| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2020-04-23 20:56:37 UTC |
|---|
| HMDB ID | HMDB0000241 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Protoporphyrin IX |
|---|
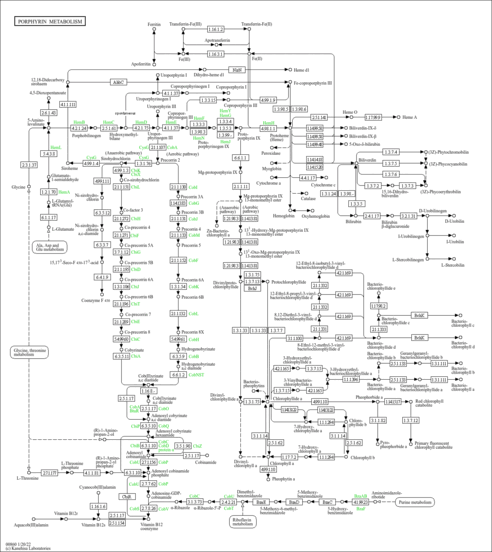
| Description | Protoporphyrins are tetrapyrroles containing 4 methyl, 2 propionic, and 2 vinyl side chains. Protoporphyrin is produced by oxidation of the methylene bridge of protoporphyrinogen. Protoporphyrin IX is the only naturally occurring isomer; it is an intermediate in heme biosynthesis, combining with ferrous iron to form protoheme IX, the heme prosthetic group of hemoglobin. Protoporphyrin IX is created by the enzyme protoporphyrinogen oxidase. The enzyme ferrochelatase converts it into heme. Protoporphyrin IX naturally occurs in small amounts in feces. Protoporphyrin IX is also responsible for the brown pigment (ooporphyrin) of birds' eggs. Protoporphyrin IX is used as a branch point in the biosynthetic pathway leading to heme (by insertion of iron) and chlorophylls (by insertion of Mg and further side-chain transformation). Protoporphyrin IX can be used to treat liver disorders, mainly as the sodium salt. Under certain conditions, protoporphyrin IX can act as a neurotoxin, a phototoxin, and a metabotoxin. A neurotoxin causes damage to nerve cells and nerve tissues. A phototoxin causes cell damage upon exposure to light. A metabotoxin is an endogenously produced metabolite that causes adverse health effects at chronically high levels. Chronically high levels of porphyrins are associated with porphyrias such as porphyria variegate, acute intermittent porphyria, and hereditary coproporphyria (HCP). In particular, it is accumulated and excreted excessively in the feces in acute intermittent porphyria, protoporphyria, and variegate porphyria. There are several types of porphyrias (most are inherited). Hepatic porphyrias are characterized by acute neurological attacks (seizures, psychosis, extreme back and abdominal pain, and an acute polyneuropathy), while the erythropoietic forms present with skin problems (usually a light-sensitive blistering rash and increased hair growth). The neurotoxicity of porphyrins may be due to their selective interactions with tubulin, which disrupt microtubule formation and cause neural malformations (PMID: 3441503 ). |
|---|
| Structure | CC1=C(CCC(O)=O)/C2=C/C3=N/C(=C\C4=C(C)C(C=C)=C(N4)/C=C4\N=C(\C=C\1/N\2)C(C=C)=C4C)/C(C)=C3CCC(O)=O InChI=1S/C34H34N4O4/c1-7-21-17(3)25-13-26-19(5)23(9-11-33(39)40)31(37-26)16-32-24(10-12-34(41)42)20(6)28(38-32)15-30-22(8-2)18(4)27(36-30)14-29(21)35-25/h7-8,13-16,35,38H,1-2,9-12H2,3-6H3,(H,39,40)(H,41,42)/b25-13-,26-13-,27-14-,28-15-,29-14-,30-15-,31-16-,32-16- |
|---|
| Synonyms | | Value | Source |
|---|
| 3,3'-(3,7,12,17-Tetramethyl-8,13-divinyl-21H,23H-porphine-2,18-diyl)-bis-propionic acid | ChEBI | | 3,7,12,17-Tetramethyl-8,13-divinylporphyrin-2,18-dipropanoic acid | ChEBI | | H2PpIX | ChEBI | | Kammerer's prophyrin | ChEBI | | Ooporphyrin | ChEBI | | Porphyrinogen IX | ChEBI | | 3,3'-(3,7,12,17-Tetramethyl-8,13-divinyl-21H,23H-porphine-2,18-diyl)-bis-propionate | Generator | | 3,7,12,17-Tetramethyl-8,13-divinylporphyrin-2,18-dipropanoate | Generator | | 3,3'-(3,7,12,17-Tetramethyl-8,13-divinylporphine-2,18-diyl)di | HMDB | | PpIX | HMDB | | Protoporphyrin | HMDB | | Protoporphyrin-"ix" | HMDB | | Protoporphyrin-IX | HMDB |
|
|---|
| Chemical Formula | C34H34N4O4 |
|---|
| Average Molecular Weight | 562.6582 |
|---|
| Monoisotopic Molecular Weight | 562.258005596 |
|---|
| IUPAC Name | 3-[20-(2-carboxyethyl)-9,14-diethenyl-5,10,15,19-tetramethyl-21,22,23,24-tetraazapentacyclo[16.2.1.1^{3,6}.1^{8,11}.1^{13,16}]tetracosa-1(21),2,4,6,8(23),9,11,13,15,17,19-undecaen-4-yl]propanoic acid |
|---|
| Traditional Name | 3-[20-(2-carboxyethyl)-9,14-diethenyl-5,10,15,19-tetramethyl-21,22,23,24-tetraazapentacyclo[16.2.1.1^{3,6}.1^{8,11}.1^{13,16}]tetracosa-1(21),2,4,6,8(23),9,11,13,15,17,19-undecaen-4-yl]propanoic acid |
|---|
| CAS Registry Number | 553-12-8 |
|---|
| SMILES | CC1=C(CCC(O)=O)/C2=C/C3=N/C(=C\C4=C(C)C(C=C)=C(N4)/C=C4\N=C(\C=C\1/N\2)C(C=C)=C4C)/C(C)=C3CCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C34H34N4O4/c1-7-21-17(3)25-13-26-19(5)23(9-11-33(39)40)31(37-26)16-32-24(10-12-34(41)42)20(6)28(38-32)15-30-22(8-2)18(4)27(36-30)14-29(21)35-25/h7-8,13-16,35,38H,1-2,9-12H2,3-6H3,(H,39,40)(H,41,42)/b25-13-,26-13-,27-14-,28-15-,29-14-,30-15-,31-16-,32-16- |
|---|
| InChI Key | KSFOVUSSGSKXFI-UJJXFSCMSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as porphyrins. Porphyrins are compounds containing a fundamental skeleton of four pyrrole nuclei united through the alpha-positions by four methine groups to form a macrocyclic structure. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Tetrapyrroles and derivatives |
|---|
| Sub Class | Porphyrins |
|---|
| Direct Parent | Porphyrins |
|---|
| Alternative Parents | Not Available |
|---|
| Substituents | Not Available |
|---|
| Molecular Framework | Not Available |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Physiological effect | |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | 300 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 0.17 mg/mL at 25 °C | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 7.58 minutes | 32390414 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 3027.8 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 483.0 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 262.3 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 234.2 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 584.8 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 1060.3 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 941.1 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 103.3 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 2171.2 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 758.2 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 2361.0 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 756.0 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 553.4 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 275.2 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 519.9 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 8.9 seconds | 40023050 |
Predicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| Protoporphyrin IX,1TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C)C(CCC(=O)O)=C4C)[NH]3 | 5372.5 | Semi standard non polar | 33892256 | | Protoporphyrin IX,1TMS,isomer #2 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O)C(CCC(=O)O[Si](C)(C)C)=C4C)[NH]3 | 5357.7 | Semi standard non polar | 33892256 | | Protoporphyrin IX,1TMS,isomer #3 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O)C(CCC(=O)O)=C4C)N3[Si](C)(C)C | 5429.1 | Semi standard non polar | 33892256 | | Protoporphyrin IX,1TMS,isomer #4 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O)=C4C)[NH]3 | 5403.2 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)[NH]3 | 5260.4 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TMS,isomer #2 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C)C(CCC(=O)O)=C4C)N3[Si](C)(C)C | 5350.2 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TMS,isomer #3 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O)=C4C)[NH]3 | 5328.0 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TMS,isomer #4 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 5332.6 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TMS,isomer #5 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)[NH]3 | 5309.9 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TMS,isomer #6 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O)=C4C)N3[Si](C)(C)C | 5368.9 | Semi standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 5237.9 | Semi standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 4475.5 | Standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 6148.1 | Standard polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #2 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)[NH]3 | 5213.5 | Semi standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #2 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)[NH]3 | 4461.0 | Standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #2 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)[NH]3 | 6237.9 | Standard polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #3 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O)=C4C)N3[Si](C)(C)C | 5282.5 | Semi standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #3 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O)=C4C)N3[Si](C)(C)C | 4484.5 | Standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #3 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O)=C4C)N3[Si](C)(C)C | 6111.8 | Standard polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #4 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 5277.2 | Semi standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #4 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 4480.4 | Standard non polar | 33892256 | | Protoporphyrin IX,3TMS,isomer #4 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 6123.2 | Standard polar | 33892256 | | Protoporphyrin IX,4TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 5182.6 | Semi standard non polar | 33892256 | | Protoporphyrin IX,4TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 4462.6 | Standard non polar | 33892256 | | Protoporphyrin IX,4TMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C)C(CCC(=O)O[Si](C)(C)C)=C4C)N3[Si](C)(C)C | 5851.9 | Standard polar | 33892256 | | Protoporphyrin IX,1TBDMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C(C)(C)C)C(CCC(=O)O)=C4C)[NH]3 | 5616.8 | Semi standard non polar | 33892256 | | Protoporphyrin IX,1TBDMS,isomer #2 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O)C(CCC(=O)O[Si](C)(C)C(C)(C)C)=C4C)[NH]3 | 5602.5 | Semi standard non polar | 33892256 | | Protoporphyrin IX,1TBDMS,isomer #3 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O)C(CCC(=O)O)=C4C)N3[Si](C)(C)C(C)(C)C | 5595.2 | Semi standard non polar | 33892256 | | Protoporphyrin IX,1TBDMS,isomer #4 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C(C)(C)C)C(CCC(=O)O)=C4C)[NH]3 | 5583.3 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TBDMS,isomer #1 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C(C)(C)C)C(CCC(=O)O[Si](C)(C)C(C)(C)C)=C4C)[NH]3 | 5648.2 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TBDMS,isomer #2 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O[Si](C)(C)C(C)(C)C)C(CCC(=O)O)=C4C)N3[Si](C)(C)C(C)(C)C | 5674.6 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TBDMS,isomer #3 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O[Si](C)(C)C(C)(C)C)=C(C)C(=CC1=N2)N5[Si](C)(C)C(C)(C)C)C(CCC(=O)O)=C4C)[NH]3 | 5664.1 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TBDMS,isomer #4 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5[NH]C(=CC1=N2)C(C)=C5CCC(=O)O)C(CCC(=O)O[Si](C)(C)C(C)(C)C)=C4C)N3[Si](C)(C)C(C)(C)C | 5666.9 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TBDMS,isomer #5 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C(C)(C)C)C(CCC(=O)O[Si](C)(C)C(C)(C)C)=C4C)[NH]3 | 5660.8 | Semi standard non polar | 33892256 | | Protoporphyrin IX,2TBDMS,isomer #6 | C=CC1=C(C)C2=CC3=C(C=C)C(C)=C(C=C4N=C(C=C5C(CCC(=O)O)=C(C)C(=CC1=N2)N5[Si](C)(C)C(C)(C)C)C(CCC(=O)O)=C4C)N3[Si](C)(C)C(C)(C)C | 5687.2 | Semi standard non polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (Non-derivatized) - 70eV, Positive | splash10-0uxu-1000290000-e22236fe3cca8eed1ecf | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (1 TMS) - 70eV, Positive | splash10-00di-6000094000-b9c12952e100c5144cab | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS ("Protoporphyrin IX,1TMS,#1" TMS) - 70eV, Positive | Not Available | 2021-10-14 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_1_1) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_1_3) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_1_4) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_2_1) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_2_2) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_2_3) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_2_4) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_2_5) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TMS_2_6) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_1_3) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_1_4) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_2_1) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_2_2) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_2_3) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_2_4) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_2_5) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Protoporphyrin IX GC-MS (TBDMS_2_6) - 70eV, Positive | Not Available | 2021-10-15 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 10V, Positive-QTOF | splash10-0002-0000090000-5d5be53410134c5719ab | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 20V, Positive-QTOF | splash10-00kb-1000590000-2d4157b9f73534d95a9c | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 40V, Positive-QTOF | splash10-0a4l-1000910000-07524a9291a1921dfafc | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 10V, Negative-QTOF | splash10-03di-0000090000-5df26c1cfb9a0b16a91a | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 20V, Negative-QTOF | splash10-0296-1000190000-2d4bcb49201fc10adc09 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 40V, Negative-QTOF | splash10-0a4l-9000230000-064fb944a966d08c06bf | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 10V, Negative-QTOF | splash10-03di-0000090000-87c1740ecd1a28f4a2d9 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 20V, Negative-QTOF | splash10-03xu-0000590000-99160b987bb7e62ab362 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 40V, Negative-QTOF | splash10-00di-0000900000-f2d9118441a9c8c47d79 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 10V, Positive-QTOF | splash10-03dj-0000090000-d428ac6dc2fc4794cc41 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 20V, Positive-QTOF | splash10-0j4j-0000090000-2b065966ec57bb97d511 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Protoporphyrin IX 40V, Positive-QTOF | splash10-0uk9-1000970000-b435817e4dc349e65ab3 | 2021-09-23 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-21 | Wishart Lab | View Spectrum |
|
|---|
| General References | - Messmann H, Knuchel R, Baumler W, Holstege A, Scholmerich J: Endoscopic fluorescence detection of dysplasia in patients with Barrett's esophagus, ulcerative colitis, or adenomatous polyps after 5-aminolevulinic acid-induced protoporphyrin IX sensitization. Gastrointest Endosc. 1999 Jan;49(1):97-101. [PubMed:9869731 ]
- Stankiewicz A, Lutz W, Krajewska B, Szulc B: [Plasma indicators of iron metabolism in persons occupationally exposed to organic solvents with normal and increased levels of protoporphyrin IX in erythrocytes]. Pol Tyg Lek. 1986 Jul 7;41(27):851-4. [PubMed:3763462 ]
- Stankiewicz A: [Erythrocyte protoporphyrin IX in occupational exposure to asbestos]. Med Pr. 1984;35(5):351-4. [PubMed:6530952 ]
- Sudworth CD, Stringer MR, Cruse-Sawyer JE, Brown SB: Fluorescence microspectroscopy technique for the study of intracellular protoporphyrin IX dynamics. Appl Spectrosc. 2003 Jun;57(6):682-8. [PubMed:14658702 ]
- Kufner G, Schlegel H, Jager R: A spectrophotometric micromethod for determining erythrocyte protoporphyrin-IX in whole blood or erythrocytes. Clin Chem Lab Med. 2005;43(2):183-91. [PubMed:15843214 ]
- Stankiewicz A: The concentration of protoporphyrin IX in workers occupationally exposed to lead. Mater Med Pol. 1989 Apr-Jun;21(2):100-2. [PubMed:2488459 ]
- Krieg RC, Fickweiler S, Wolfbeis OS, Knuechel R: Cell-type specific protoporphyrin IX metabolism in human bladder cancer in vitro. Photochem Photobiol. 2000 Aug;72(2):226-33. [PubMed:10946577 ]
- Bailey GG, Needham LL: Simultaneous quantification of erythrocyte zinc protoporphyrin and protoporphyrin IX by liquid chromatography. Clin Chem. 1986 Dec;32(12):2137-42. [PubMed:3779978 ]
- Chisolm J Jr, Brown DH: Micro-scale photofluorometric determination of "free erythrocyte pophyrin" (protoporphyrin IX). Clin Chem. 1975 Oct;21(11):1669-82. [PubMed:1164799 ]
- Casas A, Batlle AM, Butler AR, Robertson D, Brown EH, MacRobert A, Riley PA: Comparative effect of ALA derivatives on protoporphyrin IX production in human and rat skin organ cultures. Br J Cancer. 1999 Jul;80(10):1525-32. [PubMed:10408393 ]
- Smits T, Robles CA, van Erp PE, van de Kerkhof PC, Gerritsen MJ: Correlation between macroscopic fluorescence and protoporphyrin IX content in psoriasis and actinic keratosis following application of aminolevulinic acid. J Invest Dermatol. 2005 Oct;125(4):833-9. [PubMed:16185285 ]
- Bartosova J, Hrkal Z: Accumulation of protoporphyrin-IX (PpIX) in leukemic cell lines following induction by 5-aminolevulinic acid (ALA). Comp Biochem Physiol C Toxicol Pharmacol. 2000 Jul;126(3):245-52. [PubMed:11048674 ]
- Sakai T, Takeuchi Y, Ikeya Y, Araki T, Ushio K: [Automated HPLC method for determining zinc protoporphyrin IX and protoporphyrin IX in erythrocytes of workers exposed to lead]. Sangyo Igaku. 1988 Nov;30(6):467-74. [PubMed:3221502 ]
- Stankiewicz A, Lutz W, Szeszko A: [Protoporphyrin IX level in erythrocytes of persons with alcoholic liver cirrhosis]. Pol Tyg Lek. 1985 Jul 15;40(28):787-9. [PubMed:4059100 ]
- Star WM, Aalders MC, Sac A, Sterenborg HJ: Quantitative model calculation of the time-dependent protoporphyrin IX concentration in normal human epidermis after delivery of ALA by passive topical application or lontophoresis. Photochem Photobiol. 2002 Apr;75(4):424-32. [PubMed:12003134 ]
- von Beckerath M, Juzenas P, Ma LW, Iani V, Lofgren L, Moan J: The influence of UV exposure on 5-aminolevulinic acid-induced protoporphyrin IX production in skin. Photochem Photobiol. 2001 Dec;74(6):825-8. [PubMed:11783939 ]
- Gottsch JD, Graham CR Jr, Hairston RJ, Chen CH, Green WR, Stark WJ: Protoporphyrin IX photosensitization of corneal endothelium. Arch Ophthalmol. 1989 Oct;107(10):1497-500. [PubMed:2803100 ]
- Bissonnette R, Zeng H, McLean DI, Korbelik M, Lui H: Oral aminolevulinic acid induces protoporphyrin IX fluorescence in psoriatic plaques and peripheral blood cells. Photochem Photobiol. 2001 Aug;74(2):339-45. [PubMed:11547574 ]
- Rick K, Sroka R, Stepp H, Kriegmair M, Huber RM, Jacob K, Baumgartner R: Pharmacokinetics of 5-aminolevulinic acid-induced protoporphyrin IX in skin and blood. J Photochem Photobiol B. 1997 Oct;40(3):313-9. [PubMed:9372622 ]
- De Rosa FS, Marchetti JM, Thomazini JA, Tedesco AC, Bentley MV: A vehicle for photodynamic therapy of skin cancer: influence of dimethylsulphoxide on 5-aminolevulinic acid in vitro cutaneous permeation and in vivo protoporphyrin IX accumulation determined by confocal microscopy. J Control Release. 2000 Apr 3;65(3):359-66. [PubMed:10699294 ]
- Winkelman JW, Collins GH: Neurotoxicity of tetraphenylporphinesulfonate TPPS4 and its relation to photodynamic therapy. Photochem Photobiol. 1987 Nov;46(5):801-7. [PubMed:3441503 ]
|
|---|