Positive-QTOF | splash10-014l-0010500539-05ec0afe18cb6d6a4983 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 20V, Positive-QTOF | splash10-014i-0230530967-386f3f3c90b3b8a6bb6c | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 40V, Positive-QTOF | splash10-0udr-3490340563-80e35a547643c4a2b391 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 10V, Negative-QTOF | splash10-0540-1720170916-65c2b0b7a27f92bc8cb7 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 20V, Negative-QTOF | splash10-0670-5350061975-523df2bd3cff46ef40a4 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Bilirubin diglucuronide 40V, Negative-QTOF | splash10-052r-5090120520-83d0638c5c3df842db5d | 2021-09-24 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum |
|
|---|